Executive Summary
Prescription drugs play an ever-increasing role in modern medicine. New medications are improving health outcomes and quality of life, replacing surgery and other invasive treatments, and quickening recovery for patients who receive these treatments. As important as prescription drugs are, not everyone has access to them. The newest drugs are often the most expensive, and millions of Americans - especially elderly and disabled Medicare beneficiaries - have inadequate or no insurance coverage for drugs. Nearly a third of all Medicare beneficiaries have no financial protection for the costs of drugs, if they can obtain them at all. Many additional beneficiaries find themselves moving in and out of the protection provided by insurance over the course of a year.
Medicare has generally excluded coverage of outpatient prescription drugs, as was common in private health plans when the program was enacted in 1965. Since then drug coverage has become a standard feature of private insurance, and it has become clear that the omission of outpatient drug coverage represents a crucial gap in protection for the most vulnerable Medicare beneficiaries. As part of a broader plan to modernize Medicare, President Clinton has proposed a new, voluntary Medicare drug benefit that would offer all beneficiaries access to affordable, high-quality prescription drug coverage while maintaining the fiscal integrity of the program. In Congress, there has also been growing bipartisan interest in finding ways of extending drug coverage.
As policymakers consider options to ensure that every American can have access to innovative drug treatments, there is an urgent need for comprehensive and reliable information on drug coverage, drug spending, and drug prices. On October 25, 1999, the President directed the Secretary of Health and Human Services to study prescription drug costs and trends for Medicare beneficiaries. He asked that the study investigate:
- price differences for the most commonly used drugs for people with and without coverage;
- drug spending by people of various ages, as a percentage of income and of total health spending; and
- trends in drug expenditures by people of different ages, as a percentage of income and of total health spending.
This report is the Department's response to that request. It represents the work of individuals and agencies throughout the Department, including the Agency for Healthcare Research and Quality (AHRQ), the Food and Drug Administration (FDA), the Health Care Financing Administration (HCFA(now known as CMS)), and the Office of the Assistant Secretary for Planning and Evaluation (ASPE).
Chapter 1: Prescription Drug Coverage
While today, over 85 percent of Medicare beneficiaries use at least one prescription drug annually, beneficiaries must obtain drug coverage through a supplemental policy, by enrollment in a Medicare+Choice plan which includes coverage for prescription drugs, or through Medicaid. The result has been a patchwork of coverage that is not dependable, affordable, or accessible to all beneficiaries. Chapter 1 uses survey data to examine the sources of drug coverage for both the Medicare and non-Medicare population, describes the economic and demographic characteristics of those who have drug coverage and those who do not, and analyzes current trends in drug coverage. Analysis of data on the duration of coverage for the Medicare population is also presented. Differences in coverage rates by alternative measures of health status are explored. Lastly, trends in drug coverage for the Medicare and non-Medicare population are analyzed.
Key findings include:
- Only 53 percent of Medicare beneficiaries had drug coverage for the entire year of 1996, although 69 percent had coverage for at least one month during the year.
- Most sources of drug coverage are potentially unstable. Almost 48 percent of beneficiaries with drug coverage through Medigap and 29 percent who were covered through Medicare HMOs had drug coverage for only part of the year. Additionally, while employer-sponsored retiree coverage, the most prevalent single source of drug benefits, covered 32 percent of Medicare beneficiaries in 1996, 14 percent of those beneficiaries had only part year coverage from their former employers.
- Drug benefits are becoming less generous. There is considerable evidence that cost sharing for prescription drugs is increasing and that overall caps on coverage are both becoming more common and are being set at lower levels. For example, Medicare+Choice plans generally have reduced drug benefits and increased enrollee out-of-pocket costs in 2000. Eighty-six percent of plans have annual dollar limits on drugs, including 70 percent of plans with annual caps of $1000 or less, and 32 percent with caps of $500 or less per enrollee - levels that are up from 35 percent and 19 percent in 1998.
- Drug coverage is likely to decline as fewer employers offer health benefits to future retirees. For example, one employer survey recorded a drop from 40 percent in 1993 to 28 percent in 1999 in the number of large firms offering health benefits to Medicare eligible retirees. Additionally, employers have tightened eligibility rules and increased cost-shifting to retirees. Of those employers that still offer medical coverage, the survey found that 40 percent are requiring Medicare-eligible retirees to pay the full cost of their benefits, compared to 28 percent in 1995.
- Beneficiaries with incomes between 100 percent and 150 percent of poverty (that is, individuals age 65 or older with incomes between $7,527 and $11,287 in 1996) have the lowest rate of coverage. Although coverage varies by income, nearly one-fourth of beneficiaries with incomes over 400 percent of poverty lack coverage.
- Beneficiaries are less likely to have coverage if they are very old or live outside of a metropolitan area. About 37 percent of beneficiaries age 85 and above lacked coverage at any time during 1996 compared to 28 percent of beneficiaries age 65 through 69. About 43 percent of beneficiaries living in rural areas lacked drug coverage, compared to 27 percent of beneficiaries living in urban areas.
- Coverage rates vary little by self-reported health status, but are considerably higher for those with five or more chronic conditions. But by all measures, at least one-fourth of those in any category of health status lack coverage.
- Nearly one in four in the non-Medicare population never had any coverage for drugs in 1996. About 80 percent of those with full-year coverage got that coverage through employers.
Chapter 2: Effects of Prescription Drug Coverage on Spending and Utilization
Insurance coverage for prescription drugs makes a major difference in the amount of drugs people obtain, in how much they spend on drugs out of pocket, and in how much is spent in total on their behalf. People with coverage not only fill more prescriptions than those without coverage; they are likely to have access to a broader array of therapies, including more costly therapies. People without drug coverage face greater financial burdens and may sometimes be unable to follow the courses of treatment ordered by their physicians. There are even some indications that physicians themselves may recommend different therapies to people with and without coverage. Coverage increases prescription drug utilization, and reduces financial burdens for all population groups. However, access to drug coverage is most important for the elderly, simply because they require more medications, including a higher prevalence of long-term maintenance drugs for chronic conditions.
Chapter 2 presents detailed comparisons of utilization and spending (including out-of-pocket spending) for Medicare beneficiaries and the total population with and without drug coverage. It also examines some of the possible reasons for those differences and considers the consequences of being without coverage. Finally, it summarizes trends in utilization and spending and some of the factors that influence these changes.
Key findings include:
- Medicare beneficiaries with coverage fill nearly one-third more prescriptions than those without coverage.
- Although total drug spending for beneficiaries with coverage is nearly two-thirds higher, those without coverage pay nearly twice as much out of pocket ($463 versus $253).
- On average, beneficiaries with coverage pay out of pocket for about one-third of their total spending on drugs. However, the share of spending paid out of pocket varies by source of coverage, from 58 percent for those with Medigap coverage to 20 percent for those with Medicaid.
- Differences in utilization and spending between Medicare beneficiaries with and without drug coverage generally hold up across different income levels, ages, health status, and other categories.
- Drug insurance makes an especially large difference in dollar terms for those in the poorest health. Among beneficiaries with five or more chronic conditions, those with coverage had much higher total spending ($1,402 versus $944) and much lower out-of-pocket spending ($412 versus $944) than beneficiaries without coverage.
- Self-selection does not explain the difference in spending between Medicare beneficiaries with and without drug coverage. Even among beneficiaries with the same poor health status, more prescriptions are filled by people with coverage.
- Among people who are not Medicare beneficiaries, similar differences in utilization and spending exist between prescription drug users with and without coverage. Those with coverage for drugs fill two-thirds more prescriptions but spend a third less out of pocket than those without coverage.
- About a third of Medicare beneficiaries accounted for three-fourths of beneficiaries' total drug spending in 1996. Only 13 percent had no spending at all. Spending on prescription drugs in the non-Medicare population is even less evenly distributed.
- Prescription drugs take up about one-sixth of all health spending by the elderly. Out-of-pocket spending for prescription drugs is a larger proportion of health spending for the elderly than for younger people. Prescription drug spending also accounts for a larger share of spending by people with low incomes than it does for people with higher incomes.
- The burden of prescription drug costs creates access problems for some beneficiaries. Among Medicare beneficiaries, 10 percent of those with only Medicare coverage report not being able to afford a needed drug, compared to 2 percent of those with a non-Medicaid supplement.
- Drug spending has grown more quickly than other health spending throughout the 1990's. Price increases, higher utilization, and the use of newer, more expensive drugs all play a part in increasing drug spending.
Chapter 3: Prescription Drug Prices
In today's market for prescription drugs, most insurers obtain significant discounts on behalf of their insured beneficiaries. Individuals without coverage thus face not only the burden of paying for the entire cost of the drugs they need out of pocket, but they may also face higher prices for a given drug than do insurers and other large purchasers. Sorting out the differences in prices paid by those with and without coverage is not simple. The process by which prescription drug prices are determined is highly complex, involving numerous interactions and arrangements among manufacturers, wholesalers, retailers, insurers, pharmacy benefit managers (PBMs), and consumers.
In order to explain the complexity of this market, Chapter 3 begins with a description of the distribution channels for prescription drugs and how prices are established for different purchasers. It then offers an empirical analysis of whether prices paid for drugs at the retail level differ between cash customers and those with insurance coverage, using data from two sources: the Medical Expenditure Panel Survey (MEPS) and a widely used private sector data source on drug prices, IMS Health. A key limitation on the analysis of drug prices in this study, however, is our inability to incorporate the effect of rebates provided by manufacturers to insurers and PBMs. Given the greater market leverage of third party payers relative to individual consumers, it might be expected that cash customers will pay more than insurers for the same drugs at the retail pharmacy. Results from both sources, despite the absence of rebate data, support this hypothesis.
Key findings include:
- At the retail pharmacy level:
Individuals without drug coverage pay a higher price at the retail pharmacy than the total price paid on behalf of those with drug coverage (based on analysis of MEPS data that do not include rebates but look across all drug purchases holding drug type, form, strength, and quantity constant). The differences generally held up when examining the Medicare and non-Medicare populations.
Cash customers (including those without coverage and those with indemnity coverage) pay more for a given drug than those with third party payments at the point of sale (based on IMS Health data for over 90 percent of the most commonly prescribed drugs). In 1999, excluding the effect of rebates, the typical cash customer paid nearly 15 percent more than the customer with third party coverage. For a quarter of the most common drugs, the price difference between cash and third parties was even higher - over 20 percent. For the most commonly prescribed drugs, the price difference between cash customers and those with third party coverage grew substantially larger between 1996 and 1999.
The pattern of differences in the price paid by cash customers and those with third party payments is different for generic and brand name drugs (based on both MEPS and IMS Health data). Percentage differences in the price paid are often smaller for brand name drugs, but absolute differences may be larger because average prices for brand name drugs are considerably higher.
- Data on manufacturer rebates, if available, would reduce the total amount paid by the insurer or PBM on behalf of insured customers, increasing the difference in the total net price. Data on rebate arrangements, however, are confidential and unavailable to this study. In some instances, the amount of the rebate may be significantly more than the price differences observed at the retail pharmacy level. In other cases, the rebates may add only modestly to the observed differences.
- Various sources produce estimates of rebates ranging from 2 percent to 35 percent of drug sales prices. These rebates are not reflected in retail prices, but are instead paid directly to insurers and other organizations that manage drug benefits after they have already reimbursed the pharmacy.
This study presents a detailed examination of multiple factors relating to coverage, utilization, and spending for prescription drugs, particularly by the Medicare population. It also raises a variety of issues that are ripe for further investigation. Suggestive relationships between demographic factors, insurance status, and prescription drug use were revealed. However, we were unable to examine the more complex interrelationships among these factors. Future multivariate analyses will allow us to come to a more nuanced understanding of these relationships. Future research should explore what can be learned from using more sophisticated definitions of drug coverage status and severity of illness than were available for this study. In addition, if more data were available on elements of manufacturer pricing, such as rebates, further research could probe more fully the differences in prices paid by different customers. Finally, ongoing analyses will allow us to continue to use the most recent data - rapid change in the pharmaceutical market requires that analyses be refreshed and updated on a continuing basis. Some possible avenues for future research are explored at the conclusion of this report.
Introduction
Prescription drugs play an ever-increasing role in modern medicine. New medications are improving health outcomes and quality of life, replacing surgery and other invasive treatments, and quickening recovery for patients who receive these treatments. Drugs can reduce the need for bypass surgery, help prevent brain damage in stroke victims, lower cholesterol levels, or provide relief for chronic pain. Continued progress in biotechnology and genetic research promises still more innovative therapies in the coming years.
As important as prescription drugs are, not everyone has access to them. The newest drugs are often the most expensive, and millions of Americans have inadequate or no insurance coverage for drugs. The problems are especially acute for elderly and disabled Medicare beneficiaries. Many have chronic conditions for which drug treatments may be especially effective, such as osteoporosis, hypertension, diabetes, or depression. Medications for these conditions must be taken for extended periods, and beneficiaries with multiple problems may need several different drugs, many costing $1,000 a year or more. And nearly all take some drug in a given year. Yet nearly a third of all Medicare beneficiaries, many with very limited incomes, have no financial protection for the costs of these drugs, if they can obtain them at all. Many more beneficiaries find themselves moving in and out of the protection provided by insurance.
Medicare has never included coverage of outpatient prescription drugs, with very limited exceptions such as cancer chemotherapy drugs, and anti-transplant rejection drugs. The exclusion of drugs was common in private health plans when Medicare was enacted in 1965. Since then drug coverage has become a standard feature of private insurance, and it has become clear that the omission of outpatient drug coverage represents a crucial gap in protection for the most vulnerable Medicare beneficiaries. Beneficiaries without drug coverage face heavy financial burdens, and many go without needed medications or purchase less than the amount prescribed. Even many beneficiaries who have coverage today are at risk of losing it, paying higher premiums, or receiving more limited benefits as the cost of prescription drugs continues to grow. The problem is not limited to Medicare beneficiaries: millions of other Americans are without drug coverage, and some insurers and employers are responding to spending increases by requiring health plan participants to pay a larger share of their own costs.
As part of a broader plan to modernize Medicare, President Clinton has proposed a new, voluntary Medicare drug benefit that would offer all beneficiaries access to affordable, high-quality prescription drug coverage while maintaining the fiscal integrity of the program. In Congress, there has also been growing bipartisan interest in finding ways of extending drug coverage. At the same time, there has been increasing attention to the prices beneficiaries without coverage must pay for some needed medications. Congress is considering proposals that would give individual beneficiaries access to the discounts offered to group purchasers, or that would speed the availability of lower-cost substitutes for the newest and most expensive drugs.
As policymakers consider options to assure that every American can have access to innovative drug treatments, there is an urgent need for comprehensive and reliable information on drug coverage, drug spending, and drug prices. On October 25, 1999, the President directed the Secretary of Health and Human Services to study prescription drug costs and trends for Medicare beneficiaries. He asked that the study investigate:
- price differences for the most commonly used drugs for people with and without coverage;
- drug spending by people of various ages, as a percentage of income and of total health spending; and
- trends in drug expenditures by people of different ages, as a percentage of income and of total health spending.
This report is the Department’s response to that request. It represents the work of individuals and agencies throughout the Department, including the Agency for Healthcare Research and Quality (AHRQ), the Food and Drug Administration (FDA), the Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)) (HCFA(now known as CMS)), and the Office of the Assistant Secretary for Planning and Evaluation (ASPE).
In Chapter 1, the report provides estimates of the extent of prescription drug coverage among both Medicare beneficiaries and the general population. Chapter 2 provides evidence of the consequences of lacking coverage, including reduced utilization and expenditures for drugs, higher financial burdens, and failure to obtain needed treatments. Chapter 3 examines drug prices, highlighting growing disparities between what is paid by employers and insurers and what is paid by individuals who must purchase drugs on their own. In addition, several issues relating to geographic variation in prescription drug coverage, utilization, and pricing – where results are not definitive – are discussed in Appendix C.
While this initial study is informative, the research conducted for this study has uncovered numerous areas in which further investigation and analysis could provide a better understanding of problems in prescription drug financing and possible solutions. In the future, the Department will be continuing the coordinated and intensive research effort that produced this report and will provide new insights and analyses as they become available. The conclusion of this report includes a discussion of ideas for future research.
In addition, the Department commissioned a literature review by Bruce Stuart and his colleagues at the Peter Lamy Center for Drug Therapy and Aging at the University of Maryland School of Pharmacy. The literature review is included as Appendix A of this report. This review assesses the current state of research on prescription drug costs and trends for Medicare beneficiaries with and without prescription coverage and examines the use of prescription drugs and drug pricing in other nations. The detailed literature search included a traditional index search of literature in the medical, social and behavioral sciences, health services research and public policy arenas; search for relevant statistical data in domestic and international documents; identification of relevant ongoing research efforts; and identification of relevant working papers and presentations at professional associations. The resulting review summarized the literature from the perspective of what is known and what needs to be known on the issues of prescription drug coverage, pricing, utilization, and spending.
Main Data Sources for This Report
Analysis in this report is based chiefly on two household surveys conducted in 1996, the Medicare Current Beneficiary Survey (MCBS) and the Medical Expenditure Panel Survey (MEPS), and on data from pharmacy audits conducted by IMS Health. The following is a brief overview of these data sources. More detail on methodologies for MCBS and MEPS is provided in Appendix B.
MCBS and MEPS
For this report, coverage, utilization, and spending data for the Medicare population are drawn from MCBS. MCBS is a continuous, multipurpose survey of a representative sample of the Medicare population.1 Work on the MCBS is done under the direction of HCFA(now known as CMS)’s Office of Strategic Planning through its contractor, Westat, Inc. The 1996 survey included approximately 16,000 beneficiaries either in or joining the continuing sample, plus an additional one-time over-sample of approximately 2,000 beneficiaries in areas with high Medicare risk HMO penetration. Each continuing sample person, or an appropriate proxy respondent, is interviewed three times a year over a four-year period, regardless of whether he or she resides in the community or in an institution. In 1996, the sample for the Cost and Use component used in this report totaled 11,884 individuals; after excluding people who were institutionalized for the entire year, the sample includes 10,869 beneficiaries. The analysis of trends also drew upon the MCBS for 1992 through 1995.
For this report, coverage, utilization, and spending data for the non-Medicare population, along with some prescription price data for the entire population, are drawn from MEPS. MEPS, co-sponsored by the Agency for Healthcare Research and Quality and the National Center for Health Statistics (NCHS), is conducted to provide nationally representative estimates of health care utilization, expenditures, sources of payment, and insurance coverage.2 The MEPS has several components, of which two were used for this report: the Household Component (HC), a representative survey of the U.S. civilian noninstitutionalized population, and the Pharmacy Component (part of a broader Medical Provider Component), which contacts pharmacies reported by HC participants to supplement and validate information on prescription drug use and spending. The HC collects data through an overlapping panel design. In this design, data are collected through a preliminary contact followed by a series of six rounds of interviews over a two-and-a-half year period. Two calendar years of medical expenditures and utilization are collected from each household, along with information about insurance coverage. Data for 1996 are used in this report, based on a sample of 21,571 individuals.
The MCBS data used here include people who were in institutions only if they spent at least part of the year in the community. The MEPS HC is limited to noninstitutionalized people.3 This report therefore contains no information about drug coverage, utilization, or expenditures for the 1.6 million Americans, mostly elderly, who were living in nursing homes for the entire year of 1996, or for other people living in institutional settings such as long-term hospitals. Although facility residents are often heavy users of prescribed drugs, coverage and payment arrangements for these drugs are different from those for community residents. For example, drug costs may be included in a nursing home bill, rather than charged separately by a retail pharmacy. As a result, collection of comparable information for the community and institutionalized populations is not possible.
Three features common to the two surveys should be considered in interpreting the results:
- Both surveys found that some participants who failed to report having drug coverage actually had a prescription during the year for which payment was made by a third party. Payment source information might be supplied by the respondent in either MCBS or MEPS; in the case of MEPS, information could also be obtained through the pharmacy follow-back. In both surveys, the individual was deemed to have had drug coverage if self identified. In MCBS, however, there is an exception if a beneficiary reports drug coverage but has high out-of- pocket payments and no third party payments. In MEPS, the individual was also deemed to have drug coverage if identified in the pharmacy data.
- When a person reported or was otherwise identified as having more than one source of drug coverage during a year, he or she was assigned to one source of coverage. In MEPS, people with multiple sources of coverage were assigned to the type of coverage they had for the longest time during the year. If they had multiple sources for the same length of time they were assigned according to the following hierarchy: private group insurance, private nongroup insurance, private “other” group insurance, Medicaid, and Medicare HMO. MCBS also has a hierarchy: Medicare risk HMO, Medicaid, employer-sponsored, individually purchased, and other public. In MCBS, unlike MEPS, beneficiaries are assigned to the first relevant category in this listing regardless of how many months they spent in which categories.
- Both the MEPS and the MCBS analyses reported here treat individuals who reported coverage at any time during the year as covered. In fact, many people had drug coverage only for a part of the year. Data on duration of coverage for Medicare beneficiaries will be presented later in this report.
Further discussions of survey methodology can be found in Appendix B.
IMS Health
IMS Health conducts pharmacy audits to produce estimates of national sales of all pharmaceutical products sold through retail pharmacies. For each individual prescription drug, the IMS data provide information on total units sold during a given period, the acquisition costs paid by pharmacies, and the retail prices paid by three categories of purchasers: those who paid cash (for whom no insurance payment was made at the point of sale), those for whom payment was made by Medicaid, and those for whom payment was made by a third-party insurer. (Note that these categories reflect the source of payment to the pharmacy itself. If someone paid cash for a drug and was later reimbursed by an insurer, as is common under indemnity insurance plans, the transaction falls into the cash category.)
Other Data Sources
In addition to these three basic surveys, this report draws on findings from two other community-based surveys. The first is the ongoing Consumer Expenditure Survey (CES) conducted by the Bureau of Labor Statistics, which collects information on household income and spending. The second is the 1997 National Health Interview Survey (NHIS) conducted by the NCHS at the Centers for Disease Control and Prevention.
Trend estimates for aggregate U.S. drug spending are derived from the National Health Expenditures Series developed by HCFA(now known as CMS), which provides longitudinal information on aggregate spending by different payers for various categories of health services and supplies.
Finally, representatives of the Office of the Assistant Secretary for Planning and Evaluation and HCFA(now known as CMS) conducted a series of informal interviews with outside sources to collect background information for this report. These sources included representatives of drug manufacturers, pharmacy benefit managers, pharmacies, benefit consultants, consumer groups, and researchers familiar with the drug industry. They were asked for their insights on how the drug distribution system works, important trends in the industry, and any information that they could share about drug coverage, utilization, spending, and pricing.
Standard Errors and Statistical Significance Tests for Reported Results
Standard errors were produced for all of the survey-based results to ensure that the estimates derived from the surveys are robust – meaning that the reported estimates are not likely to be highly different from the values that would be produced if the entire population, rather than a sample, had been used to calculate the result. As a general rule, standard errors of greater than 30 percent of the reported result indicate that the estimate is unreliable. In these (relatively few) cases, the report will indicate that high standard errors preclude reporting the result.
Wherever possible, results noted in the text of this report have been subjected to statistical significance tests, to ensure that they are likely to be real, and not attributable to chance. The approach taken throughout this report is to indicate in a footnote only the few cases in which a result was not statistically significant. Thus, unless otherwise noted, results discussed in the text are statistically significant (at the 0.05 level, based on a two-tailed test).
The only results that were not subjected to significance tests were results based on the IMS data. The unique nature of the way IMS collects and reports its data does not allow for statistical testing of results from these audits. However, given the large sample sizes used by IMS (over 70 percent of US prescriptions filled at retail pharmacies), all results reported based on IMS data are highly likely to be statistically significant.
Strengths and Limitations of This Study
There are a number of important strengths and limitations of this study. In general, the main strengths of this work are the variety and diversity of data sources used in the analysis to create a more comprehensive picture than would have been otherwise possible. Specifically:
- The study took advantage of a comprehensive array of public data sources including newly available data on prescription drugs from the MEPS, detailed data on the Medicare population from the MCBS, and data from the NHIS, CES, and the National Health Expenditure Series.
- The study also made use of an important private sector data base on pharmaceutical prices from IMS Health. These data not only offer an enormous sample size (nearly 70 percent of prescriptions from retail pharmacies), but they are a data source accepted and widely used by industry, and have been in existence for a relatively long time.
- Taken together, the public and private data sources represent the most recent data available to address the main analysis questions.
- We also undertook a comprehensive literature review and conducted informal conversations with a wide range of industry experts to inform our analysis and to help explain the intricacies of the pharmaceutical marketplace.
As with all studies of this kind, there are limitations to the data and our analysis that are important for the reader to understand. Many of the key weaknesses stem from the short time frame in which the study was completed, which prevented more in-depth analysis on some topics, or inherent limitations of the data that are available to analyze prescription drugs. Specifically:
- The sample sizes for MEPS and MCBS are too small to do some subgroup analyses and to go beyond the simple two and three variable cross-tabs we present. In addition, both sample sizes are too small for any meaningful drug- by-drug analysis. Our use of IMS data alleviates these concerns for the analysis of drug prices.
- Although the 1996 MEPS and MCBS data represent the most recent data files available, given the dynamic nature of the pharmaceutical market, it will be important to continue to examine these issues as more information becomes available.
- Individually, each of the main data sets is not fully representative (e.g. nursing home patients are not included in this analysis, MCBS only includes information on Medicare recipients, and IMS data used in the study only cover retail pharmacies). Taken together, however, the data sources cover the range of populations more effectively than most previous studies.
- Lack of rebate information, which manufacturers consider to be highly sensitive, is a ubiquitous problem in analyzing drug prices. Given the highly competitive nature of this market, it is unlikely that there will ever be a comprehensive data source on rebates.
- This short-term study presents descriptive results using only univariate analysis with two and three variable cross-tabs. Descriptive analysis of this type cannot explain why covered individuals use more drugs or whether use of drugs leads to better outcomes. We plan to undertake future analysis that will use multivariate techniques to delve further into the data presented here, although such analysis is likely to push the limits of available data.
- A problem common to all surveys that sample individuals is their reliance on self-reported data. Self-reported data may not be accurate due to recall problems. Both MEPS and MCBS, however, use multiple rounds of interviewing of the same respondents each year in an effort to mitigate recall problems. In addition, MEPS uses a pharmacy follow-back survey to validate information reported by respondents, and the IMS data are derived directly from the retail pharmacy.
The conclusion of this report includes a discussion of ideas for future research that were generated during the course of developing this report. The strong foundations that this report lays should allow us to delve further into these topics in future analysis.
Endnotes
1 - For a description of the MCBS, see G.S. Adler, Summer 1994, “A Profile of the Medicare Current Beneficiary Survey,” Health Care Financing Review, 15(4): 153-163.
2 - For a description of MEPS, see Cohen, J.W. et al, "The Medical Expenditure Panel Survey: A National Health Information Resource," Inquiry, 33 (Winter 1996/97), pp. 373-389.
3 - A separate Nursing Home Component surveys facilities and residents.
Chapter 1. Prescription Drug Coverage
Prescription drugs play a critical role in contemporary medicine and most health insurance policies cover prescription drugs as an integral part of the benefit package. However, this was not the case in 1965 when Medicare was enacted. Consequently, Medicare does not include coverage of outpatient prescription drugs as a basic benefit. While today, over 85 percent of Medicare beneficiaries use at least one prescription drug annually, beneficiaries must obtain drug coverage through a supplemental policy, by enrollment in a Medicare+Choice plan which includes coverage for prescription drugs, or through Medicaid.
This chapter explores the sources of drug coverage and the nature of coverage that is provided to Medicare beneficiaries through different kinds of supplemental policies. Survey data is employed to analyze the sources of coverage for the Medicare and non- Medicare population in 1996 as well as the economic and demographic characteristics of those who have drug coverage and those who do not. Analysis of data on the duration of coverage for the Medicare population is also presented. Next, differences in coverage rates by alternative measures of health status are explored. Lastly, trends in drug coverage for the Medicare and non-Medicare population are analyzed.
Key findings include:
- Only 53 percent of Medicare beneficiaries had drug coverage for the entire year of 1996, although 69 percent had coverage for at least one month during the year.
- Most sources of drug coverage are potentially unstable. Almost 48 percent of beneficiaries with drug coverage through Medigap and 29 percent who were covered through Medicare HMOs had drug coverage for only part of the year. Additionally, while employer-sponsored retiree coverage, the most prevalent single source of drug benefits, covered 32 percent of Medicare beneficiaries in 1996, 14 percent of those beneficiaries had only part year coverage from their former employers.
- Drug benefits are becoming less generous. There is considerable evidence that cost sharing for prescription drugs is increasing and that overall caps on coverage are both becoming more common and are being set at lower levels. For example, Medicare+Choice plans generally have reduced drug benefits and increased enrollee out-of-pocket costs in 2000. Eighty-six percent of plans have annual dollar limits on drugs, including 70 percent of plans with annual caps of $1000 or less, and 32 percent with caps of $5Drug benefits are becoming less generous. There is considerable evidence that cost sha
- Drug coverage is likely to decline as fewer employers offer health benefits to future retirees. For example, one employer survey recorded a drop from 40 percent in 1993 to 28 percent in 1999 in the number of large firms offering health benefits to Medicare eligible retirees. Additionally, employers have tightened eligibility rules and increased cost-shifting to retirees. Of those employers that still offer medical coverage, the survey found that 40 percent are requiring Medicare-eligible retirees to pay Drug coverage is likely to decline as fewer employers offer health b
- Individuals with incomes between 100 percent and 150 percent of poverty, or individuals age 65 or older with incomes between $7,527 and $11,287 in 1996, have the lowest rate of coverage. Although coverage varies by income, nearly one-fourth of beneficiaries with incomes over 400 percent of poverty lack coverage.
- Beneficiaries are less likely to have coverage if they are very old or live outside of a metropolitan area. About 37 percent of beneficiaries age 85 and above lacked coverage at any time during 1996, compared to 28 percent of beneficiaries age 65 through 69. About 43 percent of beneficiaries living in rural areas lacked any drug coverage, compared to 27 percent of beneficiaries living in urban areas.
- Coverage rates vary little by self-reported health status, but are considerably higher for those with five or more chronic conditions. By all measures, at least one-fourth of those in any category of health status lack coverage.
- Nearly one in four in the non-Medicare population never had any coverage for drugs in 1996. About 80 percent of those with full-year coverage got that coverage through employers.
Sources and Nature of Coverage
Most people who have medical insurance obtain drug coverage through the same source that provides their overall insurance: most commonly an employer or union plan, private nongroup coverage, or Medicaid. Nearly all employer plans include drug benefits for active workers (although not necessarily retirees). Coverage of prescription drugs by Medicaid is optional under federal law, but every state’s program includes this benefit.1
Because Medicare generally does not cover outpatient prescription drugs, beneficiaries who have drug coverage obtain it as part of a supplemental plan, which usually also covers Medicare’s deductibles and coinsurance and may include benefits for other services excluded from Medicare. There are several ways for beneficiaries to obtain supplemental coverage: retiree health benefits, private Medigap policies, Medicare+Choice plans,2 Medicaid, and other federal and state programs.
Different sources of prescription drug coverage vary in the extent of coverage, the amount participants must contribute toward the cost of their drug spending, and how a participant obtains the benefits.
Employer and Retiree Plans
Employer-sponsored health benefits are the single largest source of drug coverage for both the Medicare and non-Medicare population. As will be discussed below, in 1996 employer-sponsored plans were the source of coverage for about 60 percent of the non- Medicare population and 32 percent of Medicare beneficiaries.
Until the late 1980s, prescription drug coverage was often not a distinct benefit, but was included in conventional major medical plans if it was included at all. It was subject to the overall deductible for all services and to the same coinsurance amounts (typically 20 percent) that applied to other medical care.3 Two developments have led to major changes in employer drug benefits.
The first is the widespread use of managed care plans, such as health maintenance organizations (HMOs), preferred provider organizations, and point of service plans, which now cover 89 percent of workers, although a smaller percent of retirees.4 Managed care plans offer a distinct drug benefit; only about 10 percent impose a deductible, and a copayment—for example, $5 for generic drugs and $10 for brand- name drugs—is used in place of a coinsurance percentage.5
The second is the growth of pharmacy benefits managers (PBMs).6 Most managed care plans contract with a PBM to administer their drug benefits. In addition, many employers have “carved out” prescription drug benefits from their general health plans and contract separately with a PBM.7 Unlike indemnity insurers, PBMs process and pay claims at the point of sale. They develop formularies (lists of preferred or approved drugs), negotiate discounts with manufacturers and retail pharmacies, encourage use of mail-order pharmacies, and take other steps to control drug costs. (A fuller description of PBM practices is provided in Chapter 3.) PBMs are not insurers; they usually do not accept financial risk for the costs of services, although their contracts with managed care plans or employers may include some incentives for cost reduction.
Some large managed care organizations, such as Kaiser Permanente and Aetna, manage their own drug benefits instead of contracting with a PBM. As the practices of these organizations are similar to those of PBMs, the single term PBM will be used in this report to cover all types of entities that manage prescription drug benefits. Some other insurers, such as indemnity plans or self-insured employer plans, may not manage drug benefits, but simply pay claims submitted by enrollees. Overall, an estimated 78 percent of people with non-Medicaid drug coverage are in PBMs, while the rest are in plans managing their own benefits or in unmanaged plans.8
Although still the single largest source of Medicare supplemental drug coverage, retiree health benefits have been eroding in the last decade. In part, this decline is a response to accounting rule changes that required firms to account for benefits promised to future retirees as a current liability. In some cases, firms may provide more generous coverage for active workers than for retirees because of the role of benefits in recruiting employees. According to a recent employer survey, about 41 percent of large firms offered health benefits to retirees in 1998 compared to 66 percent in 1988. Of these, 80 to 85 percent provided benefits to Medicare beneficiaries; the remainder covered only early retirees in the period before Medicare eligibility.9 Another survey recorded a drop from 40 percent in 1993 to 28 percent in 1999 in the number of large firms offering health benefits to Medicare eligible retirees.10
Smaller firms are less likely to offer health benefits to retirees. One survey found that only 8 percent of smaller firms, those with fewer than 200 workers, offered retiree coverage in 1998.11 The Mercer/Foster Higgins survey of medium-sized firms with 500 to 1000 employees, reported that 22 percent of employers offered coverage to Medicare- eligible retirees in 1999.12
The impact of the decline in employer sponsorship of retiree health benefits will be felt more strongly in the future. Most firms that have dropped coverage have done so for their active workers planning to retire in the future, rather than for current retirees.13 Since effective rates of change vary by firm and by the age of the workers, it is difficult to predict the rate of coverage decline.
Additionally, there are indications that the nature of coverage offered to retirees has changed. Of those employers that still offer medical coverage, 40 percent are requiring Medicare-eligible retirees to pay the full cost of their benefits compared to 28 percent in 1995.14 Further, many firms that continue to offer retiree coverage have tightened the eligibility rules for future retirees. For example, the percentage of large employers who require employees 55 years or older to have between 10 and 15 years of employment to qualify for benefits rose from 30 percent in 1991 to 49 percent in 1998.15
In recent years, increasing numbers of employers who provide retiree health benefits have permitted or required Medicare-eligible retirees to enroll in a Medicare+Choice plan (see below). The employer may pay the plan’s premium for its usual supplemental coverage and may also negotiate additional supplemental benefits on behalf of the retirees.16
Some drug coverage is provided in 80 percent of retiree health benefit plans.17 However, many employers are looking for ways to reduce the costs of these benefits. One recent survey of large employers found that 40 percent would consider cutting back on prescription drug coverage for Medicare-eligible retirees in the next three to five years.18
Private Medigap and Other Nongroup Coverage
Medicare beneficiaries may buy individual Medicare supplemental policies, known as Medigap plans, from private insurers. By law, an insurer selling Medigap must offer one or more of ten standardized plans. Three of these, known as plans H, I, and J, include some prescription drug coverage.19 These three Medigap plans impose a $250 deductible. They then pay 50% of covered charges up to a maximum plan payment of $1,250 for plans H and I and $3,000 for plan J. Some beneficiaries who bought policies before this law took effect have non-standardized plans; little is known about how many of these include drug coverage.20 A few states are also exempt from using the standardized plans.21
Many carriers do not offer the plans with drug coverage, and those who do may refuse coverage to applicants perceived to be high-risk. Carriers must accept all beneficiaries aged 65 and above during a limited open enrollment that ends 6 months after the beneficiary first qualifies for Medicare; no open enrollment requirement applies to disabled beneficiaries under the age of 65.22 In addition, twelve states have laws requiring community rating or preventing Medigap insurers from raising premiums as policy holders age, a practice known as attained-age rating.23
Premiums for the plans with drug coverage are much higher than for other Medigap plans, both because of the cost of the drug benefit itself and because the benefit is likely to attract beneficiaries who incur higher general medical expenses. An analysis of June 1999 premiums for some of the major Medigap carriers found that the premium for individuals aged 65 averaged $1,000 higher for plan J than for plan F, the most similar plan without drug benefits; the gap between premiums for the F and J plans increased for older beneficiaries in plans that used attained-age rating. In some cases, the incremental premium for adding drug coverage was greater than the maximum value of the benefit.
Some Medigap plans have begun to use PBMs. Since July 1998, United HealthCare, a health plan that also sells Medigap policies through AARP, has employed a PBM to negotiate lower drug prices for its H, I, and J policyholders at preferred pharmacies. Through their PBM, United HealthCare is also able to offer point of sale copayments and drug-interaction screening to its Medigap policyholders. However, most plans are still indemnity plans, which reimburse participants for their drug expenditures after the fact and do not manage the benefit.
The category of individually purchased coverage also includes a variety of additional supplements. Some individuals purchase non-Medigap policies, such as private long- term care insurance or drug-only policies. The exact number of these policies and the value of the benefits they include is unknown. The term “Medigap” will be used to describe the entire category of individually purchased Medicare supplemental insurance in the remainder of this report.
There is no reliable source of information about the nature of the drug benefits provided to non-Medicare enrollees in private nongroup insurance plans, or about how these benefits are administered.
Medicare+Choice Plans
In 2000, about 6 million Medicare beneficiaries are enrolled in Medicare+Choice (M+C) coordinated care plans. This number has grown from about 3 million in 1996 enrolled in risk HMOs, comparable to today’s Medicare+Choice plans. When a Medicare beneficiary chooses to enroll in an M+C plan (usually an HMO), the contracting organization receives a fixed monthly payment from Medicare to furnish all Medicare- covered services. If it is projected that the plan can furnish these services at a cost less than the Medicare payment, the plan must share the savings with enrollees by providing supplemental benefits. As a result of this rule, or simply to compete for market share, most M+C plans have offered enrollees supplemental coverage. These supplemental benefits often include some drug coverage, at a much lower premium than enrollees would have had to pay for an equivalent Medigap plan. In addition, unlike Medigap plans, M+C plans must accept all applicants, regardless of health status (other than individuals with End Stage Renal Disease, [ESRD]) or age. M+C plans have thus been an attractive source of supplemental coverage for beneficiaries willing to accept the restrictions on provider choice that plan membership entails.
In 1996, 95 percent of enrollees in risk HMOs had at least some drug benefit.24 More recently, some plans have reduced benefits. While complete information is not available, HCFA(now known as CMS) has estimated that in 1999, 84 percent of HMO enrollees were in plans that included drugs as a basic benefit.25 (Other enrollees may have obtained drug coverage by purchasing a high-option plan.)
Drug benefits in Medicare HMOs vary widely. About 54 percent of enrollees with drug coverage in 1999 were in plans that imposed an annual dollar limit on all drug payments, most commonly between $1,000 and $2,000. Another 24 percent were in plans that had a limit on payments for brand-name drugs but no limit for generic drugs, while 22 percent were in plans that had no payment limit for drugs. Copayments typically ranged between $5 and $10 per prescription; higher copayments were often imposed for brand-name drugs. (Note that this description is based on the basic plans offered to all enrollees. Some plans offer supplemental benefits at a higher premium. There is no information on the level of benefits offered by employer-sponsored plans through M+C contractors.) As is the case for other enrollees in managed care plans, drug benefits are commonly administered by a PBM or are managed directly by the organization.
There is considerable geographic variation in the level of drug coverage offered by M+C plans. The most generous coverage appears to be associated with high payment areas in which multiple plans compete for enrollees. For example, in 1999 all plans with unlimited drug benefits and no copayments were located in Florida. Plans with annual caps of $2000 or more were concentrated in Miami, Phoenix, and Los Angeles. In general, where plans are available in rural areas, they tend to charge higher premiums and offer no drug coverage or significantly less generous drug coverage.26
In 2000, M+C plans generally have reduced drug benefits and increased enrollee out-of- pocket costs. For example, 86 percent of plans have annual dollar limits on drugs, including 70 percent of plans with annual caps of $1000 or less, and 32 percent with annual caps of $500 or less per enrollee, up from 35 percent and 19 percent respectively in 1998. Additionally, while over one million beneficiaries lived in areas in 1999 with M+C plans available that offered zero copayments for prescription drugs, all beneficiaries will now be subject to copayments for generic and brand-name drugs.27
Medicaid
While coverage for outpatient prescription drugs is optional under Medicaid, all states provide this benefit for families and children enrolled in the program. In addition, certain low-income Medicare beneficiaries may qualify for Medicaid and receive full Medicaid benefits, including prescription drug coverage. Medicaid eligibility can be achieved in several ways. Medicare beneficiaries may qualify for Supplemental Security Income (SSI) or related cash assistance if they are elderly or disabled and meet an income limit that is well below the federal poverty level (FPL), $11,250 for a couple in 2000. In most states, receipt of SSI means automatic enrollment in Medicaid. Beneficiaries may qualify for a state’s “medically needy” Medicaid program by meeting a state-established income test, often by “spending down”—incurring medical bills sufficient to reduce their income and assets to the required level. Those in need of long-term care may meet special eligibility limits for home- and community-based service programs.
Low-income Medicare beneficiaries who fall outside these categories may qualify for Medicaid assistance that does not include prescription drug coverage.28 “Qualified Medicare Beneficiaries” (QMBs), with incomes below 100 percent of FPL, receive Medicaid assistance for Medicare cost-sharing and Medicare’s Part B premium. A state may choose to extend full Medicaid benefits to this group with federal matching funds by raising their income eligibility to 100 percent of FPL, but most have not. “Specified Low-income Medicare Beneficiaries” (SLMBs), with incomes between 100 percent and 135 percent of FPL, receive Medicaid assistance for Part B premiums only. In 1996, an estimated 90 percent of Medicare beneficiaries receiving Medicaid had drug coverage.29 This may include some cases where QMBs or SLMBs received drug coverage through state-only programs.
Medicaid programs may impose only “nominal” copayments for prescription drugs, and no copayments for certain services, such as those furnished to children. All but 17 states impose some copayments, typically $1 to $2 for prescription drugs. Although most states enroll families and children in managed care, very few have enrolled aged or disabled beneficiaries. Additionally, many states that use managed care have excluded prescription drugs from the contracts and provide the benefits directly on a fee-for-service basis.30 Thus most Medicare beneficiaries who receive their drug coverage through Medicaid receive drugs on a fee-for-service basis.
The Omnibus Budget Reconciliation Act of 1990 (OBRA 90) governs the provision of pharmaceutical benefits under Medicaid. Under the law, pharmaceutical manufacturers are required to enter into rebate agreements with the Department of Health and Human Services (HHS) in order for these companies to sell their products to state Medicaid programs (see Chapter 3 for further discussion of Medicaid rebates). The agreement establishes a basic rebate of the greater of Average Manufacturer Price (AMP) minus the manufacturer’s best price or 15.1% of the AMP for single source and innovator multiple source drugs. At the same time, OBRA 90 prohibits state Medicaid programs from using some of the same management techniques as PBMs. In particular, states may not use restrictive formularies, although they can develop programs for prior authorization and drug utilization review to manage the drug benefit. Some states control costs by capping the number of prescriptions that a beneficiary can fill within a month, restricting refills, or limiting the amount of medication that can be dispensed at one time. Arkansas, Nevada, Oklahoma, Texas, Wisconsin, and Wyoming, for example, limit Medicaid recipients to three prescriptions per month, with some exceptions. West Virginia restricts beneficiaries to ten prescriptions per year.31
Other Sources of Drug Coverage
Some Medicare beneficiaries and non-Medicare individuals receive a degree of assistance in purchasing drugs through other federal and state programs. The most important is the Department of Veterans Affairs (VA). When a VA physician prescribes a drug, a veteran may fill the prescription at a VA pharmacy, or through a VA mail- order program, usually with only a small copayment. Eligibility for VA services thus functions as the equivalent of insurance, but only for prescriptions written by a VA physician. Medicare generally does not cover VA services, so a veteran may have to pay for the physician visit to access the drug benefit. However, visits are free for the indigent and for veterans with service-related conditions.
Currently, 16 states operate pharmacy assistance programs, which cover about 750,000 individuals.32 These programs generally cover low-income elderly people who do not qualify for Medicaid drug coverage; about half are available to other population groups, typically the disabled.33 Some of the programs cover the same range of drugs covered under the state’s Medicaid program. Others are restricted to “maintenance” drugs required for certain chronic conditions.
A very small number of Medicare beneficiaries are assisted in drug purchases by Civilian Health and Medical Program of the Uniformed Services (CHAMPUS), worker’s compensation, or other coverage sources.
Some people without insurance may receive certain drugs for free. Some manufacturers operate programs that make certain drugs available to uninsured people meeting specified eligibility criteria. The Pharmaceutical Research and Manufacturers of America (PhRMA) estimates that the programs for which they have data filled 2.7 million prescriptions for nearly 1.5 million people in 1998.34 In addition, manufacturers commonly distribute samples to be dispensed by physicians and clinics. IMS Health reports that for the twelve months ending September 1999, companies gave drug samples with a retail value of about 7 billion dollars to office-based physicians.35
Other individuals without coverage may purchase discount cards from groups like AARP which enable them to receive a percentage discount for prescriptions purchased in participating pharmacies. Many pharmacies also offer discounts to seniors. Discounts of this type are not classified as drug coverage. There is no centralized source for data on the number of people taking advantage of these discounts or the value of the discounts they receive.
Prescription Drug Coverage in 1996
This section presents information from surveys on the source and extent of drug coverage in 1996. MCBS is used for the Medicare population, and MEPS for the non- Medicare population. Unless otherwise noted, all results reported in this chapter from MCBS and MEPS are statistically significant (at the 0.05 level, based on a two-tailed test). See the Introduction of this report for details. Further discussion of methodology can be found in Appendix B.
Medicare Beneficiaries
Sources of drug coverage for Medicare beneficiaries were unstable in the mid 1990s. In 1996, the most recent year for which data are available, an estimated 69 percent of Medicare beneficiaries had drug coverage for at least one month of the year, although only 53 percent had coverage for the entire year. Table 1-1 groups beneficiaries according to their primary source of Medicare supplemental coverage—that is, the source that assisted them with Medicare cost-sharing expenses. Not every beneficiary who had drug coverage obtained it through the same source that provided the beneficiary’s primary supplement. About 5 percent of those with drug coverage obtained it through a different source; this was most common for those whose primary supplement was an individually purchased plan.
| Primary supplement | With drug coverage (000s) |
Without drug coverage (000s) |
Percent without drug coverage | Source of coverage for beneficiaries with drug coverage (%) |
|---|---|---|---|---|
| Medicare risk HMO | 3,729 | 215 | 5.5% | 14.5% |
| Medicaid | 4,408 | 512 | 10.4% | 17.2% |
| Employer Sponsored | 12,045 | 1,535 | 11.3% | 47.0% |
| Individually purchased | 4,192 | 6,292 | 60.0% | 16.4% |
| Other public sources | 1,246 | 178 | 12.5% | 4.9% |
| FFS Medicare only | - | 2,891 | 100% | --- |
| Total | 25,621 | 11,623 | 31.2% | 100%\ |
| Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996. | ||||
The likelihood of having drug coverage varies by different types of supplemental coverage. Only 40 percent of beneficiaries with Medigap or other individual coverage as a primary supplement had a drug benefit. On the other hand, almost 95 percent of risk HMO enrollees had some drug coverage. Almost 3 million beneficiaries had no form of supplemental coverage, whether for drugs or for other services.
Among all beneficiaries with drug coverage for at least one month in 1996, nearly half, 47 percent, were covered through an employer-sponsored retiree plan. Medicaid provided drug benefits for 17 percent of covered beneficiaries; Medigap was the primary supplement for about 16 percent of covered beneficiaries; and 14.5 percent of those with coverage were enrolled in a Medicare risk HMO. Almost 5 percent of beneficiaries with coverage obtained it through a variety of small public programs.
However, as was noted earlier, not all beneficiaries who had drug coverage had it for the entire year. Information on this issue was not collected as part of this study. The following information (see Figure 1-1) is derived from an independent analysis of 1996 MCBS data conducted by Bruce Stuart, et al. of the University of Maryland for the Commonwealth Fund.36
Figure 1-1. Duration of Prescription Drug Coverage for Medicare Beneficiaries in 1996
Note: This analysis includes only noninstitutionalized beneficiaries enrolled in Medicare for the entire year.
Source: Bruce Stuart, Dennis Shea, and Becky Briesacher, Prescription Drug Costs for Medicare Beneficiaries: Coverage and Health Status Matter, New York, Commonwealth Fund, Issue Brief, January 2000
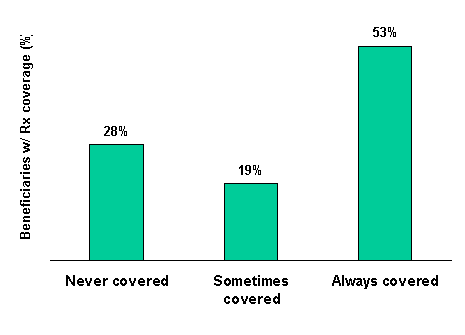
The authors’ month-to-month examination of supplemental insurance shows that prescription coverage in 1996 was often fragmented and non-continuous. Over one in four Medicare beneficiaries (about 6.3 million people) who had coverage at any time during 1996 had coverage for only part of that year. For people with part-year coverage whose period of coverage could be ascertained, the average duration of coverage was 6.6 months. Thus, while the data presented in Table 1-1 indicate that 69 percent of beneficiaries had drug coverage for at least one month in 1996, only 53 percent had coverage for the entire year.
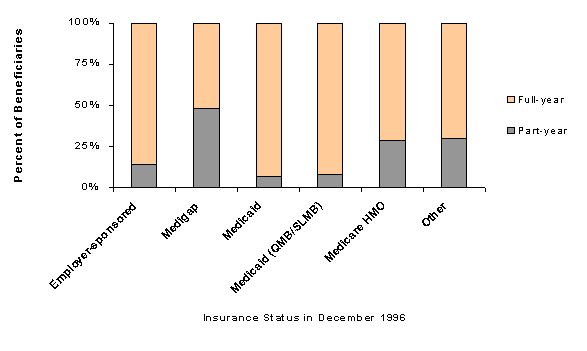
Table 1-2 shows partial and full-year drug coverage rates by source of supplemental insurance for those Medicare beneficiaries who had drug coverage for at least one month during 1996.37 Beneficiaries with every kind of Medicare supplement experienced changes in their coverage status in 1996, but changes for some types of coverage were particularly dynamic (see Figure 1-2). Almost 48 percent of beneficiaries with drug coverage through Medigap policies and 29 percent who were enrolled in Medicare HMOs had drug coverage for only part of the year. Almost 14 percent of individuals with employer-sponsored drug coverage had part-year coverage. Individuals dually eligible for Medicare and Medicaid had the most stable coverage during the year, with only 7 percent experiencing gaps in drug coverage.38
Figure 1-2. Duration of Coverage for Beneficiaries who had Coverage in 1996, by Source of Coverage
Note: This analysis includes only noninstitutionalized beneficiaries enrolled in Medicare for the entire year.
Source: Bruce Stuart, Dennis Shea, and Becky Briesacher, Prescription Drug Costs for Medicare Beneficiaries: Coverage and Health Status Matter, New York, Commonwealth Fund, Issue Brief, January 2000.
| Source of coverage in December 1996 | Of full-year beneficiaries covered at any time in 1996, percent who had: | |
|---|---|---|
| Part-year coverage | Full-year coverage | |
| Total | 26% | 74% |
| Employer-sponsored | 14% | 86% |
| Individual Medigap | 48% | 52% |
| Medicaid | 7% | 93% |
| Medicaid QMB/SLMB | 8% | 92% |
| Medicare HMO | 29% | 71% |
| Other | 30% | 70% |
|
Note: This analysis includes only noninstitutionalized beneficiaries enrolled in Medicare for the entire year. Source: Bruce Stuart, Dennis Shea, and Becky Briesacher, Prescription Drug Costs for Medicare Beneficiaries: Coverage and Health Status Matter, New York, Commonwealth Fund, Issue Brief, January 2000. |
||
Drug coverage for individuals with Medigap policies was particularly tenuous. While MCBS reports that 39.8 percent of Medicare beneficiaries with Medigap or other private nongroup policies had drug coverage in 1996, that number is misleading. Compared to other categories of supplemental insurance, beneficiaries with Medigap were most likely to have drug coverage from another source and most likely to have part year coverage. About 16 percent of Medigap policyholders with drug coverage got their coverage from a secondary source, and only 25 percent had coverage for the full year. Additionally, as noted above, this category includes an unknown number of non- Medigap policies, such as private long-term care insurance or drug-only policies. There is no data on the value of the drug coverage provided under these plans. Overall, only 21 percent of Medigap or other private nongroup insurance policy holders had drug coverage from that source for the full year.
In an additional analysis, Stuart et al. analyzed drug coverage for those individuals continuously enrolled in Medicare for 1995 and 1996.39 Only 46 percent of beneficiaries had drug coverage throughout the entire two-year period. Six percent of these beneficiaries maintained coverage throughout the period by switching from one source of coverage to another at least once. While 11 percent of beneficiaries gained coverage at some point during the period, 8 percent lost coverage and 12 percent had intermittent coverage.
Characteristics of Individuals with and Without Coverage
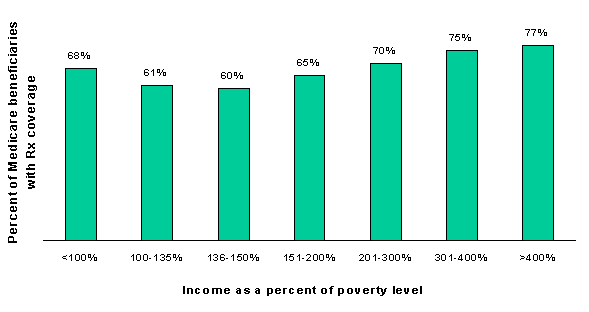
Table 1-3 shows drug coverage rates for beneficiaries with coverage for at least one month in 1996 by income as a percent of the federal poverty threshold (in 1996 the threshold was $7,525 for an individual and $9,491 for a couple age 65 or older).40 Coverage is lowest for beneficiaries between 100 and 150 percent of poverty. These beneficiaries may not qualify for Medicaid or may be eligible only for limited QMB/SLMB benefits, and they may be unable to afford to purchase coverage on their own. Above this level, coverage rises fairly steadily with income, although about 23 percent of Medicare beneficiaries with incomes above 400 percent of poverty had no drug coverage at any time in 1996 (see Figure 1-3).
| Income as percent of poverty | With drug coverage (000s) |
Without drug coverage (000s) |
Percent without drug coverage |
|---|---|---|---|
| <100% | 5,498 | 2,619 | 32.3% |
| 100-135% | 2,829 | 1,795 | 38.8% |
| 136-150% | 1,020 | 676 | 39.8% |
| 151-175% | 1,708 | 926 | 35.2% |
| 176-200% | 1,812 | 995 | 35.5% |
| 201-300% | 5,178 | 2,226 | 30.1% |
| 301-400% | 3,094 | 1,031 | 25.0% |
| >400% | 4,482 | 1,355 | 23.2% |
| Total | 25,621 | 11,623 | 31.2% |
|
Note: Not all differences in coverage rate by income groups are statistically significant. The differences between the groups with the highest rates of coverage and the groups with the lowest rates of coverage are statistically significant. The Federal poverty threshold for couples 65 and over in 1996 was $9491. Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996. |
|||
Figure 1-3. Medicare Beneficiaries Who Never Had Drug Coverage During the Year, by Income, 1996
Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Beneficiary Survey Cost and Use File, 1996
Table 1-4 shows the number of beneficiaries without coverage by income as a percent of all beneficiaries without coverage. The majority of beneficiaries without drug coverage in 1996, 56 percent, had incomes above 150 percent of poverty and 40% had incomes above 200 percent of poverty.
| Income as percent of poverty | Beneficiaries without drug coverage (000s) |
Percent of total without coverage (000s) |
|---|---|---|
| <100% | 2,619 | 22.5% |
| 100-150% | 2,471 | 21.0% |
| 151-200% | 1921 | 16.5% |
| >200% | 4612 | 40.0% |
| Total | 11,623 | 100.0% |
|
Note: The Federal poverty threshold for couples 65 and over in 1996 was $9491. Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996. |
||
Table 1-5 shows coverage rates for at least one month in 1996 by age. Disabled nonelderly beneficiaries are slightly more likely to have coverage than the elderly. More disabled beneficiaries may have incomes low enough to qualify for SSI and thus receive Medicaid; the disabled account for 12 percent of all beneficiaries but 34 percent of beneficiaries with Medicaid. On the other hand, they are less likely to have employer coverage or be enrolled in Medicare risk HMOs.
Among the elderly, rates of coverage drop steadily with age. There are a number of possible explanations. First, the very old tend to have lower incomes than younger elderly people and may not be able to afford Medigap—especially because Medigap premiums often rise with attained age. Second, retiree health benefits are a relatively recent phenomenon; the very old may be more likely than the younger elderly to have retired before such coverage became prevalent.
| AGE | With drug coverage (000s) |
Without drug coverage (000s) |
Percent without coverage |
|---|---|---|---|
| 0-44 | 1,038 | 432 | 29.4% |
| 45-64 | 2,093 | 781 | 27.2% |
| 65-69 | 6,841 | 2,644 | 27.9% |
| 70-74 | 6,025 | 2,658 | 30.6% |
| 75-79 | 4,599 | 2,229 | 32.6% |
| 80-84 | 2,964 | 1,640 | 35.6% |
| 85+ | 2,061 | 1,238 | 37.5% |
| Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996. | |||
Table 1-6 shows rates of coverage for at least one month in 1996 by sex. Males are slightly more likely to have coverage than females; as male beneficiaries tend to be younger than female beneficiaries, this finding is consistent with the age differences shown in Table 1-5. Also, males are more likely to have worked and have access to retiree insurance.
| SEX | With drug coverage (000s) |
Without drug coverage (000s) |
Percent without coverage |
|---|---|---|---|
| Male | 11,561 | 4,917 | 29.8% |
| Female | 14,059 | 6,706 | 32.3% |
| Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996. | |||
Table 1-7 shows coverage for any part of 1996 by race. Differences in coverage rates by race are not statistically significant.
| RACE | With drug coverage (000s) | Without drug coverage (000s) | Percent without coverage |
|---|---|---|---|
| White | 21,804 | 10,176 | 31.8% |
| Black | 2,397 | 1,016 | 29.8% |
| Other | 1,420 | 431 | 23.3% |
|
Note: “Other” includes Asian, Hispanic, and North American Native. Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996. |
|||
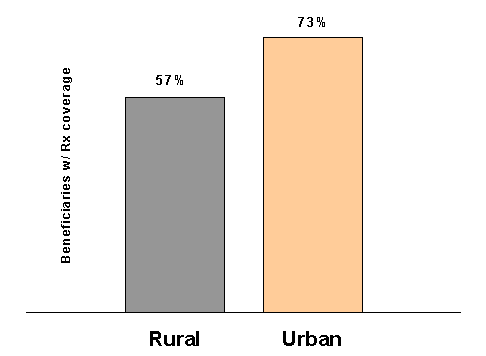
Table 1-8 and Figure 1-4 show coverage for at least one month in 1996 according to whether the beneficiary resides in a metropolitan statistical area. Beneficiaries outside metropolitan areas have a much lower rate of coverage. While 57 percent of beneficiaries living in rural areas had coverage for at least one month in 1996, 73 percent of beneficiaries living in metropolitan areas had some drug coverage. Rural beneficiaries also had less full-year coverage; only 44 percent had coverage throughout the year.41 By contrast, 49 percent of beneficiaries living in urban areas had coverage for the full year. One factor is probably more limited availability of Medicare HMOs, which tend to be concentrated in metropolitan areas. HCFA(now known as CMS) projects that, in 2000, about 29 percent of beneficiaries live in areas with no access to a Medicare+Choice plan. In addition, nonmetropolitan beneficiaries are less likely to have employer-sponsored drug benefits, probably because their jobs were less likely to have offered retiree benefits.
Figure 1-4. Beneficiaries Who Never Had Drug Coverage During the Year, by Metropolitan Residence, 1996
Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Beneficiary Survey Cost and Use File, 1996
| Residence | With drug coverage (000s) | Without drug coverage (000s) | Percent without coverage |
|---|---|---|---|
| Metropolitan | 19,932 | 7,414 | 27.1% |
| Non-metropolitan | 5,660 | 4,203 | 42.6% |
| Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Beneficiary Survey Cost and Use File, 1996 | |||
Table 1-9 and Figure 1-5 show coverage for at least one month in 1996 by self-reported health status. Differences in coverage rates by health status are not statistically significant. However, differences in source of coverage are significant: those reporting poor health are more likely to have coverage through Medicaid and less likely to have coverage from employers or HMOs.
Figure 1-5. Beneficiaries Who Never Had Drug Coverage During the Year, by Health Status, 1996
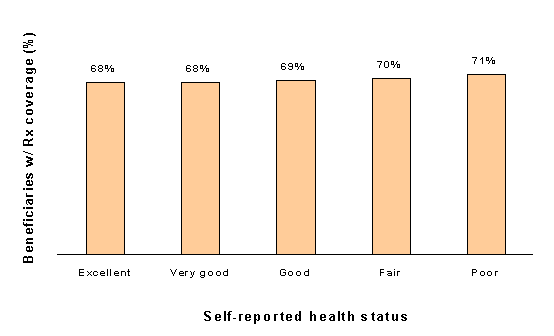
Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Beneficiary Survey Cost and Use File, 1996
| Health Status | With drug coverage (000s) | Without drug coverage (000s) | Percent without coverage |
|---|---|---|---|
| Excellent | 4,186 | 1,967 | 32.0% |
| Very Good | 6,742 | 3,193 | 32.1% |
| Good | 7,693 | 3,468 | 31.1% |
| Fair | 4,496 | 1,970 | 30.5% |
| Poor | 2,451 | 1,000 | 29.0% |
| Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996. | |||
Additionally, unstable drug coverage was evident for individuals with self-reported poor health status. Almost 21 percent of individuals with fair to poor health status had only part-year coverage in 1996.42
However, self-reported health status is a subjective measure that may or may not be related to actual need for drugs or other health services. The next three tables use different measures as alternative indicators of a likely need for prescription drugs.
The first is functional status, which measures the extent to which an individual requires assistance in performing “activities of daily living” (ADLs), such as bathing, dressing, toileting, or eating, or with “instrumental activities of daily living” (IADLs), such as meal preparation or managing money. Prior analysis of MCBS data has shown that poorer functional status is strongly correlated with the need for acute care services.43 As Table 1-10 shows, drug coverage does not differ very much by functional status. The most severely disabled beneficiaries have about the same rate of coverage as those with no limitations.
| Functional Status | With drug coverage (000s) | Without drug coverage (000s) | Percent without coverage |
|---|---|---|---|
| No limitations | 19,592 | 8,949 | 31.4% |
| Requires assistance with: | |||
| IADL only | 1,130 | 458 | 28.8% |
| 1 or 2 ADLs | 2,979 | 1,384 | 31.7% |
| 3 or more ADLs | 1,920 | 832 | 30.2% |
| Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996. | |||
Table 1-11 shows coverage rates for any part of 1996 by the number of chronic conditions reported by a beneficiary from a list of ten possible conditions.44 Those reporting 5 or more chronic conditions had a somewhat higher coverage rate than those reporting fewer chronic conditions, and considerably higher than those reporting none. They were also more likely to have coverage through Medicaid or other public programs. Yet, many had gaps in their coverage. Of the more than 74 percent of these beneficiaries that had coverage for at least one month during the year, 21 percent only had coverage for part of the year.45
| Chronic Conditions | With drug coverage (000s) | Without drug coverage (000s) | Percent without coverage |
|---|---|---|---|
| 0 | 2,419 | 1,311 | 35.1% |
| 1-2 | 11,255 | 5,383 | 32.4% |
| 3-4 | 9,221 | 3,986 | 30.2% |
| 5+ | 2,727 | 943 | 25.7% |
| Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996. | |||
Finally, table 1-12 shows coverage rates for any part of 1996 for beneficiaries who did and did not have an inpatient hospital admission during the year. The two groups had about the same rates of coverage.
| Any inpatient admission | With drug coverage (000s) | Without drug coverage (000s) | Percent without coverage |
|---|---|---|---|
| No | 21,118 | 9,522 | 31.1% |
| Yes | 4,503 | 2,101 | 31.8% |
| Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996. | |||
In sum, on three measures that might be related to need for services — self-reported health status, functional status, and inpatient hospital use — MCBS shows no statistically significant difference in drug coverage between healthier and sicker or more disabled individuals. These findings do not rule out the possibility of self-selection; within each defined subpopulation there are likely to be some individuals with a greater need for prescription drugs than others, and these individuals may be more prone to obtain insurance. Moreover, on one measure, number of chronic conditions, sicker individuals do have significantly higher rates of coverage. Evidence about utilization and spending related to the question of selection is reviewed in Chapter 2. These findings, however, do suggest that healthy Medicare beneficiaries are not the only ones who lack coverage for prescription drugs. In general, an equal proportion of covered and non-covered beneficiaries have health problems.
Non-Medicare Population
To put drug coverage for Medicare beneficiaries in perspective, Table 1-13 and Figure 11-6 show sources of drug coverage for the non-Medicare population in 1996.46 Nearly one in four people had no drug coverage during the year.
Figure 1-6. Sources of Drug Coverage for Non-Medicare Population, 1996
Source: Center for Cost and Financing Studies, Agency for Healthcare Research and Quality: Medical Expenditure Panel Survey Household Component
| Source of coverage | Number (000s) | Percent |
|---|---|---|
| Employer | 140,303 | 60.7% |
| Private nongroup and other private | 8,268 | 3.6% |
| Medicaid | 26,291 | 11.4% |
| All other | 2,991 | 1.3% |
| Total with coverage | 177,853 | 77.0% |
| No drug coverage | 53,109 | 23.0% |
| Total | 230,962 | 100.0% |
| Source: Center for Cost and Financing Studies, Agency for Healthcare Research and Quality: Medical Expenditure Panel Survey Household Component, 1996. | ||
The sources of coverage are very different for the non-Medicare population. Three- fifths of the nonelderly, or about 80 percent of those with coverage, have drug coverage through employer groups, compared to a third of the elderly. Many fewer non- Medicare individuals purchase individual coverage. The non-Medicare population is slightly more likely to have drug coverage through Medicaid. While Medicaid benefits for Medicare beneficiaries (specifically QMBs and SLMBs) may or may not include drug coverage, all states provide drug benefits to the non-Medicare classes of Medicaid eligibles, such as pregnant women and children, certain families, and disabled people who do not receive Medicare. Of those without drug coverage, the majority lacked any form of health insurance that covered physician and hospitalization expenses.
Table 1-14 shows coverage rates by ratio of family income to the federal poverty threshold. The near-poor, those between 100 and 200 percent of FPL, have the lowest coverage rate; over a third are without drug coverage. This is the same income range with lowest coverage rates in the MCBS data for Medicare beneficiaries. As with the latter group, individuals at this income threshold may earn too much to qualify for Medicaid but lack the means to obtain their own coverage. However, there is more of a correlation between income and drug coverage in the non-Medicare population compared to the Medicare population. Thus, about 23 percent of Medicare beneficiaries with incomes above 400 percent of poverty lack drug coverage, compared to 14 percent of the non-Medicare population. This probably is in part a result of the types of jobs held by those at different income levels: low-wage workers are less likely to be offered health insurance by their employers.
| Income as a percent of poverty | With drug coverage (000) | Without drug coverage (000) | Percent without coverage |
|---|---|---|---|
| < 100 | 22,425 | 9,987 | 30.8% |
| 100-199 | 26,797 | 15,371 | 36.5% |
| 200-399 | 59,563 | 16,459 | 21.9% |
| > 400 | 68,796 | 11,229 | 14.0% |
| Total | 177,853 | 53,109 | 23.0% |
|
Note: The total includes 335,000 people with negative family income; the sample size for this group is too small for reliable coverage estimates. Source: Center for Cost and Financing Studies, Agency for Healthcare Research and Quality: Medical Expenditure Panel Survey Household Component, 1996. |
|||
Finally, coverage in the non-Medicare population varies by individual health status. Table 1-15 shows rates of drug coverage by self-reported health status. Those reporting poor health have the highest rate of coverage, but those in excellent or very good health have higher coverage rates than those in good or fair health.47 Further research would be needed to determine whether confounding variables, such as employment status or income, are affecting the results.
| Health status | Percent without drug coverage |
|---|---|
| Total | 23.0% |
| Excellent | 22.7% |
| Very good | 21.9% |
| Good | 24.6% |
| Fair | 25.0% |
| Poor | 16.4% |
|
Note: Total row includes persons with unknown perceived health status. Source: Center for Cost and Financing Studies, Agency for Healthcare Research and Quality: Medical Expenditure Panel Survey Household Component, 1996. |
|
Trends in Drug Coverage
Medicare Beneficiaries
The percentage of Medicare beneficiaries with drug coverage for at least one month of the year increased from 57 percent in 1992 to 69 percent in 1996.48 Within the most recent two years for which we have data, the share of beneficiaries with prescription drug coverage for at least part of the year grew from 65 percent in 1995 to 69 percent in 1996, largely because of rapid growth in Medicare HMO enrollment during 1996.
Data for more recent years are not yet available.49 However, as William Scanlon of the General Accounting Office (GAO) and Lisa Alecxih of the Lewin Group recently testified before the House Commerce Committee, there are reasons to believe that coverage rates will erode in the future, if they have not already.50
- Employers are continuing to cut back retiree benefits or requiring enrollees to pay much or all of the cost. Most of the changes affect current workers retiring after a given future date, not current retirees. It is likely that fewer new beneficiaries will have access to this source of drug coverage in coming years. However, given different effective dates of changes in retiree coverage, it is difficult to predict accurately how rapidly coverage might decline.
- Some Medicare HMOs have been reducing their drug benefits. If all 1999 enrollees remained in the same plans in 2000, the number with drug coverage as part of the basic package would drop from 84 percent to 82 percent. This figure does not include enrollees whose plans have terminated their contracts and who may not be able to find drug coverage elsewhere. Continuing growth in the cost of drugs may cause further reductions in drug benefits in the future. It could also increase the premiums charged for thSome Medicare HMOs have been reducing their drug benefits. If all 1999 enr
- In addition to these factors that may lead to a declining rate of coverage in future years, other factors (discussed above) may make the coverage that remains less complete. There is considerable evidence that cost sharing for prescription drugs is increasing and that overall caps on coverage are both becoming more common and are being set at lower levels (especially for Medicare+Choice plans).
General Population
Multi-year data on trends in drug coverage for the non-Medicare population are not readily available. However, there is evidence that health insurance coverage generally has eroded slightly since 1996. Table 1-16 shows the proportion of noninstitutionalized civilians without Medicare receiving coverage from different sources at any time during 1996 and 1998, as measured by the March supplement to the Census Bureau’s Current Population Survey (CPS). While enrollment in employer plans increased somewhat, the gain was more than offset by a sharp decline in Medicaid participation. Over the two years, the share of the non-Medicare population with no coverage grew by nearly a percentage point.
| Source of coverage | 1996 | 1998 |
|---|---|---|
| Employer | 64.7% | 65.5% |
| Other private | 9.9% | 9.2% |
| Medicaid | 11.5% | 9.8% |
| Other | 3.2% | 3.1% |
| None | 18.1% | 18.9% |
|
Note: Columns sum to more than 100 percent because some people had multiple sources of coverage during the year. In 1998, use of Indian Health Service facilities is no longer treated as insurance. Source: Institute for Health Policy Solutions analysis of March 1997 and March 1999 Supplements, Current Population Survey.51 |
||
For people with coverage, prescription drug benefits may be less generous now than in 1996. Because rising prescription drug spending has been driving overall growth in employer health plan costs (see Chapter 2), many employers are reportedly focusing on restraining the cost of drug benefits. In the last year, a reported 32 percent of employers with 500 or more workers modified drug benefit design, for example, by increasing financial incentives for participants to use generic or on-formulary drugs. Ten percent have limited coverage for some new drugs or other treatments.
Endnotes
1 - Access to Medicaid drug coverage by Medicare beneficiaries will be discussed below.
2 - Medicare+Choice plans are not literally supplemental plans but serve as an alternative to the traditional Medicare fee-for-service program and typically include supplemental benefits.
3 - Congressional Research Service, Health Insurance and the Uninsured: Background Data and Analysis, Washington, 1988.
4 - Mercer/Foster Higgins. National Survey of Employer-Sponsored Health Plans, 1999. These results are based on a survey of employers with 500 or more employees.
5 - “Brand-name” drugs are those sold by the manufacturer that initially developed them or by another manufacturer with a license from the initial developer. “Generic” drugs are those sold by other manufacturers after the patent protection for a brand-name drug has expired; they are similarly formulated and have biologic effects equivalent to those of a brand-name drug. Copayments may also vary depending on whether the drug is on the PBMs preferred list or formulary. According to the Mercer/Foster Higgins survey, in 1999, 32 percent of large employers raised one or more cost-sharing components to their benefit plans.
6 - For more information on the role of PBMs see Anna Cook, et al. The Role of PBMs in Managing Drug Costs: Implications for a Medicare Drug Benefit. Report prepared for the Kaiser Family Foundation, January 2000.
7 - Kaiser Family Foundation and Health Research and Educational Trust (HRET), Employer Health Benefits: 1999 Annual Survey. Kaiser-HRET estimates that 34 percent of employees were in carve-outs in 1999.
8 - Testimony of Jeff Sanders, Pharmaceutical Care Management Association, before the Senate Finance Committee, 1999.
9 - Kaiser /HRET. For this survey, large employers are defined as those employing 200 or more workers.
10 - Mercer/Foster Higgins.
11 - Kaiser/HRET.
12 - Mercer/Foster Higgins.
13 - Kaiser/HRET.
14 - Mercer/Foster Higgins.
15 - Kaiser/HRET.
16 - In results using the MCBS in this report, all beneficiaries in Medicare+Choice plans are classed in the Medicare+Choice category even though some have supplemental coverage paid for by their former employer.
17 - Mercer/Foster Higgins.
18 - Hewitt Associates, Retiree Health Coverage: Recent Trends and Employer Perspectives on Future Benefits, Kaiser Family Foundation, Oct. 1999.
19 - Insurers may offer a high deductible version of Plan J which generally has lower premiums than the regular Plan J. Under this high-deductible option, the purchaser pays $1,530 out-of-pocket per year before the plan pays anything. The purchaser must still meet a $250 deductible on prescription drugs before drug coverage begins.
20 - Rice et al. found that 15 percent of nonstandardized policies sold in a sample of six states in 1994 included prescription drug benefits. There is no data on the value of these benefits. “The Impact of Policy Standardization on the Medigap Market,” Inquiry, 1997.
21 - Federal law permits insurers in Minnesota, Massachusetts, and Wisconsin to offer different state- regulated Medigap policies. Wisconsin law requires Medicare Supplement policies to contain a catastrophic prescription drug benefit. This benefit must cover at least 80% of the charges for outpatient prescription drugs after the purchaser meets a drug deductible of no more than $6,250 per calendar year.
22 - Medigap carriers must also offer open enrollment to beneficiaries who have lost supplemental coverage as a result of the termination of a Medicare risk HMO contract, or in certain other circumstances. However, they are not required to offer one of the plans with drug coverage during this special open enrollment period.
23 - HHS analysis.
24 - MCBS.
25 - HCFA(now known as CMS) analysis of Medicare Compare. MCBS data capture information on drug coverage in 1996 for enrollees of Medicare managed care plans who have employer-sponsored drug coverage through the Medicare contracting organization as well as information on beneficiaries whose source of drug coverage is a ”high option” plan offered by a Medicare contracting organization (today’s M+C organization). HCFA(now known as CMS) administrative files, used for the 1999 analysis, do not include information on whether beneficiaries obtain drug coverage by selecting high option plans rather than as part of the basic benefit package. This understates the overall proportion of beneficiaries with drug coverage in M+C plans.
26 - HCFA(now known as CMS) analysis.
27 - HCFA(now known as CMS), Medicare+Choice: Changes for the Year 2000, 1999.
28 - The discussion here omits a variety of other special population groups defined in law and accounting for very small numbers of participants.
29 - MCBS.
30 - One reason is that states cannot receive Medicaid rebates on drugs provided through HMOs.
31 - National Pharmaceutical Council. Pharmaceutical Benefits under State Medical Assistance Programs, December 1998.
32 - National Conference of State Legislatures, news release, March 10, 2000.
33 - This count does not include disease-specific programs that may provide some assistance with drug costs, such as the AIDS drug assistance programs funded by the Ryan White CARE Act.
34 - Direct communication, PhRMA.
35 - IMS Health. Integrated Share of Voice Report. (It should be noted that the MCBS and MEPS data on utilization and spending to be presented in Chapter 2 exclude samples or other free medications.)
36 - Stuart, Bruce, Dennis Shea, and Becky Briesacher, Prescription Drug Costs for Medicare Beneficiaries: Coverage and Health Status Matter, New York, Commonwealth Fund, Issue Brief, January 2000.
37 - Note that the sample for Table 1-2 is somewhat different from the Table 1-1. Numbers for Table 1-2 are derived from an MCBS sample of persons enrolled in Medicare for the entire year and thus excludes decedents and new Medicare entrants.
38 - These data are based on a beneficiary’s source of coverage in December. The proportions would probably vary somewhat if a different month were used as the basis for comparison.
39 - Bruce Stuart, et al., “Dynamics in Prescription Coverage of Medicare Beneficiaries: Finders, Losers, Switchers,” Commonwealth Fund Issue Brief, forthcoming.
40 - Income includes that of the beneficiary and a spouse, if any, not that of other family or household members.
41 - Note sample size for part-year coverage is based on an MCBS sample of persons enrolled in Medicare for the entire year and thus excludes decedents and new Medicare entrants. Bruce Stuart, University of Maryland, direct communication.
42 - Stuart et al.
43 - J. Feder and J. Lambrew, “Why Medicare Matters to People Who Need Long-Term Care,” Health Care Financing Review, (Winter 1996): 99-112.
44 - The conditions were heart problems, cancer, arthritis, lung disease, mental disorders, Alzheimer’s, diabetes, hypertension, bone disease, and stroke.
45 - Stuart et al.
46 - Individuals who had drug coverage at any time during the year are classified as covered in this survey.
47 - The differences between “poor” and “fair,” “very good” and “good,” and “very good” and “fair” are statistically significant; those between “excellent” and the other groups are not.
48 - Because of changes in the methodology used by MCBS to measure drug coverage, figures from 1992 - 1994 are not fully comparable with data collected in 1995 and 1996.
49 - Current Population Survey data on general insurance coverage are available. However, the CPS is not a reliable source for estimating supplementary coverage of Medicare beneficiaries, because it does not separately identify Medicare HMO enrollees. Some may report themselves as having Medicare only, others as having private nongroup coverage. Between 1996 and 1998, the CPS shows the share of beneficiaries with employer coverage and with Medicaid remaining approximately the same. It shows a sharp decline in other private coverage; nearly 1.6 million fewer beneficiaries reported this coverage in 1998 than in 1996. But there is no way of knowing how much of the change is related to actual loss of coverage, shifts to HMO enrollment, or simply changes in how respondents reported their coverage.
50 - Testimony before the House Commerce Subcommittee on Health and Environment, “Seniors’ Access to Affordable Prescription Drugs: Models for Reform,” February 16, 2000. For a fuller analysis of factors that might lead to declining coverage, see the White House report, Disturbing Truths and Dangerous Trends: The Facts about Medicare Beneficiaries and Prescription Drug Coverage, July 1999.
51 - The Census Department advises that Medicaid coverage is underreported in the CPS compared with enrollment and participation data from the HCFA(now known as CMS). Changes in Medicaid coverage estimates from one year to the next should be viewed with caution.
Chapter 2. Effects of Prescription Drug Coverage on Spending and Utilization
Insurance coverage that includes coverage of prescription drugs plays a critical role in assuring access to needed medications. People with coverage not only fill more prescriptions than people without coverage; they are likely to have access to a broader array of therapies, including more costly therapies. People without drug coverage face greater financial burdens and may sometimes be unable to follow the courses of treatment ordered by their physicians. There are even some indications that physicians themselves may recommend different therapies to people with and without coverage.
As will be seen, the effects of prescription drug coverage persist across different age groups, income levels, and health statuses. Coverage increases prescription drug utilization, changes the mix of different drugs received, and reduces financial burdens for all population groups. However, access to drug coverage is most important for the elderly, simply because they require more medications, including a higher prevalence of long-term maintenance drugs for chronic conditions.
This chapter will first present detailed comparisons of utilization and spending for Medicare beneficiaries and the total population with and without drug coverage. Second, it will examine some of the possible reasons for those differences and will consider the consequences of being without coverage. Finally, it will summarize trends in utilization and spending and some of the factors that influence these changes.
Key findings include:
- Medicare beneficiaries with coverage fill nearly one-third more prescriptions than those without coverage.
- Although total drug spending for beneficiaries with coverage is nearly two-thirds higher, those without coverage pay nearly twice as much out of pocket ($463 versus $253).
- On average, beneficiaries with coverage pay out of pocket for about one-third of their total spending on drugs. However, the share of spending paid out of pocket varies by source of coverage, from 58 percent for those with Medigap coverage to 20 percent for those with Medicaid.
- Differences in utilization and spending between Medicare beneficiaries with and without drug coverage generally hold up across different income levels, ages, health status, and other categories.
- Drug insurance makes an especially large difference in dollar terms for those in the poorest health. Among beneficiaries with five or more chronic conditions, those with coverage had much higher total spending ($1,402 versus $944) and much lower out-of-pocket spending ($412 versus $944) than beneficiaries without coverage.
- Among people who are not Medicare beneficiaries, similar differences in utilization and spending exist between prescription drug users with and without coverage. Those with coverage for drugs fill two-thirds more prescriptions but spend a third less out of pocket than those without coverage.
- About a third of Medicare beneficiaries accounted for three-fourths of beneficiaries’ total drug spending in 1996. Only 13 percent had no spending at all. Spending on prescription drugs in the non-Medicare population is even less evenly distributed.
- Self-selection does not explain the difference in spending between Medicare beneficiaries with and without drug coverage. Even among beneficiaries with the same poor health status, more prescriptions are written for people with coverage, and people without coverage are less likely to fill prescriptions.
- Prescription drugs take up about one sixth of all health spending by the elderly. Out of pocket spending for prescription drugs is a larger proportion of health spending for the elderly than for younger people. Prescription drug spending also accounts for a larger share of spending by people with low incomes than it does for people with higher incomes.
- The burden of prescription drug costs creates access problems for some beneficiaries. High spenders with incomes below the poverty line spend more than one-fourth of their income on drugs. Among Medicare beneficiaries, 10 percent of those with only Medicare coverage report not being able to afford a needed drug, compared to 2 percent of those with a non-Medicaid supplement.
- Drug spending has grown more quickly than other health spending throughout the 1990’s. Price increases, higher utilization, and the use of newer, more expensive drugs all play a part in increasing drug spending.
Comparisons of Utilization and Spending
This section will present, for Medicare beneficiaries and for the non-Medicare population, comparisons of utilization by covered and noncovered people grouped by various demographic and health characteristics. Unless otherwise noted, all results reported in this chapter are statistically significant at the 0.05 level, based on a two-tailed test. See the Introduction of this report for details.
The comparisons here are limited to simple descriptive tables, primarily focusing on the ratio between people with and without drug coverage. It is important to note that these descriptive comparisons, which look at one factor at a time, may not isolate the key determinants of drug spending. Multivariate analysis would be needed to establish which factors are most important in explaining differences between the covered and noncovered populations.
Most of the estimates provided in this chapter are drawn from Medicare Current Beneficiary Survey (MCBS) and Medical Expenditure Panel Survey (MEPS) data on prescription drugs. It should be noted that over-the-counter drugs are not included here. Drugs that are available both over-the-counter and in prescription strength are included if the drug was obtained in prescription strength.
MCBS and MEPS use different methods to gather data on utilization and spending. However, both find the same patterns in utilization and spending for people with and without coverage. Their estimates of the magnitude of the gaps between the covered and uncovered are sometimes different, though rarely contradictory. A detailed summary of the survey methods and of the differences in results is provided in appendix B.
Overall differences
Table 2-1, drawn from 1996 MCBS data, compares drug utilization and spending for Medicare beneficiaries with and without drug coverage. As in the previous chapter, beneficiaries are included in the covered category if they had drug coverage at any point during the year. The next section of this chapter will explore the differences in spending and utilization depending on the duration of their coverage.
| All Medicare beneficiaries | Beneficiaries who filled at least one prescription | |||||
|---|---|---|---|---|---|---|
| Covered | Not covered | Ratio, covered/ not covered | Covered | Not covered | Ratio, covered/ not covered | |
| Percent who filled at least one prescription | 89.4% | 80.3% | 1.11 | 100% | 100% | 1.00 |
| Average # of prescriptions | 21.14 | 16.01 | 1.32 | 23.64 | 19.93 | 1.19 |
| Average annual spending | $768.90 | $463.15 | 1.66 | $859.99 | $576.43 | 1.49 |
| Retail price per prescription | $36.37 | $28.93 | 1.26 | $36.37 | $28.93 | 1.26 |
| Average out-of-pocket spending | $252.65 | $463.15 | 0.55 | $282.58 | $576.43 | 0.49 |
| % out-of-pocket | 33% | 100% | 0.33 | 33% | 100% | 0.33 |
| Average out-of-pocket per prescription | $11.95 | $28.93 | 0.41 | $11.95 | $28.93 | 0.41 |
| Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996. | ||||||
The table shows that:
- Beneficiaries who have prescription drug coverage fill nearly one-third more prescriptions than those who do not have coverage, and total drug spending for beneficiaries with coverage is nearly two-thirds higher.
- The cost of prescriptions filled by beneficiaries with coverage is higher on average than the cost of prescriptions filled by beneficiaries without coverage. This is not because the price of any specific medication is higher for people with coverage. Instead, the difference seems to come from the fact that people with drug coverage receive a different mix of drugs than noncovered people receive, or different size prescriptions. The next chapter will show that for a given prescription, prices are generallyThe cost of prescriptions filled by beneficiaries with coverage is higher on average than the
- Beneficiaries with coverage pay out of pocket for 33 percent of their total spending on prescription drugs; those without coverage, of course, pay 100 percent. For each prescription they fill, beneficiaries without coverage pay on average more than twice as much out of pocket as those who have coverage. Overall, their annual out-of-pocket costs are nearly twice as high -- $200 more -- even though they use fewer medications (see Figure 2-1).
Figure 2-1. Out-of-pocket and Insurer Spending on Prescription Drugs by Medicare Beneficiaries with and without Drug Coverage, 1996
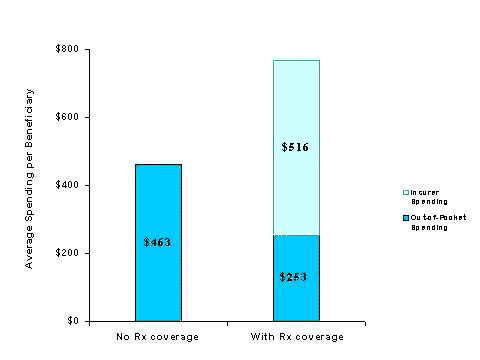
Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996.
- Beneficiaries with coverage are more likely to fill at least one prescription. The differences between those with and without coverage persist even when looking only at beneficiaries who fill prescriptions (see Figure 2-2).
Figure 2-2. Medicare Beneficiaries Who Filled a Prescription and Number of Prescriptions Filled, by Coverage Status, 1996
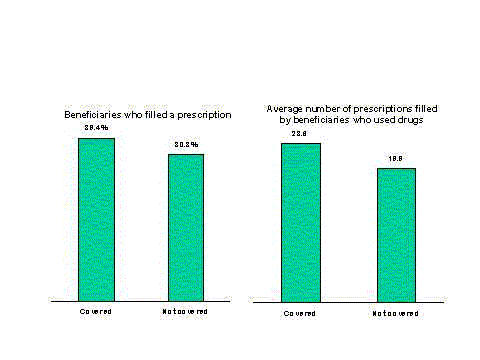
Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996
Table 2-2 presents similar information for the non-Medicare population.1 The differences in utilization and total spending are much greater. This is because a very large number of non-Medicare individuals without coverage — nearly two-thirds — receive no prescription drugs at all. In contrast, only 13 percent of Medicare beneficiaries fill no prescription during the year.2 If one considers only users of prescription drugs, the patterns are more similar to those shown in table 2-1. Those who have coverage receive more, and more costly, prescriptions.3 Those without coverage pay over twice as much out-of-pocket for each prescription they fill (see Figure 2-3).
| Total non-Medicare population | People who filled at least one prescription | |||||
|---|---|---|---|---|---|---|
| Covered | Not covered | Ratio, covered/ not covered | Covered | Not covered | Ratio, covered/ not covered | |
| Percent who filled at least one prescription | 70.0 | 33.9 | 2.06 | 100% | 100% | 1.00 |
| Average # of prescriptions | 6.80 | 2.02 | 3.36 | 9.73 | 5.92 | 1.64 |
| Average annual spending | $222.01 | $58.94 | 3.77 | $317.64 | $172.64 | 1.84 |
| $ per prescription | $32.65 | $29.17 | 1.12 | $32.65 | $29.17 | 1.12 |
| Average out-of-pocket spending | $80.59 | $58.94 | 1.37 | $115.30 | $172.64 | 0.67 |
| % out-of-pocket | 36% | 100% | 0.36 | 36% | 100% | 0.36 |
| Average out-of-pocket per prescription | $11.85 | $29.17 | 0.41 | $11.85 | $29.17 | 0.41 |
| Source: Center for Cost and Financing Studies, Agency for Healthcare Research and Quality: Medical Expenditure Panel Survey Household Component, 1996. | ||||||
Figure 2-3. Out-of-pocket and Insurer Spending on Prescription Drugs by Non-Medicare Beneficiaries with and without Drug Coverage, 1996
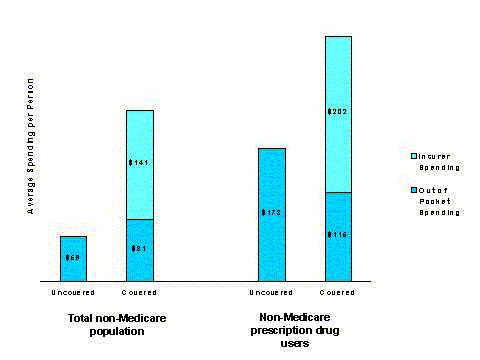
Source: Center for Cost and Financing Studies, Agency for Healthcare Research and Quality: Medical Expenditure Panel Survey Household Component, 1996.
It should be emphasized that, throughout this chapter, “out-of-pocket costs” include only the net costs to an individual, after any insurance payments, for any prescriptions received. Premiums paid for insurance coverage are not included. This analysis excludes premiums because it is difficult with survey data to separate drug premiums from overall insurance premiums. In fact, in many cases, calculating truly separate drug premiums would be impossible even with complete information. In many benefit packages, prescription drugs are considered a key piece, and the interactions with other benefits make it difficult to separate the incremental cost of a drug benefit from the package as a whole.4 Moreover, the insurance that these premiums buy has a value in and of itself, even if there is no drug utilization.
Duration of coverage
Table 2-3, drawn from an independent analysis of MCBS data by Bruce Stuart et al. for the Commonwealth Fund, shows differences in utilization and spending for Medicare beneficiaries who had drug coverage throughout 1996, during part of the year, or at no time during the year. It has been observed for other types of medical coverage that people with part-year coverage use more services during their covered period than people with full-year coverage use during a similar period; in effect, they are using their coverage to deal with previously unattended problems. However, this sort of catch-up effect does not appear to be applicable for prescription drugs. While someone can postpone elective surgery, for example, people who do not obtain needed medications during a period without coverage cannot make up for this when they obtain coverage.
Instead, it seems likely that people who have coverage for only part of a year will act like noncovered people during their period without coverage and like other covered people during their period with coverage. In fact, beneficiaries even with 7 to 9 months of coverage spend only 12 percent ($58) more than beneficiaries who are uncovered all year. Table 2-3 and Figure 2-4 show that the more substantial increase in utilization occurs when a beneficiary has drug coverage for at least 10 months.
| Months of drug coverage during 1996 | |||||||
|---|---|---|---|---|---|---|---|
| Total | Never covered | 1-3 months | 4-6 months | 7-9 months | 10-11 months | Always covered | |
| Annual prescription drug spending | $688 | $468 | $524 | $539 | $526 | $655 | $828 |
| Annual prescription spending out of pocket | $316 | $468 | $341 | $341 | $204 | $237 | $219 |
| Proportion of drug spending paid out of pocket | 46% | 100% | 65% | 63% | 39% | 36% | 26% |
| Proportion of beneficiaries | 95%* | 28% | 2% | 5% | 5% | 2% | 53% |
| *The total equals 95% because duration of insurance coverage could not be determined for 4.7% of beneficiaries (had claims paid but did not report coverage. Source: Bruce Stuart tabulations of MCBS, 1996, for noninstitutionalized beneficiaries enrolled in Medicare for the entire year. |
|||||||
Figure 2-4. Out-of-pocket and Insurer Spending on Prescription Drugs by Medicare Beneficiaries, by Duration of Drug Coverage, 1996
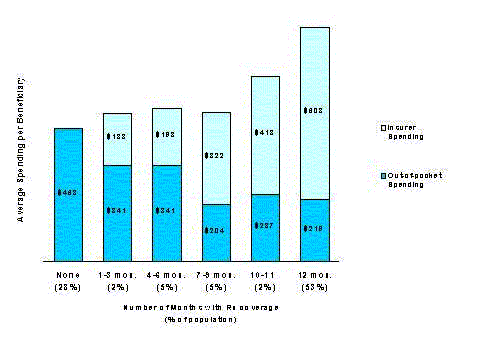
Note: 4.7% of beneficiaries had drug coverage for an unknown number of months (had claims paid but did not report coverage).
Source: Bruce Stuart tabulations of MCBS, 1996 for noninstitutionalized beneficiaries enrolled in Medicare for the entire year.
Much of the MCBS data presented elsewhere in this report, and all the MEPS data, treat individuals as having drug coverage if they have coverage at any time during the year. Because people with part-year coverage have lower utilization and spending than those with full-year coverage, their inclusion lowers the averages for the covered group. As a result, the real differences between people who do and do not have coverage at a given point in time are understated throughout this report.
Source of Coverage
Table 2-4 and Figure 2-5 show that utilization and spending by Medicare beneficiaries varies according to their primary source of supplemental coverage.5 The gaps between beneficiaries with and without drug coverage also vary.
| Source of coverage | Covered | Not covered | Ratio, covered/not covered | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Average number of prescriptions | Average total spending | Average out-of-pocket spending | Out-of-pocket as % of total spending | Average number of prescriptions | Average total spending (all out-of-pocket) | Average number of prescriptions | Average total spending | Average out-of-pocket spending | |
| TOTAL | 21.14 | $768.90 | $252.65 | 33% | 16.01 | $463.15 | 1.32 | 1.66 | 0.55 |
| Risk HMO | 17.43 | $573.02 | $188.90 | 33% | * | $151.33 | * | 3.79 | 1.25 |
| Medicaid | 28.50 | $882.13 | $177.57 | 20% | 14.95 | $380.05 | 1.91 | 2.32 | 0.47 |
| Employer Sponsored | 19.07 | $805.89 | $238.72 | 30% | 17.42 | $530.03 | 1.10 | 1.52 | 0.45 |
| Individually Purchased | 20.78 | $711.28 | $416.41 | 58% | 17.61 | $524.70 | 1.18 | 1.36 | 0.79 |
| Other | 27.43 |
$790.96 |
$292.71 | 37% | 17.87 | $543.66 | 1.53 | 1.45 | 0.54 |
| Medicare Only | 12.39 | $326.65 | |||||||
|
* Sample size is too small to produce a reliable estimate. Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996. |
|||||||||
Among beneficiaries with drug coverage, both total spending and the proportion that beneficiaries pay out-of-pocket vary by type of insurance. Beneficiaries who have individually purchased (mostly Medigap) insurance pay a higher proportion of their drug costs than any other covered group and have relatively low total spending compared to other coverage groups. Beneficiaries enrolled in Medicaid who have drug coverage have the highest utilization and spending and the lowest out-of-pocket costs of any covered group.6
Figure 2-5. Out-of-pocket and Insurer Spending on Prescription Drugs by Medicare Beneficiaries, by Type of Drug Coverage, 1996
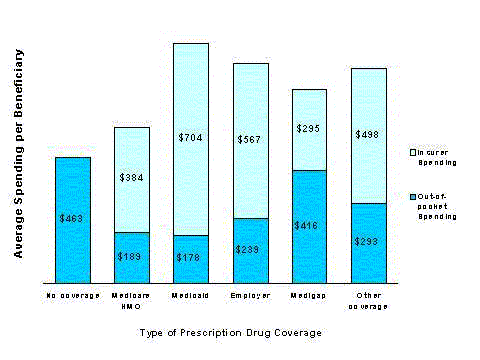
Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996.
With few exceptions, Medicare beneficiaries with drug coverage had more prescriptions, higher spending, and lower out-of-pocket costs than those without drug coverage as a whole and within each coverage group.7
- The spending differences between Medicaid beneficiaries with and without drug coverage are dramatic. Beneficiaries who have Medicaid with drug coverage get nearly twice as many prescriptions and spend over twice as much on drugs as beneficiaries who receive Medicaid assistance only with cost sharing or premiums. Very low-income people who lack drug coverage may be least likely to be able to purchase needed medications on their own.
- Beneficiaries with employer-sponsored supplemental insurance show the smallest utilization difference between those with and without a drug benefit; the difference in the number of prescriptions per person is not statistically significant. The difference in total spending is much larger, because prescriptions received by those with a drug benefit were 39 percent more costly than those received by retirees without a drug benefit. Despite their higher total spending, those with drug coverage had out-of-pockeBeneficiaries with employer-sponsored supplemental insurance show the smallest u
- The utilization difference for people with individually purchased coverage with and without a drug benefit is also quite small. In contrast to those with employer-sponsored insurance, however, those with drug coverage spend nearly as much out of pocket (almost 80 percent as much) as those without it. This is partly due to the deductible and cost-sharing requirements imposed by standard Medigap plans. On average, beneficiaries with a drug benefit and Medigap pay 58 percent of their costs out of pocket.
There are also notable differences among beneficiaries without drug coverage based on whether they had other supplemental coverage. With the exception of beneficiaries in HMOs, people with no drug coverage but some supplemental coverage had higher spending than people who had only Medicare coverage. One possible explanation is that people with no supplement are healthier than people who choose to obtain some form of supplemental coverage. Another possible factor is that beneficiaries without a supplement were less likely to see a physician, and hence less likely to receive a prescription. The issues of self-selection, moral hazard, and lack of access will be considered further below.
Income
Table 2-5 and Figure 2-6 show average prescription drug spending and average out-of-pocket spending for Medicare beneficiaries by income.8 Average spending for people with coverage is consistently higher than for people without coverage across income groups.9 It might be expected that spending differences would diminish steadily with higher income, because higher-income people without coverage are better able to pay for drugs on their own. The difference in average spending for covered and noncovered beneficiaries in poverty is indeed greater than for other income groups. However, among the higher income groups, there is no significant difference in the size of the spending gap between covered and noncovered beneficiaries. This suggests that insurance coverage for prescription drugs matters, irrespective of income.
| Income as a percent of poverty | Covered | Not Covered | Ratio, Covered/ Not Covered | |||
|---|---|---|---|---|---|---|
| Average Total Spending | Average Out-of-pocket Spending | Out-of-pocket as % of Total Spending | Average Total Spending (all out-of-pocket) | Average Total Spending | Average Out-of-pocket Spending | |
| Total | $769 | $253 | 33% | $463 | 1.66 | 0.55 |
| <Poverty | $800 | $200 | 25% | $368 | 2.18 | 0.54 |
| Poverty-135% | $767 | $269 | 35% | $476 | 1.61 | 0.57 |
| 136-150% | $673 | $272 | 40% | $555 | 1.21 | 0.49 |
| 151-175% | $790 | $279 | 35% | $453 | 1.74 | 0.62 |
| 176-200% | $791 | $255 | 32% | $512 | 1.54 | 0.50 |
| 201-300% | $778 | $284 | 36% | $487 | 1.60 | 0.58 |
| 301-400% | $782 | $264 | 34% | $453 | 1.72 | 0.58 |
| >400% | $717 | $248 | 35% | $525 | 1.37 | 0.47 |
| Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996. | ||||||
Figure 2-6. Out-of-pocket and Insurer Spending on Prescription Drugs by Medicare Beneficiaries with and without Drug Coverage, by Income, 1996
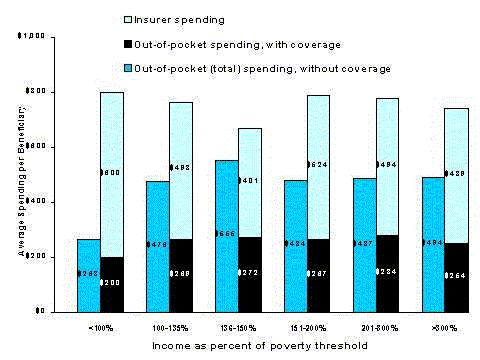
Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996.
Among covered beneficiaries, those below the poverty threshold pay the smallest share of their own costs out of pocket. Poor beneficiaries are more likely to have drug coverage through Medicaid, which imposes only nominal copayments. Still, even with drug coverage, beneficiaries in poverty paid 25 percent of their own costs. This may be partly because people are counted as covered if they have coverage for any part of the year. The out-of-pocket costs might have been incurred by part-year eligibles during a period without coverage. Another possible factor is the fact that some state Medicaid programs limit the number of prescriptions beneficiaries may receive in a given period.
Table 2-6 shows spending by income for the non-Medicare population. Again, it is helpful to look at people who used any prescription drugs, rather than at the total population. Among users, patterns for the non-Medicare population are similar to those shown in Table 2-5 for the Medicare population as a whole. In all income ranges, there is a difference between spending by those with coverage and those without.10 Like Medicare beneficiaries, covered non-Medicare beneficiaries pay the smallest share of their own costs – just over a fourth – when they are in poverty. However, actual out-of-pocket spending for drug users in this group is only $76 on average, as opposed to $200 for covered Medicare beneficiaries in poverty.
| Income as a percent of poverty | Covered | Not Covered | Ratio, Covered/ Not Covered | |||
|---|---|---|---|---|---|---|
| Average Total Spending | Average Out-of-pocket Spending | Out-of-pocket as % of Total Spending | Average Total Spending (all out-of-pocket) | Average Total Spending | Average Out-of-pocket Spending | |
| Total non-Medicare population | $222.01 | $80.59 | 36% | $58.94 | 3.77 | 1.37 |
| <Poverty | $191.14 | $49.64 | 26% | $62.18 | 3.07 | 0.80 |
| 100-200% | $195.96 | $76.98 | 39% | $63.45 | 3.09 | 1.21 |
| 200-400% | $210.66 | $79.10 | 38% | $51.33 | 4.10 | 1.54 |
| >400% | $252.34 | $93.58 | 37% | $61.36 | 4.11 | 1.53 |
| People using prescription drugs | $317.64 | $115.30 | 36% | $172.64 | 1.84 | 0.67 |
| <Poverty | $291.65 | $75.74 | 26% | $176.60 | 1.65 | 0.43 |
| 100-200% | $285.74 | $112.26 | 39% | $187.13 | 1.53 | 0.60 |
| 200-400% | $305.98 | $114.89 | 38% | $156.70 | 1.95 | 0.73 |
| >400% | $346.89 | $128.65 | 37% | $172.10 | 2.02 | 0.75 |
|
Note: The totals include persons with negative family income. Source: Center for Cost and Financing Studies, Agency for Healthcare Research and Quality: Medical Expenditure Panel Survey Household Component, 1996. |
||||||
Health status and other health indicators
Not surprisingly, as health status worsens, beneficiaries use more drugs. However, Table 2-7 and Figure 2-7 show that while utilization rises, a gap remains between beneficiaries with and without coverage. Within each health status category, beneficiaries with coverage are more likely to fill at least one prescription, and fill more prescriptions when they do use drugs. Beneficiaries without coverage who are in fair or poor health are as likely to fill no prescriptions as covered beneficiaries in very good health.
| % filling at least one prescription | Number of prescriptions filled by users | ||||
|---|---|---|---|---|---|
| Covered | Not covered | Covered | Not covered | Ratio, covered/ not covered | |
| Excellent | 80% | 67% | 13.76 | 11.42 | 1.20 |
| Very Good | 86% | 79% | 17.18 | 15.45 | 1.11 |
| Good | 92% | 84% | 22.40 | 20.36 | 1.10 |
| Fair | 96% | 86% | 33.22 | 26.44 | 1.26 |
| Poor | 96% | 86% | 39.67 | 31.43 | 1.26 |
| Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996. | |||||
Figure 2-7. Percentage of Medicare Beneficiaries with and without Drug Coverage Filling at Least One Prescription, by Health Status, 1996
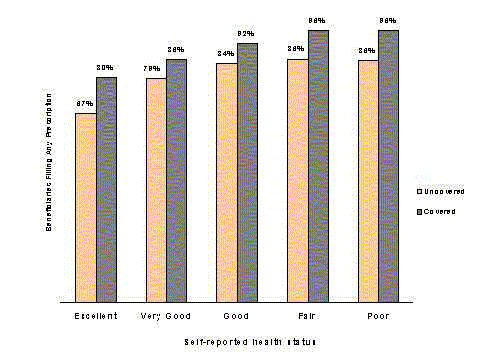
Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996.
Table 2-8 and Figure 2-8 show that spending follows the utilization pattern: as Medicare beneficiaries report poorer health, their spending on prescription drugs increases. Again, within each health status category, beneficiaries with drug coverage have higher spending than the noncovered. Spending for beneficiaries in poor health who had drug coverage was $590 more than for those who lacked coverage; for beneficiaries with fair health, the difference was $480. Beneficiaries spent nearly twice as much out of pocket as those who had coverage unless they were in excellent health.
| Covered | Not Covered | Ratio, Covered/Not Covered | ||||
|---|---|---|---|---|---|---|
| Self-reported health status | Average total spending | Average out-of-pocket spending | Out-of-pocket as % of total spending | Average total spending (all out-of-pocket) | Average total spending | Average out-of-pocket spending |
| Total | $769 | $253 | 33% | $463 | 1.66 | 0.55 |
| Excellent | $414 | $141 | 34% | $207 | 2.01 | 0.68 |
| Very Good | $554 | $190 | 34% | $382 | 1.45 | 0.50 |
| Good | $759 | $256 | 34% | $497 | 1.53 | 0.51 |
| Fair | $1,120 | $350 | 31% | $640 | 1.75 | 0.55 |
| Poor | $1,340 | $423 | 32% | $749 | 1.79 | 0.56 |
| Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996. | ||||||
Figure 2-8. Out-of-pocket and Insurer Spending for Medicare Beneficiaries with and without Drug Coverage, by Health Status, 1996
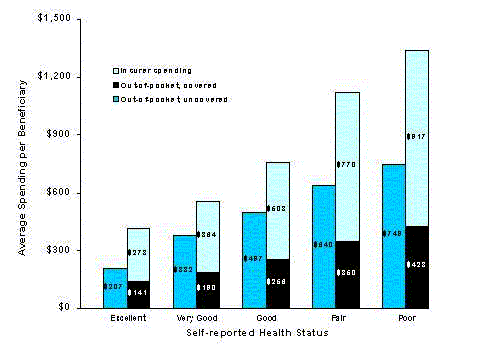
Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996.
Table 2-9 shows that the same patterns are reinforced when beneficiaries are split according to the duration of their drug coverage. Beneficiaries who always had drug coverage had nearly twice the spending of beneficiaries who never had drug coverage at any time during the year, regardless of health status. Those in fair or poor health who were covered for only part of the year had total spending slightly closer to those who never had coverage.
| Always Covered | Sometimes Covered | Never Covered | |||||
|---|---|---|---|---|---|---|---|
| Self-reported health status | Average total spending | Average out-of-pocket spending | Out-of-pocket as % of total spending | Average total spending | Average out-of-pocket spending | Out-of-pocket as % of total spending | Average total spending (all out-of-pocket) |
| Total | $828 | $219 | 26% | $626 | $358 | 57% | $468 |
| Excellent/Good | $655 | $184 | 28% | $480 | $282 | 59% | $386 |
| Fair/Poor | $1,327 | $318 | 24% | $1,003 | $552 | 55% | $732 |
|
Source: Bruce Stuart, Dennis Shea, and Becky Briesacher, “Prescription Drug Costs for Medicare Beneficiaries: Coverage and Health Status Matter,” New York, Commonwealth Fund Issue Brief, January 2000. |
|||||||
That spending and utilization differences persist across different health statuses suggests that the overall difference in spending for covered and noncovered people cannot simply be attributed to different levels of need for prescription drugs between those with and without drug coverage. However, self-reported health status is not necessarily a good indicator of relative need for drugs or other health services. Table 2-10 uses three other indicators to compare spending by covered and noncovered Medicare beneficiaries: number of chronic conditions, number of functional limitations, and use of inpatient services during the year.
| Covered | Not covered | Ratio, covered/not covered | ||||
|---|---|---|---|---|---|---|
| Average total spending | Average out-of-pocket Spending | Out-of-pocket as % of total spending | Average total spending (all out-of-pocket) | Average total spending | Average Out-of-pocket Spending | |
| Total | $769 | $253 | 33% | $463 | 1.66 | 0.55 |
| Chronic Conditions | ||||||
| 0 | $222 | $83 | 37% | $148 | 1.50 | 0.56 |
| 1-2 | $600 | $191 | 32% | $342 | 1.76 | 0.56 |
| 3-4 | $932 | $325 | 35% | $617 | 1.51 | 0.53 |
| 5+ | $1,401 | $412 | 29% | $944 | 1.48 | 0.44 |
| Functional status | ||||||
| No Limitations | $662 | $218 | 33% | $415 | 1.60 | 0.53 |
| IADL Only1 | $1,160 | $394 | 34% | $666 | 1.74 | 0.59 |
| 1 or 2 ADLs2 | $1,051 | $348 | 33% | $582 | 1.80 | 0.60 |
| 3+ ADLs2 | $1,190 | $378 | 32% | $674 | 1.77 | 0.56 |
| Inpatient use | ||||||
| No admission | $699 | $230 | 33% | $412 | 1.70 | 0.56 |
| Admission | $1,097 | $361 | 33% | $694 | 1.58 | 0.52 |
| 1 Beneficiaries who need assistance with one or more instrumental activities of daily living, such as meal preparation or managing money. 2Beneficiaries who need assistance with activities of daily living, such as bathing, dressing, toileting, or eating. Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996. |
||||||
On all three measures, the pattern is the same as for self-reported health status. Beneficiaries who report more chronic conditions, worse functional status, or a hospital stay during the year spend more on drugs whether or not they have coverage. In each category, beneficiaries with drug coverage spend much more than uncovered beneficiaries in the same category; at the same time, their out-of-pocket spending is much lower.
Because spending rises so dramatically with worsening health status, the dollar gaps between the covered and the noncovered rise with poorer health. For example, among beneficiaries with five or more chronic conditions, those with coverage had average total spending $457 higher than that for beneficiaries without coverage, while the difference for beneficiaries with no chronic conditions was only $74. Out of pocket spending for uncovered beneficiaries with five or more chronic conditions was $532 higher than for those with coverage. Drug insurance clearly makes an important difference for people with severe health problems.
Table 2-11 provides MEPS data for the total population (including people with and without Medicare) by self-reported health status. The figures include only people who used any prescription drugs. Again, spending rises with poorer health status for both the covered and the noncovered population,11 and the dollar difference in spending is greatest for those in poor health.12
| Covered | Not covered | Ratio, covered/not covered | ||||
|---|---|---|---|---|---|---|
| Average number of prescriptions | Average annual spending | Average number of prescriptions | Average annual spending | Average number of prescriptions | Average annual spending | |
| TOTAL | 12.59 | $427.75 | 8.77 | $269.04 | 1.43 | 1.59 |
| Excellent | 6.00 | $175.75 | 4.34 | $144.38 | 1.38 | 1.22 |
| Very good | 10.22 | $339.21 | 7.66 | $220.50 | 1.33 | 1.54 |
| Good | 14.59 | $528.28 | 8.09 | $221.31 | 1.80 | 2.39 |
| Fair | 25.11 | $875.10 | 16.80 | $584.61 | 1.49 | 1.50 |
| Poor | 37.86 | $1,309.97 | 25.80 | $793.25 | 1.47 | 1.65 |
| Source: Center for Cost and Financing Studies, Agency for Healthcare Research and Quality: Medical Expenditure Panel Survey Household Component, 1996. | ||||||
Age
Table 2-12 and Figure 2-9 show data on spending for prescription drugs by age. There is a notable difference between elderly and non-elderly Medicare beneficiaries. Non-elderly Medicare beneficiaries qualify for the program on the basis of disability or end stage renal disease, and thus may be more likely to need prescription drugs. The spending gap between covered and noncovered beneficiaries is largest for nonelderly beneficiaries, particularly those under 45. Nonelderly beneficiaries with drug coverage spend much more than elderly beneficiaries with drug coverage; beneficiaries under 45 without coverage spend much less than elderly noncovered beneficiaries.
| Age | Covered | Not covered | Ratio, covered/not covered | |||
|---|---|---|---|---|---|---|
| Average total spending | Average out-of-pocket Spending | Out-of-pocket as % of total spending | Average total spending (all out-of-pocket) | Average total spending | Average Out-of-pocket Spending | |
| Total | $769 | $253 | 33% | $463 | 1.66 | 0.55 |
| 0-44 | $1,077 | $241 | 22% | $268 | 4.01 | 0.90 |
| 45-64 | $1,300 | $347 | 27% | $588 | 2.21 | 0.59 |
| 65-69 | $662 | $218 | 33% | $395 | 1.68 | 0.55 |
| 70-74 | $692 | $229 | 33% | $483 | 1.43 | 0.47 |
| 75-79 | $762 | $276 | 36% | $461 | 1.65 | 0.60 |
| 80-84 | $743 | $264 | 36% | $519 | 1.43 | 0.51 |
| 85+ | $708 | $280 | 40% | $487 | 1.45 | 0.58 |
| Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996. | ||||||
Figure 2-9. Out-of-pocket and Insurer Spending for Medicare Beneficiaries with and without Drug Coverage, by Age, 1996
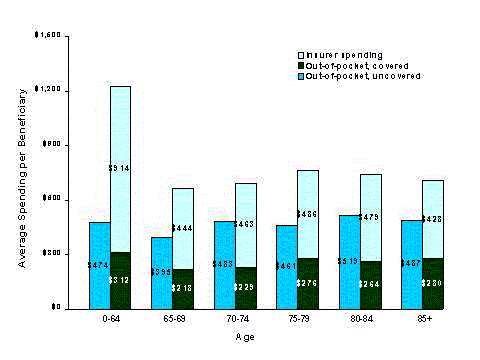
Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996.
Among elderly beneficiaries, spending by those with coverage rises with age, except that it drops slightly for those aged 80 and older. 13 This may be because the sickest among the oldest beneficiaries are likely to be in institutions and are therefore omitted from the data. A similar, but slightly less clear, pattern is present for the elderly without coverage. The gap between those with and without coverage persists, but does not seem to follow any pattern as beneficiaries age.
Race
Table 2-13 shows spending by covered and noncovered beneficiaries by race. Among Medicare beneficiaries, average spending by people with coverage is slightly higher for whites than for blacks or people of other races, but the difference in spending for the covered and the noncovered is slightly greater for nonwhites. However, these differences are not statistically significant. Multivariate analysis would be needed to determine the extent to which any spending differences by race are associated with other factors, such as income, health status, or source of coverage.
| Race/ethnicity | Covered | Not covered | Ratio, covered/not covered | |||
|---|---|---|---|---|---|---|
| Average total spending | Average out-of-pocket Spending | Out-of-pocket as % of total spending | Average total spending (all out-of-pocket) | Average total spending | Average Out-of-pocket Spending | |
| Total | $769 | $253 | 33% | $463 | 1.66 | 0.55 |
| White | $781 | $263 | 34% | $478 | 1.63 | 0.55 |
| Black | $699 | $199 | 28% | $369 | 1.90 | 0.54 |
| Other | $699 | $192 | 27% | $330 | 2.12 | 0.58 |
|
Note: “Other” includes Asian, Hispanic, and North American Native. Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996. |
||||||
Distribution of Spending
Table 2-14 and Figure 2-10 show the distribution of spending by beneficiaries with and without drug coverage in 1996. Only 4 percent of Medicare beneficiaries accounted for almost a quarter of all beneficiaries’ drug spending in 1996, and the top third of beneficiaries spent three-fourths of the total spent on drugs. The distribution of spending is different for covered beneficiaries compared to those without coverage. Compared to beneficiaries with coverage, those without coverage were almost twice as likely to spend nothing, and about half as likely to spend over $1,000.
| Spending on Prescription Drugs | Percent of population | Percent of Spending | ||||
|---|---|---|---|---|---|---|
| Total | Covered | Not covered | Total | Covered | Not covered | |
| Total | 100% | 100% | 100% | 100% | 100% | 100% |
| No Spending on Drugs | 13% | 11% | 20% | 0% | 0% | 0% |
| $1 – 249 | 27% | 26% | 29% | 4% | 4% | 6% |
| $250 – 499 | 17% | 16% | 19% | 9% | 8% | 15% |
| $500 – 749 | 12% | 12% | 11% | 11% | 10% | 15% |
| $750 – 999 | 9% | 9% | 8% | 11% | 10% | 15% |
| $1,000 - 1,249 | 6% | 6% | 4% | 9% | 9% | 9% |
| $1,250 - 1,499 | 4% | 5% | 3% | 8% | 8% | 8% |
| $1,500 - 1,749 | 3% | 3% | 2% | 6% | 7% | 5% |
| $1,750 - 1,999 | 2% | 3% | 2% | 7% | 7% | 8% |
| $2000 - 2,499 | 3% | 3% | 2% | 9% | 10% | 7% |
| $2,500+ | 4% | 5% | 1% | 24% | 28% | 10% |
| Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996. | ||||||
Figure 2-10. Distribution of Medicare Beneficiaries and Prescription Drug Expenditures by Amount of Spending, 1996
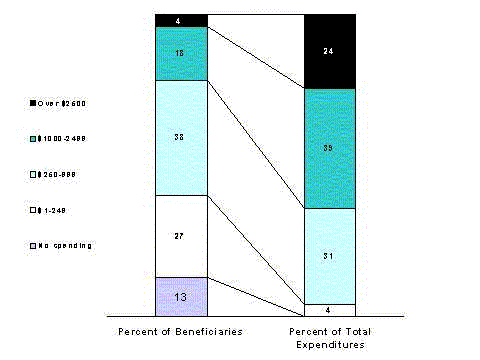
Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996.
Tables 2-15 through 2-17 provide a fuller picture of the characteristics of the subset of Medicare beneficiaries with the highest spending. They compare the 20 percent of the population with the highest spending (those with annual expenditures over $1,066) with the total population. The top 20 percent group is referred to as the “highest drug spenders” in these tables. Overall, 24 percent of covered beneficiaries fell into this group, and 12 percent of noncovered beneficiaries. Of the high spenders, 82 percent had drug coverage, compared to 69 percent of all beneficiaries.
The data show that, in general, the differences explored in this report between Medicare beneficiaries with and without drug coverage do not appear to be driven by high spenders. In other words, the gaps in spending and utilization between Medicare beneficiaries with and without drug coverage do not appear to result because of unique characteristics of those with higher spending. Differences in source of supplemental coverage for high spenders with and without drug coverage mirror the differences seen between all beneficiaries with and without drug coverage, as shown in Table 2-15. Overall, the highest spenders are more likely to have employer based insurance or Medicaid, and less likely to have individually purchased insurance or be in a risk HMO.
| Primary Medicare supplement | Covered | Not covered | Total | |||
|---|---|---|---|---|---|---|
| Percent of all beneficiaries | Percent of highest drug spenders | Percent of all beneficiaries | Percent of highest drug spenders | Percent of all beneficiaries | Percent of highest drug spenders | |
| TOTAL | 100% | 100% | 100% | 100% | 100% | 100% |
| Risk | 15% | 10% | 2% | * | 11% | 8% |
| Medicaid | 17% | 19% | 4% | 5% | 13% | 17% |
| Employer Sponsored | 47% | 50% | 13% | 18% | 36% | 44% |
| Individually Purchased | 16% | 16% | 54% | 61% | 28% | 24% |
| Other | 5% | 5% | 2% | * | 4% | 5% |
| FFS Medicare | 0% | 0% | 25% | 14% | 8% | 3% |
|
*Less than 1 percent; sample is too small to produce a stable estimate. Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996. |
||||||
For the demographic characteristics shown in Table 2-16, the highest spenders do not differ very much from the general Medicare population. Across the total population with and without drug coverage, high spenders’ income distribution is similar; so is their age distribution, except that high spenders are slightly more likely to be nonelderly. Their distribution by race, sex, and metropolitan residence is also comparable. Within both the covered and the non-covered categories, the highest spenders are also similar to the general population of Medicare beneficiaries, except that people without coverage who are below poverty are less likely to be high spenders. This suggests that beneficiaries in all income and socioeconomic groups are at risk of having high drug spending, but that both health status and drug coverage can influence that spending.
In contrast, as might be expected, the highest spenders tend to have poorer health than other Medicare beneficiaries, as measured by each of the indicators used. Table 2-17 shows that these beneficiaries are more likely to report poorer health status, multiple chronic conditions, and more functional limitations, and they are more likely to have had an inpatient admission. This is true for beneficiaries with and without coverage.
| Covered | Not covered | Total | ||||
|---|---|---|---|---|---|---|
| Percent of all beneficiaries | Percent of highest drug spenders | Percent of all beneficiaries | Percent of highest drug spenders | Percent of all beneficiaries | Percent of highest drug spenders | |
| Total | 100% | 100% | 100% | 100% | 100% | 100% |
| Poverty/Income | ||||||
| <Poverty | 21% | 22% | 23% | 18% | 22% | 22% |
| Poverty-135% | 11% | 11% | 15% | 17% | 12% | 12% |
| 136-150% | 4% | 3% | 6% | 7% | 5% | 4% |
| 151-175% | 7% | 7% | 8% | 7% | 7% | 7% |
| 176-200% | 7% | 8% | 9% | 9% | 8% | 8% |
| 201-300% | 20% | 21% | 19% | 20% | 20% | 21% |
| 301-400% | 12% | 12% | 9% | 9% | 11% | 12% |
| >400% | 17% | 16% | 12% | 13% | 16% | 16% |
| Age | ||||||
| 0-44 | 4% | 5% | 4% | 2% | 4% | 5% |
| 45-64 | 8% | 13% | 7% | 11% | 8% | 13% |
| 65-69 | 27% | 23% | 23% | 18% | 25% | 22% |
| 70-74 | 24% | 21% | 23% | 23% | 23% | 21% |
| 75-79 | 18% | 18% | 19% | 17% | 18% | 18% |
| 80-84 | 12% | 12% | 14% | 16% | 12% | 13% |
| 85+ | 8% | 8% | 11% | 12% | 9% | 8% |
| Sex | ||||||
| Male | 45% | 40% | 42% | 39% | 44% | 40% |
| Female | 55% | 60% | 58% | 61% | 56% | 60% |
| Race | ||||||
| White | 85% | 88% | 88% | 92% | 86% | 88% |
| Black | 9% | 8% | 9% | 6% | 9% | 8% |
| Other | 6% | 4% | 4% | 2% | 5% | 4% |
| Metro status | ||||||
| Metro | 78% | 77% | 64% | 62% | 73% | 74% |
| Nonmetro | 22% | 23% | 36% | 38% | 26% | 25% |
| Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996. | ||||||
| Covered | Not covered | Total | ||||
|---|---|---|---|---|---|---|
| Percent of all beneficiaries | Percent of highest drug spenders | Percent of all beneficiaries | Percent of highest drug spenders | Percent of all beneficiaries | Percent of highest drug spenders | |
| Health Status | ||||||
| Excellent | 16% | 7% | 17% | 3% | 17% | 6% |
| Very Good | 26% | 17% | 27% | 17% | 27% | 17% |
| Good | 30% | 30% | 30% | 32% | 30% | 30% |
| Fair | 18% | 28% | 17% | 31% | 17% | 28% |
| Poor | 10% | 18% | 9% | 17% | 9% | 18% |
| Functional Status | ||||||
| No Limitations | 76% | 65% | 77% | 63% | 77% | 64% |
| IADL Only | 4% | 8% | 4% | 8% | 4% | 8% |
| 1 or 2 ADLs | 12% | 16% | 12% | 17% | 12% | 17% |
| 3+ ADLs | 7% | 11% | 7% | 12% | 7% | 11% |
| Chronic Conditions | ||||||
| 0 | 9% | 2% | 11% | 2% | 10% | 2% |
| 1-2 | 44% | 30% | 46% | 26% | 45% | 30% |
| 3-4 | 36% | 45% | 34% | 50% | 35% | 46% |
| 5+ | 11% | 22% | 8% | 21% | 10% | 22% |
| Inpatient Stay | ||||||
| No | 82% | 74% | 82% | 67% | 82% | 73% |
| Yes | 18% | 26% | 18% | 33% | 18% | 27% |
| Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996. | ||||||
The distribution of drug spending is even more concentrated for the non-Medicare population. Table 2-18 shows the distribution for the non-Medicare population in three coverage groups: coverage other than Medicaid, Medicaid, and no coverage. The highest spenders account for much of the spending in each category. Among individuals with Medicaid or with other drug coverage, the top 5 percent account for over half of total spending. The top 5 percent of individuals without drug coverage account for almost three-quarters of total spending for people without drug coverage. People with Medicaid are somewhat more likely than people with other coverage to have had no drug spending during the year, while people without coverage are much more likely to have had no drug spending.
| Amount of prescription drug spending | Percent of population | Percent of total spending | ||||
|---|---|---|---|---|---|---|
| Covered | Medicaid | Not covered | Covered | Medicaid | Not covered | |
| Total | 100% | 100% | 100% | 100% | 100% | 100% |
| $0 | 29% | 36% | 66% | 0% | 0% | 0% |
| $1-250 | 51% | 50% | 29% | 16% | 16% | 27% |
| $250-500 | 9% | 4% | 2% | 14% | 8% | 13% |
| $500-1,000 | 6% | 4% | 2% | 19% | 17% | 18% |
| $1,000 -2,000 | 3% | 3% | 1% | 20% | 24% | 19% |
| $2,000+ | 2% | 2% | <1% | 32% | 34% | 24% |
|
Note: The total includes persons with negative family income. Percents may not add to 100 because of rounding. Source: Center for Cost and Financing Studies, Agency for Healthcare Research and Quality: Medical Expenditure Panel Survey Household Component, 1996. |
||||||
Explaining Utilization and Spending Differences
There are a number of reasons why people with prescription drug coverage might have higher utilization and spending than people without coverage:
- People who anticipate large drug expenditures might be more likely to seek drug coverage than people who do not expect to spend much on drugs. This phenomenon is known as adverse selection or self-selection.
- More prescriptions, or prescriptions for more costly drugs, might be written for people with coverage. This could be because physicians consider a patient’s coverage when deciding on appropriate treatment. People with coverage may also be more likely to request drug therapies, especially more costly therapies. Higher demand by the consumer in these situations is also known as moral hazard.
- People without coverage may be less likely to fill the prescriptions they receive, or they may make a prescription last longer by not taking the recommended dosage. Those who have no health insurance at all (a large share of the nonelderly without drug coverage) may never visit a physician to obtain a prescription in the first place. These problems will be referred to here as lack of access.
All of these factors probably play a part in the observed utilization and expenditure differences. They are notoriously difficult to disentangle, both for drugs and for medical care in general. Distinguishing between self-selection, moral hazard, and lack of access requires information about the relative quantity of drugs actually needed by people with and without coverage. This would require a much higher level of clinical detail than is available in the data used for this report. Some factors will be presented here to highlight fruitful areas for further analysis, not necessarily to resolve the issues. The emphasis will be on the Medicare population.
Adverse Selection
People without Medicare may decide whether or not to obtain health insurance, and anticipated need is certainly a factor in this choice. However, most non-Medicare individuals do not make a separate decision about whether to obtain coverage specifically for prescription drugs. The vast majority who have insurance are covered through employer plans or Medicaid programs and automatically receive whatever level of drug coverage their insurance provides. The primary exceptions are people whose employers offer cafeteria plans where drugs are an option, and people who have a choice of benefit packages when they seek out individual policies.
The story is similar for those Medicare beneficiaries who have employer-based coverage or Medicaid: they often do not control whether or not drugs are a part of their benefit package. However, a larger proportion of Medicare beneficiaries do not have access to these two sources of coverage. These beneficiaries must decide whether to obtain coverage to supplement their Medicare benefits, and whether that supplemental coverage should include drugs. It would be reasonable to expect that this decision would be influenced by the amount of health and drug spending that the beneficiary expects to incur. However, as Chapter 1 discussed, Medigap rules may make coverage more difficult to obtain for those in poor health. It is also likely that many Medicare beneficiaries who choose to buy supplemental coverage that includes drug coverage do so because they want to insure against the possibility of having high costs, not because they already know that their costs will be high.
Research to date has not focused specifically on the question of adverse selection into prescription coverage by Medicare beneficiaries. There is, however, a substantial body of research on selection into Medicare supplemental policies independent of drug coverage. Recent studies of Medicare HMO enrollment find strong evidence of favorable rather than adverse selection – that is, that enrollees in Medicare HMOs are actually healthier than average.14 Indications of adverse selection in the Medigap market have been found by some researchers but not others.15 Most researchers assume that there is no selection into retiree plans given the nature of plan sponsorship, but one recent work finds evidence of adverse selection in this market.16 These thoroughly mixed findings do little to clarify the issue of possible adverse selection into prescription drug coverage.
As Chapter 1 showed (see Tables 1-8 through 1-11), Medicare beneficiaries are slightly more likely to have coverage if they report poorer health status or more chronic conditions, but many of the differences are not statistically significant. Further analysis is needed to explore whether there is evidence of adverse selection when controlling for other factors.
Given the small differences in health status between beneficiaries with and without coverage, the spending differences between the covered and noncovered cannot be explained by assuming that sick people obtain coverage and healthy people forgo it. Furthermore, data presented earlier in this chapter in Tables 2-7 through 2-11 indicate poor health does not appear to explain the significant differences in utilization and spending among the covered and uncovered.17 This suggests that other factors, such as higher demand by those with coverage and lack of access by those without coverage, are also at work.
Moral Hazard and Barriers to Access
The RAND Health Insurance Experiment of the late 1970’s and early 1980’s found that even in the absence of adverse selection, insurance can lead to higher rates of use.18 In the experiment, higher cost-sharing deterred people from obtaining both necessary and unnecessary care.19 Several studies focusing on drug use by Medicare beneficiaries reach the same basic conclusion — drug coverage increases the probability of drug use. The empirical estimates of insurance effects produced by the studies vary within a relatively narrow range. The addition of drug coverage is estimated to increase the probability of any prescription being filled by between 4 and 16 percent, depending on population subgroup and generosity of drug coverage.20 In a study of Pennsylvania elderly, prescription coverage increased drug use by approximately 3 percent for every 10 percent reduction in out-of-pocket cost to beneficiaries, all else being equal.21
It is difficult to separate whether these effects are due to higher demand by people with coverage, or lack of access for people without coverage. If two people have similar characteristics, and the one with coverage receives more drugs than the one without coverage, is this because the one with coverage is receiving the appropriate drugs and the one without coverage is not? Or is it because the person with coverage is encouraged to receive excessive or unnecessary treatments and the one without coverage is thriftily purchasing only the care he or she needs? Both answers could be correct. Many people without coverage report difficulty in obtaining needed medications; data on the extent of this problem will be presented in the next section of this chapter. At the same time, however, studies have shown that some elderly people receive inappropriate drugs or inappropriately large dosages, with potential adverse effects.22
Consequences of Lack of Drug Coverage
There are several ways to explore the burden that drug spending places on Medicare beneficiaries without coverage as compared to those who do have coverage: drug spending as a proportion of income, drug spending as a proportion of other spending, self-reported problems in affording drugs, and self-reported differences in the mix of drugs used. These measures show that beneficiaries without drug coverage are likely to have more trouble meeting their prescription drug costs, and are more likely to forgo filling a prescription as a result.
Out-Of-pocket Drug Spending and Income
This report has shown that, while people without drug coverage have lower total drug spending than those with coverage, they incur higher out-of-pocket costs. Tables 2-20 through 2-23 show this out-of-pocket spending as a share of income for covered and noncovered Medicare beneficiaries, in the total population and in the 20% with the highest spending. Among all beneficiaries, those without coverage spend over twice as much of their income for drugs as those with coverage (2.2 percent versus 1 percent of income). Among the highest spenders, the ratio is over three to one (8.1 percent versus 2.6 percent of income). Within the group of high spenders, some classes of beneficiaries without coverage are spending a significant proportion of income (frequently over 10 percent, and sometimes over 20 percent) on out-of-pocket payments for drugs.
Table 2-19 and Figure 2-11 show that at all income levels, beneficiaries with no drug coverage spend a larger proportion of their income on out-of-pocket prescription drug costs. Beneficiaries in the high spending group whose incomes are below the poverty threshold spend more than a quarter of their income on prescription drugs.
| Income | All beneficiaries | Highest drug spenders | ||
|---|---|---|---|---|
| Covered | Not covered | Covered | Not covered | |
| TOTAL | 1.0% | 2.2% | 2.6% | 8.1% |
| <Poverty | 3.3% | 5.8% | 7.8% | 27.8% |
| Poverty-135% | 2.6% | 4.6% | 6.2% | 16.5% |
| 136-150% | 2.1% | 4.4% | 6.3% | 15.9% |
| 151-175% | 2.0% | 3.1% | 4.7% | 12.4% |
| 176-200% | 1.5% | 3.1% | 3.7% | 11.8% |
| 201-300% | 1.3% | 2.2% | 3.2% | 7.7% |
| 301-400% | 0.8% | 1.5% | 2.1% | 5.8% |
| >400% | 0.4% | 0.7% | 1.0% | 2.8% |
| Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996. | ||||
Figure 2-11. Out-of-pocket Spending on Prescription Drugs as a Proportion of Income for Medicare Beneficiaries With and Without Drug Coverage, by Income Level, 1996
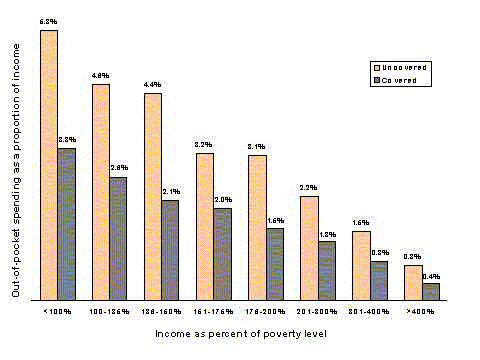
Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996.
Table 2-20 shows the relative income share by type of coverage. Shares of income devoted to out-of-pocket spending are quite close for beneficiaries with individually purchased coverage that does and does not include a drug benefit. This result is consistent with earlier results showing that Medigap coverage tends to offer the least protection against drug costs. High spenders with no supplemental insurance spend almost 12 percent of their income on prescription drugs; those who are enrolled in Medicaid but have no drug coverage spend 23 percent of their income on drugs.
| Source of supplemental coverage | All beneficiaries | Highest drug spenders | ||
|---|---|---|---|---|
| With drug coverage | Without drug coverage | With drug coverage | Without drug coverage | |
| Total | 1.0% | 2.2% | 2.6% | 8.1% |
| Risk HMO | 0.8% | * | 2.1% | * |
| Medicaid | 2.2% | 4.3% | 5.5% | 23.0% |
| Employer Sponsored | 0.7% | 1.7% | 1.8% | 6.2% |
| Individually Purchased | 1.7% | 2.2% | 4.7% | 7.8% |
| Other | 1.7% | 3.2% | 3.8% | * |
| FFS Medicare; no supplement | 2.4% | 11.6% | ||
|
*Sample size too small for a reliable estimate. Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996. |
||||
Table 2-21 shows out-of-pocket spending on prescription drugs as a proportion of income for different demographic groups. For Medicare beneficiaries as a whole, the relative income share for the covered and noncovered is fairly constant across these subgroups. Those without coverage almost always spend a higher percentage of their income on drugs. Among the highest spenders who have no drug coverage, certain demographic groups have extraordinarily high costs as a proportion of their income. The youngest and oldest beneficiaries in this group both spend more than 10 percent of their income on drugs. Non-white beneficiaries without coverage in the high spending group spend over 15 percent of their income on prescription drugs.
| All beneficiaries | Highest drug spenders | |||
|---|---|---|---|---|
| Covered | Not covered | Covered | Not covered | |
| TOTAL | 1.0% | 2.2% | 2.6% | 8.1% |
| Age | ||||
| 0-44 | 2.0% | 2.0% | 4.2% | 13.6% |
| 45-64 | 2.0% | 3.2% | 3.7% | 8.4% |
| 65-69 | 0.7% | 1.7% | 2.2% | 9.8% |
| 70-74 | 0.8% | 2.0% | 2.1% | 6.6% |
| 75-79 | 1.1% | 2.2% | 2.5% | 7.3% |
| 80-84 | 1.3% | 2.6% | 2.7% | 8.2% |
| 85+ | 1.3% | 2.8% | 3.5% | 10.3% |
| Sex | ||||
| Male | 0.8% | 1.8% | 2.1% | 6.8% |
| Female | 1.3% | 2.5% | 3.0% | 9.3% |
| Race | ||||
| White | 1.0% | 2.1% | 2.5% | 7.7% |
| Black | 1.4% | 3.1% | 4.1% | 15.9% |
| Other | 1.3% | 2.3% | 3.8% | 15.4% |
| Metro status | ||||
| Metro | 0.9% | 2.0% | 2.3% | 7.7% |
| NonMetro | 1.4% | 2.5% | 3.8% | 8.8% |
| Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996. | ||||
Table 2-22 shows spending on drugs as a percent of income by various measures of health status. Uncovered high spenders who have the worst health or functional status or the most chronic conditions spend over a tenth of their income on prescription drugs.
| All beneficiaries | Top 20% by spending | |||
|---|---|---|---|---|
| Covered | Not covered | Covered | Not covered | |
| Health status | ||||
| Excellent | 0.4% | 0.9% | 1.0% | 7.4% |
| Very Good | 0.7% | 1.5% | 1.9% | 5.3% |
| Good | 1.1% | 2.6% | 2.5% | 7.4% |
| Fair | 1.9% | 3.7% | 3.2% | 9.8% |
| Poor | 2.6% | 3.9% | 4.3% | 12.5% |
| Functional status | ||||
| No Limitations | 0.8% | 1.9% | 2.1% | 7.7% |
| IADL Only | 2.2% | 3.7% | 3.8% | * |
| 1 or 2 ADLs | 1.9% | 3.2% | 3.7% | 8.4% |
| 3+ ADLs | 2.3% | 3.6% | 4.3% | 12.2% |
| Chronic conditions | ||||
| 0 | 0.3% | 0.7% | 3.0% | 8.1% |
| 1-2 | 0.7% | 1.6% | 2.0% | 6.9% |
| 3-4 | 1.4% | 2.8% | 2.8% | 7.8% |
| 5+ | 2.0% | 4.7% | 3.1% | 11.0% |
| Inpatient stay | ||||
| No | 0.9% | 1.9% | 3.0% | 8.1% |
| Yes | 1.7% | 3.4% | 2.3% | 6.8% |
|
*Cell size is too small to produce a reliable estimate Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1996. |
||||
Even some people with coverage are devoting a considerable share of their income to prescription drugs. MCBS estimates that 8 percent of beneficiaries with drug coverage have total spending of $2,000 per year or more. Table 2-23 shows the out-of-pocket costs that would be incurred by someone with $2,000 in spending under different types of drug coverage. Beneficiaries with Medigap plans would pay over half this cost out of pocket.
| Source of drug coverage | Deductible | Copay | Over cap | Total out-of-pocket | Percent paid out-of-pocket |
|---|---|---|---|---|---|
| HMO ($7.50 copay, $1,500 cap)1 | -- | $412 | $88 | $500 | 25% |
| Medigap H/I/J ($250 deductible, $1,250 or $3,000 cap, 50% coinsurance) 2 | $250 | $875 | -- | $1,125 | 56% |
| Employer ($7.50 copay)1 | -- | $412 | -- | $412 | 21% |
| Medicaid ($2 copay)1 | -- | $110 | -- | $110 | 6% |
| Medicare FFS only | -- | -- | -- | $2,000 | 100% |
|
1 For non-Medigap plans, typical cost-sharing rules are assumed; there are plans with higher and lower cost sharing. The $2,000 spending was divided by the average cost of prescriptions for Medicare beneficiaries with drug coverage ($36.37) to generate the number of prescriptions (55) used for the examples in this table. The cap on HMO payments applies to spending by the plan. 2 For Medigap plans, out-of-pocket spending in this table is calculated directly from the dollar amount of spending ($2,000). After the beneficiary has met the $250 deductible, Plans H and I will cover 50% of $2500 in total spending, for a total plan payment of $1250. Plan J will pay a total of $3000. NOTE: This table does not attempt to account for premiums paid or the different purchasing power that $2000 might have under different discount arrangements negotiated by HMOs, employers, and Medicaid. |
|||||
Table 2-24 shows data from MEPS on out-of-pocket spending as a proportion of income for non-Medicare beneficiaries. Overall, this group spends a lower share of their income on drugs than Medicare beneficiaries spend. Among all non-Medicare drug users, those with coverage spend about half as much of their income on prescription drugs as people without coverage. The difference between covered and noncovered users persists across all income levels.23
| Income quintile | Total Population | Users | ||
|---|---|---|---|---|
| Covered | Noncovered | Covered | Noncovered | |
| TOTAL | 0.98% | 0.93% | 1.41% | 2.72% |
| 20% | * | * | * | * |
| 40% | 0.41% | 0.28% | 0.59% | 0.84% |
| 60% | 0.23% | 0.15% | 0.33% | 0.46% |
| 80% | 0.14% | 0.08% | 0.19% | 0.29% |
| 100% | 0.08% | 0.06% | 0.11% | 0.14% |
|
* Estimates are not stable. Source: Center for Cost and Financing Studies, Agency for Healthcare Research and Quality: Medical Expenditure Survey Household Component, 1996. |
||||
Out-of-pocket Drug Spending and Other Health Spending
Spending for drugs is a substantial component of total out-of-pocket health spending for all ages and income levels. The following tables and charts were prepared using the Consumer Expenditure Survey (CEX) data prepared by the Bureau of Labor Statistics. This analysis differs in several respects from others reported in this chapter. Expenditures are for the consumer unit (basically a family or unrelated individuals who share living expenses) rather than the individual. Consumer units are grouped here according to age of the reference person.24 A unit with an elderly reference person may contain non-elderly individuals, and vice versa.
Table 2-25 shows the average breakdown of spending by consumer units in 1997-98. Spending for non-health necessities -- defined here as food, shelter, utilities, transportation, and clothing -- makes up a majority of total spending for all ages and income groups.25 Out-of-pocket health spending, including insurance premiums as well as out-of-pocket payments for drugs and other medical services and supplies, is an important component of discretionary spending (spending other than for non-health necessities) for all ages and incomes. Payments for over-the-counter drugs are included in the total for out-of-pocket health spending. As in the rest of this chapter, they are not included in spending on prescription drugs.
| Average expenditures per consumer unit | Consumer units whose reference person is: | |||||
|---|---|---|---|---|---|---|
| Under age 65 | Age 65 or over | |||||
| All incomes | Income <$15,0002 | Income >$40,0002* | All incomes | Income <$15,0002 | Income >$40,0002 * | |
| Total expenditures | $37,306 | $16,746 | $53,740 | $25,598 | $15,999 | $51,377 |
| Spending for non-health necessities | $23,602 | $12,184 | $32,247 | $15,032 | $9,986 | $27,573 |
| Discretionary spending (including health)3 | $13,704 | $4,562 | $21,493 | $10,566 | $6,013 | $23,804 |
| Health spending4 | $1,499 | $703 | $2,045 | $2,993 | $2,178 | $4,210 |
| Prescription drug spending | $154 | $117 | $173 | $516 | $438 | $502 |
| Drugs as % of all spending | 0.4% | 0.7% | 0.3% | 2.0% | 2.7% | 1.0% |
| Drugs as % of discretionary | 1.1% | 2.6% | 0.8% | 3.8% | 5.3% | 1.8% |
| Drugs as % of health | 10.2% | 16.6% | 8.5% | 17.2% | 20.1% | 11.9% |
|
* The statistics in this income group are less reliable than for other series, due to a lower sample size. 1Estimates of single-year prescription drug expenditures are unpublished data that do not meet the reliability standards of published estimates. Therefore, data are presented as averages of two years to generate sufficient sample size needed to make the prescription drug estimates meet the Bureau of Labor Statistics’ reliability standards. 2All income ranges are before taxes. 3Health spending is included with discretionary spending because spending for health is frequently forgone by low income households. 4Out-of-pocket health care spending includes out of pocket insurance payments, prescription and nonprescription drug expenses, and payments for all other medical services and supplies. Source: Consumer Expenditure Survey data from the Bureau of Labor Statistics as analyzed by the Office of the Actuary, HCFA(now known as CMS). |
||||||
As Figure 2-12 shows, out-of-pocket spending for prescription drugs is a larger proportion of health spending for units with an elderly reference person than for units with a younger reference person.26 Prescription drug spending also accounts for a larger share for people with low incomes than for people with higher incomes. For the lower income units with an elderly reference person, spending for drugs accounted for 20 percent of health spending, and 5 percent of all discretionary spending.
Figure 2-12. Out-of-pocket Prescription Drug Spending as a Proportion of Health Spending, by Age and Income, 1997-1998

Note: Expenditures are for the consumer unit and shown by the age of the reference person (see text).
Source: Office of the Actuary, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Bureau of Labor Statistics Consumer Expenditure Survey, 1997-1998
Because the CEX information on drug coverage is not definitive, this analysis is not able to compare spending by those with coverage to those without coverage. However, this chapter showed earlier for Medicare beneficiaries that as a percentage of income, drug spending for those without coverage was double the spending of those with coverage (see Table 2-19). This implies that for uncovered beneficiaries, an even larger proportion of spending goes to prescription drugs than is shown here.
Table 2-26 shows changes in the components of spending for units with an elderly reference person between 1992-93 and 1997-98. Out-of-pocket spending for drugs rose much more rapidly than out-of-pocket health spending. This was particularly true for low income families. In dollar terms, out-of-pocket drug spending for higher-income units increased more than for lower-income units. As a share of health spending, however, the increase was much greater for the lowest-income units: increased spending for drugs accounted for 92 percent of the total increase in health spending during the period. As a result, drugs as a share of health spending rose from 16.3 percent to 17.2 percent for all consumer units with an elderly reference person, and from 17.6 percent to 20.1 percent for the subset of these units with the lowest incomes.
| Average expenditures Per consumer unit | All income groups | Income <$15,000 before taxes | ||||
|---|---|---|---|---|---|---|
| 1992-93 | 1997-98 | % change | 1992-93 | 1997-98 | % change | |
| Total expenditures | $21,214 | $25,598 | 20.7% | $14,054 | $15,999 | 13.8% |
| Spending for non-health necessities | $12,520 | $15,032 | 20.1% | $8,760 | $9,986 | 14.0% |
| Discretionary spending (including health)2 | $8,694 | $10,566 | 21.5% | $5,294 | $6,013 | 13.6% |
| Health spending3 | $2,644 | $2,993 | 13.2% | $2,106 | $2,178 | 3.4% |
| Prescription drug spending | $432 | $516 | 19.4% | $372 | $438 | 17.9% |
| Drugs as % of all spending | 2.0% | 2.0% | 2.6% | 2.7% | ||
| Drugs as % of discretionary | 5.0% | 4.9% | 7.0% | 7.3% | ||
| Drugs as % of health | 16.3% | 17.2% | 17.6% | 20.1% | ||
|
1Estimates of single-year prescription drug expenditures are unpublished data that do not meet the reliability standards of published estimates. Therefore, data are presented as averages of two years to generate sufficient sample size needed to make the prescription drug estimates meet the Bureau of Labor Statistics’ reliability standards. 2Health spending is included with discretionary spending because spending for health is frequently forgone by low income households. 3Out-of-pocket health care spending includes out of pocket insurance payments, prescription and nonprescription drug expenses, and payments for all other medical services and supplies. Source: Consumer Expenditure Survey data from the Bureau of Labor Statistics as analyzed by the Office of the Actuary, HCFA(now known as CMS). |
||||||
Access to Needed Drugs
Because out-of-pocket spending for prescription drugs can take up a significant share of spending, some people may not be able to buy all of the medications they need. Data from the 1997 National Health Interview Survey confirm that, both among Medicare beneficiaries and in the total population, people without coverage are going without medications because they cannot afford them.27
Table 2-27 shows, for the total (Medicare and non-Medicare) population, NHIS estimates of people who reported that they needed prescription drugs but did not get them because they could not afford to. These data do not necessarily reflect cases in which people postponed filling a prescription, purchased less than the quantity prescribed, or took the medication less often than prescribed. About 3 percent of all people with health insurance, and 16 percent of people without health insurance, failed to get needed prescription medicine because they could not afford it. Note that health insurance in this table includes insurance that does not cover prescription drugs; thus all elderly people with Medicare fall into the insured column. If data were available on drug coverage, the differences would likely be even larger.
| Percent reporting that they needed prescription medicine in the last 12 months but did not get it because they (or a parent) could not afford it | |||
|---|---|---|---|
| All persons | With health insurance | Without health insurance | |
| Total | 4.9% | 3.0% | 16.4% |
| Income (as % of Poverty) | |||
| <Poverty | 11.5% | 7.6% | 21.0% |
| 100-150% | 11.2% | 8.0% | 19.2% |
| 150-200% | 7.0% | 4.8% | 14.9% |
| 200-300% | 5.2% | 3.4% | 17.0% |
| 300-400% | 2.7% | 1.9% | 12.2% |
| >400% | 1.4% | 0.9% | 13.7% |
| Health Status | |||
| Excellent | 2.1% | 1.1% | 8.9% |
| Very Good | 3.7% | 2.1% | 13.7% |
| Good | 6.9% | 4.2% | 19.6% |
| Fair | 13.3% | 9.2% | 35.7% |
| Poor | 21.9% | 16.1% | 60.5% |
| Source: Division of Health Interview Statistics, National Center for Health Statistics, Centers for Disease Control, National Health Interview Survey (NHIS), 1997. | |||
People without insurance are consistently more likely than people with insurance to report that they couldn’t afford needed medicines. Among the uninsured, the problem is greatest for people with low incomes and for people in poor health. Over 60 percent of uninsured people in poor health reported not getting medicines they needed.
Table 2-28 and Figure 2-13 show the responses by more detailed insurance categories and income. Again, for both Medicare beneficiaries and the non-Medicare population, people without insurance and those with the lowest incomes were most likely to go without a prescription drug.28 About a sixth of low-income beneficiaries without supplemental coverage, and just over a fifth of low-income uninsured non-Medicare beneficiaries, reported not receiving a needed drug because of cost.
Figure 2-13. Medicare Beneficiaries Who Did Not Get a Needed Drug Because They Could Not Afford It, by Source of Insurance and Income, 1997
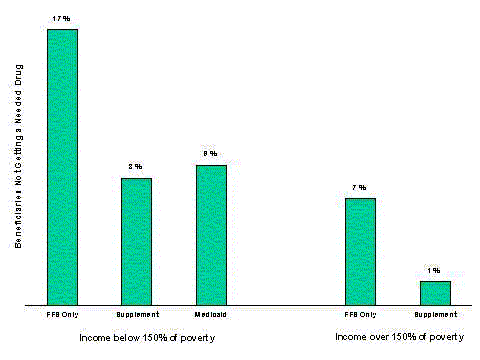
Note: Medicaid is not included at higher income level because sample size is too small to produce reliable estimates.
Source: Division of Health Interview Statistics, National Center for Health Statistics, Centers for Disease Control, National Health Interview Survey (NHIS), 1997.
| Health Insurance | Percent reporting that they needed prescription medicine in the last 12 months but did not get it because they (or a parent) could not afford it | |||
|---|---|---|---|---|
| Total | 0-150% of poverty | 150-300% of poverty | 300% of poverty or more | |
| Medicare beneficiaries | ||||
| Medicare FFS only | 9.9% | 16.9% | 7.6% | * |
| Medicare + non-Medicaid supplement | 2.2% | 7.8% | 2.4% | 0.6% |
| Medicare + Medicaid | 8.8% | 8.6% | * | * |
| Non-Medicare population | ||||
| Medicaid | 6.2% | 6.6% | 7.6% | * |
| Private | 2.4% | 7.1% | 3.8% | 1.2% |
| No Insurance | 16.4% | 20.3% | 16.0% | 13.0% |
|
* Small cell size; estimates not reliable Source: Division of Health Interview Statistics, National Center for Health Statistics, Centers for Disease Control, National Health Interview Survey (NHIS), 1997. |
||||
Medicare beneficiaries with no supplemental coverage were over four times as likely as those with a non-Medicaid supplement to report not receiving a drug they needed. Among the non-Medicare population, people without any insurance were almost eight times as likely as people with private insurance to report going without a needed drug.
Again, this difference between people with and without coverage would likely be even stronger if information were available on which supplements included drug coverage. As Chapter 1 showed, while many Medicare beneficiaries with supplemental coverage have drug coverage, most beneficiaries with an individually purchased policy do not have coverage for their drugs. This distinction may be less important for non-Medicare beneficiaries, who usually have drug coverage if they have health insurance.
Mix of Drugs
Another consequence of not having drug coverage may be the use of a different mix of drugs. In addition to not filling some prescriptions, some people without drug coverage may obtain different prescriptions compared to the prescriptions that people with drug coverage get. Preliminary analysis of MCBS data on Medicare beneficiary utilization of medications commonly associated with the treatment of hypertension (categorized as diuretics, calcium channel blockers, beta blockers and anti-hypertensives)29 suggests that:
- Beneficiaries with prescription drug coverage were more likely to be dispensed a generic drug for all of the drug categories; beneficiaries without coverage were more likely to be dispensed a brand name product. Presumably, beneficiaries with coverage are more likely to be encouraged (or required) to use generic products by the health plan or pharmacy benefit manager.30
- The average number of units dispensed per prescription was higher for people with prescription drug coverage. Individuals with prescription drug coverage may be purchasing a supply of drugs to last a longer period of time (for example, a 30 day supply rather than a 14 day supply). Individuals without coverage also may be skipping doses to avoid higher out-of-pocket costs (for example, taking medication once per day instead of twice per day).
One issue the analysis raises for future work is the possibility of a connection between insurance status and “step therapy.” That is, beneficiaries with insurance could be more likely to have a drug or combination of drugs prescribed and dispensed that is more responsive to a particular patient (and perhaps more expensive). For example, a physician may prescribe an increased drug dose, substitute another drug, or add a second agent from a different class of drugs if a patient is not responding adequately to current drug therapy. More work is necessary to discern whether or not beneficiaries with drug coverage are more likely to be prescribed or fill those additional prescriptions, and take them at appropriate dosages.
Trends in Prescription Drug Spending
Prescription drug spending and utilization have become increasingly important issues in part because of their rapid growth. Spending on prescription drugs has risen faster than spending on other types of health care throughout the 1990’s. This section explores growth in spending for the total population and for Medicare beneficiaries. It will also examine some of the components of this growth.
Growth in Drug Spending for the Total Population
The nation’s spending for prescription drugs has grown dramatically in recent years. Table 2-29 shows estimated total national health expenditures and prescription drug expenditures for selected years from 1965 through 1998, along with HCFA(now known as CMS)’s projections to 2008. All numbers are in nominal dollars and thus reflect general inflation during this period. Figure 2-14 shows the trend in drug spending in 1998 dollars: even when controlling for general inflation, there has been a dramatic increase in drug spending, especially since the mid-1980s.
Figure 2-14. Real Spending for Prescription Drugs for the Total Population, 1965-1998 and Projected 1999-2008, in 1998 Dollars

Source: National Health Statistics Group, Office of the Actuary, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): National Health Accounts . Adjusted for inflation using the historical CPI-U through 1998 and OMB projections of CPI-U for 1999 through 2008.
| Year | National health expenditures | Drug expenditures | Drugs as % of NHE | ||
|---|---|---|---|---|---|
| Total (millions) | Annual change | Total (millions) | Annual change | ||
| 1965 | $41,100 | $3,715 | 9.0% | ||
| 1970 | $73,243 | 12.2% | $5,497 | 8.2% | 7.5% |
| 1980 | $247,273 | 12.9% | $12,049 | 8.2% | 4.9% |
| 1990 | $699,361 | 11.0% | $37,677 | 12.1% | 5.4% |
| 1991 | $766,783 | 9.6% | $42,148 | 11.9% | 5.5% |
| 1992 | $836,537 | 9.1% | $46,598 | 10.6% | 5.6% |
| 1993 | $898,496 | 7.4% | $50,632 | 8.7% | 5.6% |
| 1994 | $947,717 | 5.5% | $55,189 | 9.0% | 5.8% |
| 1995 | $993,725 | 4.9% | $61,060 | 10.6% | 6.1% |
| 1996 | $1,042,522 | 4.9% | $69,111 | 13.2% | 6.6% |
| 1997 | $1,092,385 | 4.8% | $78,888 | 14.1% | 7.2% |
| 1998 | $1,149,100 | 5.1% | $90,648 | 14.9% | 7.9% |
| 2003 (proj.) | $1,590,359 | 6.7% | $152,363 | 11.1% | 9.6% |
| 2008 (proj.) | $2,176,620 | 6.5% | $243,437 | 9.8% | 11.2% |
| Source: National Health Statistics Group, Office of the Actuary, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): National Health Accounts. | |||||
Through the 1970s and 1980s, drug spending grew somewhat more slowly than overall health spending—at an annual rate of 10 percent a year, compared to 12 percent for all health spending. In the 1990s, growth in drug spending began to outpace growth in spending for other kinds of health services. As a result, the share of health expenditures going to prescription drugs increased throughout the 1990’s, growing from 5.4 percent in 1990 to 7.8 percent in 1998 (see Figure 2-15). Part of this trend may be attributed to a slowing of the growth in health expenditures associated with the expansion of managed care. Some analysts contend that growth in drug spending also contributed to the moderation in spending growth for other services. They suggest that some new drug therapies replaced more costly treatments; for example, new asthma drugs may have reduced emergency room utilization. While these substitution effects have been identified in specific instances, it is not possible to assess the aggregate degree of substitution that may be occurring. This could be a fruitful area for future research.
Figure 2-15. Prescription Drug Expenditures as a Percent of National Health Expenditures, 1965-1997
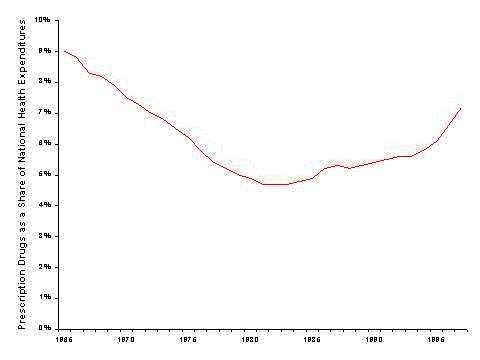
Source: National Health Statistics Group, Office of the Actuary, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): National Health Accounts.
HCFA(now known as CMS) projects that growth in drug spending will moderate slightly in the coming years. This is partly because the patent protection for many top-selling brand-name drugs will expire over the next decade. The likely appearance of generic equivalents for these drugs will depress spending growth slightly. Still, many new medications are expected to appear, and drug spending is projected to consume a steadily larger share of total spending by all the major payment sources.
Between 1965 and 1998, spending on drugs by every major payment source – private insurers, Medicaid, consumers’ out-of-pocket payments, and other sources – increased faster than general inflation. However, expenditures by some sources have increased much faster than others. As Table 2-30 and Figure 2-16 show, the result has been a substantial shift in the share of drug expenditures that each source accounts for.
| Year | Annual Growth | Share of Drug Spending | ||||||
|---|---|---|---|---|---|---|---|---|
| Private Insurance | Medicaid | Out-of-pocket | All Other | Private Insurance | Medicaid | Out-of-pocket | All Other | |
| 1965 | 3.5% | 0.0% | 92.6% | 3.9% | ||||
| 1970 | 30.1% | - | 5.7% | -14.4% | 8.8% | 7.6% | 82.4% | 1.2% |
| 1975 | 15.3% | 15.9% | 6.0% | 13.8% | 12.2% | 10.8% | 75.4% | 1.6% |
| 1980 | 19.8% | 10.1% | 5.5% | 16.1% | 20.1% | 11.7% | 66.0% | 2.2% |
| 1985 | 21.1% | 12.1% | 8.1% | 17.8% | 29.9% | 11.8% | 55.4% | 2.9% |
| 1990 | 15.4% | 15.2% | 9.2% | 19.1% | 34.4% | 13.5% | 48.3% | 3.8% |
| 1991 | 17.0% | 21.7% | 6.1% | 4.0% | 36.0% | 14.7% | 45.8% | 3.6% |
| 1992 | 18.1% | 8.6% | 5.7% | 4.2% | 38.5% | 14.4% | 43.8% | 3.4% |
| 1993 | 12.2% | 15.6% | 3.8% | 2.2% | 39.7% | 15.3% | 41.8% | 3.2% |
| 1994 | 16.6% | 10.4% | 1.1% | 12.8% | 42.5% | 15.5% | 38.8% | 3.3% |
| 1995 | 21.9% | 12.9% | -3.3% | 15.4% | 46.8% | 15.8% | 33.9% | 3.4% |
| 1996 | 17.5% | 14.7% | 5.3% | 16.1% | 48.8% | 16.1% | 31.6% | 3.5% |
| 1997 | 18.7% | 17.2% | 4.9% | 16.2% | 50.8% | 16.5% | 29.1% | 3.6% |
| 1998 | 19.7% | 19.2% | 5.4% | 18.0% | 52.7% | 17.1% | 26.6% | 3.6% |
| 2003 (proj.) | 11.1% | 13.0% | 8.8% | 14.1% | 53.0% | 18.7% | 24.1% | 4.2% |
| 2008 (proj.) | 10.0% | 11.6% | 8.2% | 8.9% | 53.3% | 20.3% | 22.4% | 4.1% |
| Source: National Health Statistics Group, Office of the Actuary, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): National Health Accounts. | ||||||||
Figure 2-16. Spending for Prescription Drugs by Payment Source, Total Population, 1965-1998, in 1998 Dollars
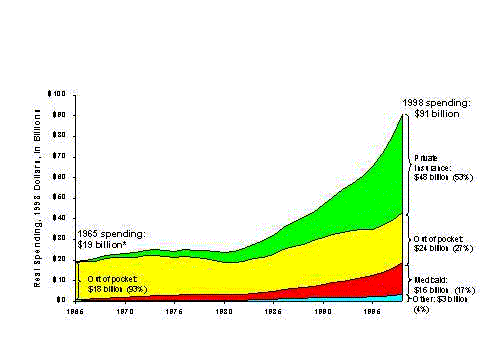
*Nominal spending in 1965 was $3.7 billion.
Source: National Health Statistics Group, Office of the Actuary, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): National Health Accounts. Adjusted for inflation using the CPI-U.
Private insurance payments accounted for only 3.5 percent of all prescription drug expenditures in 1965; by 1998, private insurers paid just over half of all drug costs. One of the major factors in this shift has been the addition of outpatient prescription drugs to the standard benefit package that private insurers offer. In 1965, it was much less common than it is today for health plans to include drugs in their benefit package. More recently, the trend toward an increasing insurer share may also be related to the shift to managed care and the corresponding move from benefit packages with deductibles and coinsurance to no-deductible plans with fixed copayments.31
Medicaid has also covered an increasing share of prescription drug expenditures since its creation in 1965. In 1975, Medicaid paid for just over a tenth of all drug expenditures. The program now covers almost a fifth of all drug costs.
While the population’s out-of-pocket spending on prescription drugs grew significantly between 1965 and 1998 in dollar terms, the share of total drug expenditures that were paid out-of-pocket dropped from 93 percent to only 27 percent. HCFA(now known as CMS) projects that out-of-pocket spending will begin to grow more rapidly than it has in recent years, as insurers and employers respond to higher drug costs by passing some of these costs on to health plan enrollees. However, HCFA(now known as CMS) projects that insurer spending on drugs will grow even more rapidly, continuing the slow shift from out-of-pocket expenditures to other sources of payment.
It is not possible to separate Medicare beneficiaries from the rest of the population in the National Health Expenditures data. However, some of the findings of this report suggest that the sources of spending have been different for Medicare beneficiaries compared to the rest of the population. As Chapter 1 showed, many beneficiaries continue to lack access to a source of coverage for their prescription drug costs. As a result, the trend toward increasing coverage of drug costs by insurers has likely been much less dramatic over time for Medicare beneficiaries than for the rest of the population. The next section will explore overall spending and utilization trends for Medicare beneficiaries more recently, using the Medicare Current Beneficiary Survey.
Spending Trends for Medicare Beneficiaries
Table 2-31 shows MCBS data on changes in drug utilization and spending for Medicare beneficiaries between 1992 (the first year of MCBS) and 1996, results that are generally consistent with the National Health Expenditures estimates for the total population. Total per beneficiary drug spending (including both third-party and out-of-pocket spending) rose at an annual rate of 9 percent in this period. Utilization grew 4.1 percent a year, slightly faster than the 3.9 percent annual growth in total retail prescriptions over the same period (NACDS). Spending per prescription grew 4.7 percent — well above general inflation, which averaged 2.8 percent from 1992 to 1996. Figure 2-17 shows that expenditures per Medicare beneficiary grew more rapidly than prescriptions per beneficiary throughout this period.
As it did for the total population, out-of-pocket spending on prescription drugs by Medicare beneficiaries grew less rapidly than total spending. This is due both to a growth in prescription drug coverage and particularly to shifts into managed care, including expanding Medicare risk HMO enrollment and increased use of managed care in retiree health benefit plans. Thus, this slower growth in out-of-pocket spending by Medicare beneficiaries may not reflect current or future trends.
Figure 2-17. Growth in Drug Spending and Number of Prescriptions per Medicare Beneficiary, 1993-1996
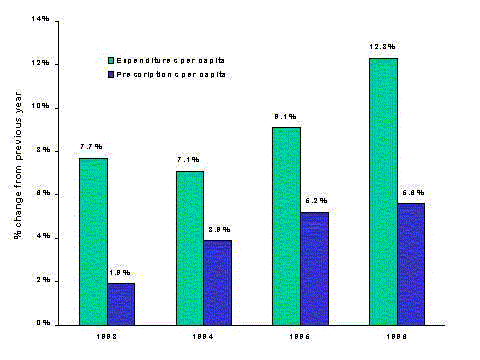
Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1992-1996.
| 1992 | 1993 | 1994 | 1995 | 1996 | Average Annual Change | |
|---|---|---|---|---|---|---|
| Per Capita Total Expenditures | $477 | $513 | $550 | $600 | $674 | 9.0% |
| Per Capita Out-of-pocket | $277 | $284 | $287 | $303 | $318 | 3.5% |
| Per Capita Scripts | 16.62 | 16.94 | 17.60 | 18.52 | 19.54 | 4.1% |
| Total Exp./Script | $28.66 | $30.30 | $31.22 | $32.39 | $34.47 | 4.7% |
| Out-of-pocket/Script | $16.67 | $16.77 | $16.28 | $16.34 | $16.29 | -0.6% |
| Percent Out-of-pocket | 58% | 55% | 52% | 50% | 47% | -5.1% |
| Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1992-1996. | ||||||
Table 2-32 shows changes in spending between 1995 and 1996, by primary source of supplemental coverage and whether or not the beneficiary had drug coverage. Total spending and out-of-pocket spending both grew more rapidly between 1995 and 1996 than in the preceding two years. Spending growth was highest for enrollees in risk HMOs and for Medicaid beneficiaries, and enrollees in other public programs. Spending actually declined for beneficiaries with no supplemental coverage.
| Primary Medicare Supplement | Growth in per capita total drug expenditures | Growth in per capita out-of-pocket drug expenditures | ||||
|---|---|---|---|---|---|---|
| Total | Covered | Not covered | Total | Covered | Not covered | |
| All Beneficiaries | 12.3% | 11.6% | 7.0% | 5.2% | 8.5% | 7.0% |
| No Supplemental Coverage (FFS Medicare only) | -7.2% | na | -7.2% | -7.2% | na | -7.2% |
| Supplemental Coverage: | ||||||
| Medicare Risk HMO | 17.9% | 19.7% | * | -4.8% | -4.1% | * |
| Medicaid2 | 17.0% | 16.4% | 38.5% | 16.1% | 11.4% | 38.5% |
| Employer-sponsored3 | 11.1% | 10.2% | 10.6% | 4.5% | 5.9% | 10.6% |
| Individually-purchased only | 10.3% | 6.1% | 11.2% | 11.7% | 16.3% | 11.2% |
| All other4 | 27.9% | 25.4% | 45.1% | 24.7% | 21.6% | 45.1% |
|
1Each person has been assigned to one supplementary insurance category but they may or may not obtain their drug insurance coverage from that source. 2 Includes beneficiaries receiving full Medicaid benefits, as well as QMBs and SLMBs. 3 Includes those who only had employer-sponsored supplemental insurance and those who had both employer-sponsored and individually-purchased supplemental insurance. 4 Includes other public programs such as VA, DOD, and State Pharmaceutical Assistance Programs for low-income elderly, as well as non-risk HMOs (cost and HCPP plans). * Number is unreliable because of small sample size. Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1995 and 1996. |
||||||
Factors in Spending Growth
The very rapid growth in drug spending in recent years has been attributed to a variety of factors:
- Part of the growth in drug spending is attributable to price increases for existing drugs. Although price increases have been less than one percentage point above general inflation since 1994, these increases (including the effect of general inflation) still contribute significantly to the total increase in drug spending.32
- Second, the nature of the drugs people receive has changed. There has been a shift toward newer drugs. The number of new drugs introduced each year has grown rapidly, from 23 new drugs introduced in 1990 to 53 new drugs introduced in 1996; the Pharmaceutical Research and Manufacturers of America (PhRMA) estimates that 100 new drugs were introduced in 1997 and 1998.33 Many of these drugs have been breakthrough treatments; others are merely incremental improvements over existing therapies, or “me tooSecond, the nature of the drugs people receive has changed. There has been a shift toward newer drugs. The number of new drugs introduced each year has grown rapidly, from 23 new drugs introduced in 1990 to 53 new drugs introduced in 1996; the Pharmaceutical Research and Manufacturers
- Finally, utilization has grown dramatically. The number of prescriptions filled by retail and mail-order pharmacies grew by 32 percent between 1992 and 1998 (NACDS). There are a number of possible reasons. An aging population may have a higher incidence of chronic conditions for which drug therapy is appropriate, although these demographic changes are occurring only gradually. The use of prescription drugs is also likely growing because of changes in medical practice, including the rise in managed care, Finally, utilization has grown dramatically. The number of prescriptions filled by retail and mail-order pharmacies grew by 32 percent between 1992 and 1998 (NACDS). There are a number of possible reasons. An aging population may have a higher incidence of chronic conditions for which drug therapy is appropriate, although these demographic changes are occurring only gradually. The use of prescription drugs is also likely growing because of changes in medical practice, including the rise
A recent study by the Barents Group for the National Institute for Health Care Management (NIHCM) attempted to measure the relative importance of different factors in the growth of drug spending, as shown in Table 2-33. In general, the study split inflation into two categories: “price” effects and “utilization” effects. Each of these effects were further split between older drugs (drugs that entered the market before 1992) and new drugs (drugs that entered the market in 1992 or later).
The study found that about two thirds of spending growth from 1993 to 1998 was attributable to price. Of this portion, 22 percentage points were attributable to pure price increases for older drugs. Another 42 percentage points reflected the fact that newer drugs cost more than older drugs: the study estimated that the average 1998 price for drugs introduced in 1992 or later was $71.49 per prescription, compared to $30.47 for previously existing drugs. This difference reflects higher initial introduction prices as well as price increases after introduction. The study did not attempt to measure how much of this difference reflects changes in quality as better, newer drugs replace older, less effective medications.
The study reported that increased utilization accounted for about one third of spending growth. If price levels and the mix of prices had not changed between 1993 and 1998, 36 percent of the total spending growth would still have occurred as a result of the increased number of prescriptions. Increased utilization of newer drugs contributed almost twice as much as utilization of older drugs to this increase.
| Percent of rise in drug spending attributable to prices (at introduction and subsequent increases) | Percent of rise in drug spending attributable to utilization | Total | |
|---|---|---|---|
| New drugs (1992 or later) | 42% | 23% | 65% |
| Older drugs | 22% | 13% | 35% |
| Total | 64% | 36% | 100% |
| Source: National Institute for Health Care Management Research and Educational Foundation, Factors Affecting the Growth of Prescription Drug Expenditures, Washington, 1999. | |||
Table 2-34 shows some of the components of spending growth for Medicare beneficiaries. Both utilization and cost per prescription grew from 1995 to 1996. This analysis did not attempt to measure changes in the mix of drugs. Average cost per prescription grew by 6.4 percent, more rapidly than during the preceding years. The cost change was larger than the change in utilization for HMO enrollees and those in employer plans. The reverse was true for Medicaid beneficiaries and purchasers of individual coverage; for these groups utilization grew faster than price. For beneficiaries with no Medicare supplement, both utilization and average price dropped. This may mean that these beneficiaries were less able to afford prescription drugs. However, it might also suggest that the highest spenders in 1995 were disproportionately represented among beneficiaries who newly gained drug coverage in 1996.
| Type of Insurance Coverage | Growth in Average Number of Prescriptions Per Beneficiary | Growth in Average Expenditure per Prescription | ||||
|---|---|---|---|---|---|---|
| Total | Covered | Not covered | Total | Covered | Not Covered | |
| All Beneficiaries | 5.5% | 4.2% | 4.4% | 6.4% | 7.1% | 2.5% |
| No Supplemental Coverage (FFS Medicare only) | -2.8% | -2.8% | -4.6% | -4.6% | ||
| Supplemental Coverage: | ||||||
| Medicare Risk HMO | 7.9% | 8.5% | * | 9.3% | 10.3% | * |
| Medicaid2 | 8.5% | 8.1% | 20.6% | 7.8% | 7.7% | 14.9% |
| Employer-sponsored3 | 2.9% | 2.5% | 3.9% | 8.0% | 7.4% | 6.5% |
| Individually-purchased only | 7.2% | 6.0% | 6.8% | 2.9% | 0.2% | 4.1% |
| All other4 | 19.0% | 17.4% | 27.6% | 7.4% | 6.9% | 13.7% |
|
1Each person has been assigned to one supplementary insurance category but they may or may not obtain their drug insurance coverage from that source. 2Includes beneficiaries receiving full Medicaid benefits, as well as QMBs and SLMBs. 3 Includes those who only had employer-sponsored supplemental insurance and those who had both employer-sponsored and individually-purchased supplemental insurance. 4 Includes other public programs such as VA, DOD, and State Pharmaceutical Assistance Programs for low-income elderly, as well as non-risk HMOs (cost and HCPP plans). * Number is unreliable because of small sample size. Source: Information and Methods Group, Office of Strategic Planning, Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)): Medicare Current Beneficiary Survey Cost and Use File, 1995 and 1996. |
||||||
Endnotes
1 - This and subsequent MEPS-based tables on the non-Medicare population are limited to nonelderly people without Medicare. They omit the very small number of elderly people—an estimated 150,000—who are not covered by Medicare.
2 - Appendix B contains a further discussion of use rates and how users are treated in this report.
3 - The difference in the price of prescriptions purchased by covered people compared to those purchased by noncovered people is only statistically significant at the five percent level on a one-tail test.
4 - In some cases, people with supplemental insurance may even be paying premiums for the addition of a drug benefit that cost more on average than the maximum value of the drug benefit.
5 - Note that not every beneficiary with drug coverage obtained that coverage through the same source that provided his or her primary Medicare supplemental coverage.
6 - However, Medicaid beneficiaries’ spending, utilization, and out-of-pocket spending is not significantly different from the next closest group in each case: their spending is only different from spending by those with employer-sponsored coverage on a one-tail test, their utilization is not significantly different from beneficiaries with other public insurance, and their out-of-pocket spending is not significantly different from beneficiaries enrolled in an HMO.
7 - Differences among beneficiaries enrolled in Medicare HMOs are not statistically significant, primarily because of the small number of beneficiaries in HMOs without drug coverage.
8 - Income is given as a percentage of the federal poverty threshold, which differs by age and household size. For beneficiaries over 65, these calculations use a poverty threshold of $7,525 for an individual, and $9,491 for those who lived with others. For beneficiaries under 65 they use $8,163 for an individual, and $10,564 for those who lived with others.
9 - In the income range between 136 percent and 150 percent of poverty, the difference in spending between beneficiaries with and without coverage is not statistically significant.
10 - However, the smallest difference, between users with and without coverage with incomes between 100 and 200 percent of poverty, is statistically significant only on a one-tail test.
11 - For persons without coverage, the difference in spending between those with good health status and those with very good health status is not statistically significant, nor is the difference between persons with fair health and those with poor health.
12 - Among beneficiaries with the same health status, the spending difference between those with and without coverage is statistically significant for all beneficiaries except those in excellent health.
13 - The difference between spending by covered beneficiaries aged 65 to 69 and those aged 85 and over is not statistically significant.
14 - Hellinger (1995), PPRC (1996), Riley (1996), Hamilton (1999), and Call et al (1999); see bibliography in Appendix A for detailed citations.
15 - Ettner (1997), Wolfe and Goddeeris (1991), and Atherly (1999) found evidence of selection. Cartwright, Hu, and Huang (1992), Hurd and McGarry (1997), and Lillard et al. (1999) did not. See bibliography in Appendix A for detailed citations.
16 - Atherly, A. (1999). “The Effect of Medicare Supplemental Insurance on Medicare Expenditures.” Department of Health Policy and Management. School of Public Health. Emory University. Atlanta, GA. Unpublished Paper.
17 - None of the measures used are direct indicators of the need for prescription drugs. However, that drug spending rises with deteriorating health for both covered and noncovered people suggests that the measures are at least strongly correlated with prescription drug need.
18 - The experimental design allowed researchers to eliminate the effects of self-selection by randomly assigning participants to plans that required various levels of copayment, ranging from full first-dollar coverage to 95 percent coinsurance (to simulate non-coverage).
19 - Brook, Robert, et al., “Does Free Care Improve Adults’ Health? Results from a Randomized Controlled Trial,” NEJM, 309 (23), Dec. 8, 1983, 1426-1434; Lohr, Kathleen, et al., “Use of Medical Care in the RAND Health Insurance Experiment: Diagnosis and Service-Specific Analyses in a Randomized Controlled Trial,” Medical Care 24(9) (Supplement), Sept. 1986, S74-S77; Manning, Willard G., et al. "Health Insurance and the Demand for Medical Care: Evidence from a Randomized Experiment," American Economic Review, 77(3), June 1987, 251-277.
20 - Stuart and Grana (1998), Stuart and Zacker (1999), Ya-chen (1999), and Lillard, Rogowski, and Kingston (1999); see bibliography in Appendix A for detailed citations.
21 - Coulson NE. and Stuart, BC. (1995) “Insurance Choice and the Demand for Prescription Drugs.” Southern Economic Journal. 61(4): 1146-1157.
22 - General Accounting Office. Prescription Drugs and the Elderly: Many Still Receive Potentially Harmful Drugs Despite Recent Improvements, March 1996.
23 - The difference between covered and noncovered users in the next to highest quintile is significant only on a one tail test.
24 - A consumer unit comprises either: (1) all members of a household who are related by blood, marriage, adoption, or other legal arrangements; (2) a person living alone, or a person sharing a household who is financially independent; or (3) two or more persons living together who pool their income to make joint expenditure decisions. The reference person is the person named when the respondent is asked to "Start with the name of the person or one of the persons who owns or rents the home." It is with respect to this person that the relationship of the other consumer unit members is determined.
25 - Total reported spending for units with reported income below $15,000 actually exceeds $15,000. Data in the tables are for complete income reporters. These are units that provide values for at least one of the major sources of their income. However, even complete income reporters do not necessarily provide full accounting of their income. In addition, some units may spend more than their income by borrowing or by using up assets; the latter may be more likely for the elderly.
26 - Note that these figures cannot be compared to the MCBS and MEPS out-of-pocket numbers for individuals because the year is different, and many units have more than one individual.
27 - The MCBS Access to Care component for 1996 asked whether the respondent had failed to fill one or more prescriptions during the year, and why. By contrast, the NHIS question asks whether a respondent failed to receive a needed drug. This study does not include an analysis of the MCBS question because it does not directly capture people who might never have had a drug prescribed because they did not go to the doctor.
28 - The exception is for people with Medicaid, for whom the difference between income groups is not statistically significant.
29 - Note that the analysis included beneficiary use of drugs associated with hypertension, but some of these medications may have been prescribed for purposes other than treatment of hypertension. For example, diuretics could be prescribed for a number of purposes.
30 - However, Chapter 3 will show that people with and without coverage purchase a similar mix of brand-name and generic drugs. More analysis is needed on this issue.
31 - Copayments are not necessarily less burdensome than coinsurance. On a $35 prescription, 20 percent coinsurance would be $7, less than the copayments imposed by many plans. However, the elimination of deductibles probably reduced consumer liabilities considerably. In addition, coinsurance rises automatically with drug inflation, while insurers have to change copayments periodically to keep pace with inflation.
32 - HCFA(now known as CMS), based on the CPI and the GDP deflator.
33 - PhRMA, Drug Utilization And Managed Care, Sept. 1998.
Chapter 3. Prescription Drug Prices
One of the charges of this study was to examine the differences in drug prices paid by people with and without prescription drug coverage. Prices and price differentials are important measures for understanding the market for pharmaceuticals in the United States. Analyses of pharmaceutical pricing, however, are complicated by the intricacies of this market: the process by which drug prices are determined is highly complex, involving numerous interactions and arrangements among manufacturers, wholesalers, retailers, insurers, pharmacy benefit managers (PBMs), and consumers.
In order to explain the complexity of the market for pharmaceuticals, this chapter begins with a description of the distribution channels for prescription drugs and how prices are established for different purchasers. The chapter then explores the question of whether prices paid for drugs at the point of purchase differ between cash customers and insurers, using data from the Medical Expenditure Panel Survey (MEPS) and from a widely used private sector data source, IMS Health.
A key limitation on the analysis of prescription drug prices in this study is our inability to incorporate the effect of rebates provided by manufacturers to insurers or PBMs. In many instances, a manufacturer will provide a cash rebate to an insurer or PBM if the manufacturer's drugs are used by the insurer's or PBM's enrollee. Unfortunately, information about the relative size, prevalence and characteristics of these rebate arrangements is quite limited - these are confidential, private arrangements negotiated between manufacturers and insurers or PBMs. Information from industry sources and the available literature indicate that rebate amounts vary considerably by type of arrangement and by drug. They are quite considerable in some instances and relatively modest in others. Because these rebates primarily affect the ultimate price paid for prescriptions of insured individuals, failure to account accurately for the value of such rebates tends to understate the price differences facing uninsured and insured individuals.
Although we were unable to obtain information on rebates, we were able to obtain specific data from two sources, MEPS and IMS Health, that enable us to examine differences in the prices paid by consumers at the retail pharmacy point of sale. These data compare the amount paid to the pharmacy by uninsured customers to the reimbursement received by the pharmacy for insured customers (i.e., reimbursement from an insurer or PBM plus the customer's cost sharing). Given the greater market leverage of third-party payers relative to individual consumers, it might be expected that uninsured customers will pay more than insurers for the same drugs at the retail pharmacy level. Our results from both MEPS and IMS Health support this hypothesis.
Key findings include:
- At the retail pharmacy level:
- Individuals without drug coverage pay a higher price at the retail pharmacy than the total price paid on behalf of those with drug coverage (based on analysis of MEPS data that do not include rebates but look across all drug purchases holding drug type, form, strength, and quantity constant). The differences generally held up when examining the Medicare and non-Medicare populations.
- Cash customers (including those without coverage and those with indemnity coverage) pay more for a given drug than those with third-party payments at the point of sale (based on IMS Health data for over 90 percent of the most commonly prescribed drugs). In 1999, excluding the effect of rebates, the typical cash customer paid nearly 15 percent more than the customer with third-party coverage. For a quarter of the most common drugs, the price difference between cash and third parties was even higher - over 20 percent. For the most commonly prescribed drugs, the price difference between cash customers and those with third-party coverage grew substantially larger between 1996 and 1999.
- The pattern of differences in the price paid by cash customers and those with third-party payments is different for generic and brand name drugs (based on both MEPS and IMS Health data). Percentage differences in the price paid are often smaller for brand name drugs, but absolute differences may be larger because average prices for brand name drugs are considerably higher.
- Data on manufacturer rebates, if available, would reduce the total amount paid by the insurer or PBM on behalf of insured customers, increasing the difference in the total net price. Data on rebate arrangements, however, are confidential and unavailable to this study. In some instances, the amount of the rebate may be significantly more than the price differences observed at the retail pharmacy level. In other cases, the rebates may add only modestly to the observed differences.
- Various sources produce estimates of rebates ranging from 2 percent to 35 percent of drug sales prices. These rebates are not reflected in retail prices, but are instead paid directly to insurers and other organizations that manage drug benefits after they have already reimbursed the pharmacy.
Distribution and Pricing of Prescription Drugs
Most noninstitutionalized people, regardless of their coverage status, obtain prescription drugs through some form of retail pharmacy, including independent pharmacies, chains, pharmacies in supermarkets or mass merchandisers, and mail-order pharmacies. In 1998, sales through retail outlets accounted for 90 percent of total outpatient prescription drug sales (excluding sales to hospitals and long-term care facilities or agencies).1
The following description of this system will begin with the simplest series of transactions, those that culminate in a retail purchase by a consumer who pays for a prescription in full at the point of sale. Some of these customers, referred to as cash customers in this chapter, may file a claim with their insurer for reimbursement after the transaction. This description will be followed by a summary of how prices are established for private insurers and PBMs, along with a review of the special pricing systems for Medicaid, the VA, and certain other favored purchasers.
The prices paid by these various types of customers are illustrated in Table 3-1, which portrays illustrative pricing for brand name drugs. The prices in the table are based on a composite of commonly prescribed brand name drugs and reflect documented relationships among the prices for different transactions.2 Ranges of prices are included where more precise information, particularly on drug rebates, cannot be documented. Actual price relationships vary substantially by drug, and are quite different for generic drugs. Generic drugs, for example, have much lower prices on average and the fixed costs for the pharmacy of dispensing the prescription represent a much higher proportion of the final retail price. Furthermore, approaches to pricing generic drugs in the industry are different.
| Cash Customers (No 3rd Party Payment at Point of Sale) | Insurers and PBMs | HMO ** | Medicaid | Federal Supply Schedule | |
|---|---|---|---|---|---|
| List price (AWP) | $50 | ||||
| Manufacturer's price (manufacturer to wholesaler or other entity) | $40 (AWP-20%) |
$40* (AWP-20%) |
$34 (AWP-33%) |
$40* | $24 (AWP-52%) |
| Acquisition price (wholesaler to pharmacy) | $41 | $41 | n/a | $41 | n/a |
| Retail price at pharmacy (total of amounts paid by customer and reimbursed by 3rd party payer) | $52 (AWP+ 4%) |
$46* (AWP-13% +$2.50) |
$41 +$2.50 | ||
| Retail price, less typical manufacturer rebate | n/a | $30 to $44 (5% to 35% rebate) |
$30 to $37 (15.1% to 30% rebate) |
||
| Ultimate (net) amount paid by final purchaser and/or consumer | $52 | $30 to $44 | $34 (avg.) | $30 to $37 $34 (avg.) |
$24 |
|
n/a = not applicable * without rebate ** This column refers only to HMOs that buy directly from manufacturers. Notes: (1) Prices are based on a composite of several commonly prescribed brand-name drugs for a typical quantity of pills. For some cells in the table, the relative relationships have been calculated based on relationships reported in How Increased Competition from Generic Drugs Has Affected Prices and Returns in the Pharmaceutical Industry ( CBO, 1998) study and on other relationships widely reported by industry sources. |
|||||
The share of purchasers who pay in full at the time of the transaction (referred to as cash customers) has been steadily decreasing in recent years. This category includes both those with no insurance coverage for drugs and those with indemnity coverage who file claims after the retail transaction is complete. In 1990, 63 percent of retail prescriptions involved cash customers, while 37 percent involved billing by the pharmacy to third-party payers or Medicaid (Figure 3-1). By 1998, only 25 percent of prescriptions were paid for by cash customers.3
Figure 3-1. Payment Sources for Prescription Drug Purchases, 1990-1998
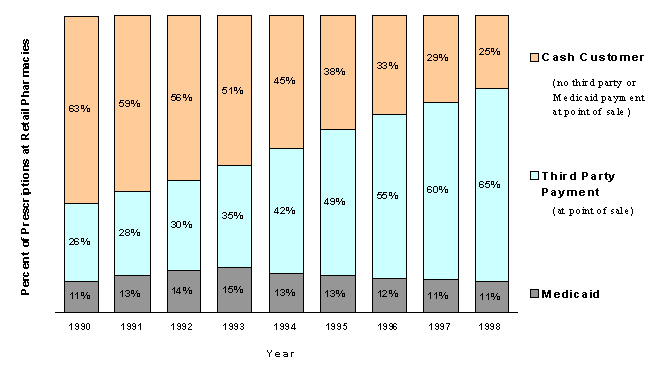
Source: IMS Health Retail Method-of-Payment ReportTM, 1999.
This trend does not represent a growth of coverage as much as it represents a shift in how drug coverage works. During the 1990s, the common approach has shifted from indemnity coverage to coverage that is managed at the point of sale.
With indemnity insurance, a customer typically pays cash for the full cost of the prescription at the pharmacy and then files a claim for reimbursement from the insurer. As noted in Chapter 1, most people with private group coverage for prescription drugs have some form of managed drug benefit, administered by a PBM or sometimes directly by an HMO or other insurer. Under PBM administration, point of sale transactions are now the norm. Under such a transaction, the pharmacist uses a computer system to determine the deductible, copayment, or coinsurance, which the customer pays at the retail counter.
Management of drug benefits has become the norm for group coverage. For nongroup coverage, including Medigap, it is much less common. In the case of Medigap, for example, the insurer's liability is limited to half of total spending between a deductible and a cap; insurers generally have not seen enough potential for savings to invest in benefit management.
As noted previously, the ultimate prices paid for prescription drugs by or on behalf of consumers are established through an intricate system involving pharmaceutical manufacturers, wholesalers, retailers, and insurers or other managers of drug benefits. Accordingly, the descriptions below, despite the apparent level of detail provided, are necessarily simplified in order to convey the key characteristics of this system.
Pricing for Cash Customers
Cash customers at retail pharmacies include people without coverage and people with indemnity coverage who pay for their own prescriptions and are later reimbursed by their insurer.4 Prices are set by a series of transactions linking the manufacturer to the cash customer through the wholesaler and the retail pharmacy, as shown in Table 3-1.
Sale by Manufacturer to Wholesaler
In the first transaction, the manufacturer sells the drug to a wholesaler. The manufacturer establishes a price that varies by the form and strength of the product; for example, a 500 milligram tablet of a given drug will have a different price from a 250 milligram tablet of the same drug. Price may also vary by packaging; for example, a package containing 1,000 tablets of a given medication might have a lower price per tablet than a package of 500 tablets. When there is only a single manufacturer of a drug, as is often the case with a brand-name drug, there is only one price for a specific product and package size. Once generic versions of the drug become available, the equivalent medication (in form, strength, and package) may be offered at different prices by different manufacturers.5
Wholesalers may sometimes receive discounts from manufacturers, based on volume or prompt payment. A manufacturer of a multi-source drug (i.e., one that is produced by more than one manufacturer) may offer a discount to induce wholesalers to promote its particular version of the drug. Thus the manufacturer's price is only a guideline, and may not represent the price that all wholesalers ultimately pay for the drug. The manufacturer's price itself represents both the cost of producing the drug and a share of the manufacturer's research and development costs, taxes, and profits. For any particular drug, the price may reflect the market position of the drug more than the cost of its production; for example, a company may set a higher price for an innovator drug than for one which has several competitors. Further breakdown of these components of the manufacturer's price is outside the scope of this study.
Sale by Wholesaler to Retail Pharmacy
In the second transaction, the wholesaler sells the drug to a retail pharmacy at a price reflecting its cost of acquiring the drug plus a markup. This price may be referred to as the wholesale price or acquisition price. A price that is commonly cited in the industry is the "average wholesale price," or AWP. Despite what this name would suggest, the AWP is not the average of the amounts actually paid by retail pharmacies to wholesalers for a particular drug. Instead it is a published wholesale price or "list price" suggested by the manufacturer of the drug. A wholesaler may sell specific drugs to all pharmacies at prices below the AWP, or may grant a general discount to certain pharmacies. Thus, although the AWP is often used by pharmacies as a cost basis for pricing purposes, it does not represent the actual cost to a retail pharmacy of acquiring the drug. It is merely a wholesale list price that can be used as a benchmark in comparing retail and wholesale prices.6
Industry sources suggest that the price charged by the manufacturer to the wholesaler typically runs about 20 percent below the list price or AWP. In the example in Table 3-1, the acquisition price (paid by the pharmacy to the wholesaler) is $9 below the AWP. The markup added by the wholesaler is generally small, perhaps 2 percent to 4 percent.
Sale by Retail Pharmacy to the Consumer
In the third transaction, the pharmacy sells the drug to a consumer at a price that includes its cost for acquiring the drug from the wholesaler plus a retail markup. Part of this markup is a fixed cost that is not related to the cost of acquiring a specific drug. This is because the cost to the pharmacy of filling a prescription for a low-price drug is likely to be the same as for a high-price drug. As a result, the fixed cost is a higher percentage markup over acquisition cost for a low-price drug than for a high-price one. Different pharmacies have different fixed costs. Because of economies of scale, a large chain pharmacy may have lower costs than a small independent one.
Part of the markup varies by drug. Pharmacies employ a variety of pricing strategies when determining this markup for their sales to cash customers. For example, they may set a lower markup for maintenance medications and a higher markup for acute medications, or may routinely discount certain commonly used medications as "loss leaders," in order to attract cash customers who will then buy other medications or other merchandise.
Some industry sources have suggested that retail markups in the range of 20 percent to 25 percent over the pharmacy's acquisition price are typical. This markup includes both the fixed operating costs of the pharmacy as well as taxes and profits. These same sources also suggest that the fixed costs represent most of this markup amount. In the example in Table 3-1, the $52 cash price is 4 percent above the AWP. The pharmacy in this example includes a retail markup of about 25 percent over its acquisition cost in the price charged to its cash customers.
Pharmacies may also offer across-the-board discounts on drugs to certain groups of cash customers, such as senior citizens. In addition, some organizations negotiate discounts on behalf of people without drug coverage or people who pay cash because their coverage is an indemnity plan. For example, AARP offers its members a Member Choice Program operated by RPS, Inc. In return for a $15 annual fee, members receive access to discounts negotiated with pharmacies by RPS. AARP reports that members receive average discounts of $7.26 per prescription or about $160 per year. Some insurers that sell both indemnity coverage and coverage that pays pharmacies directly may offer their indemnity purchasers access to the discounts they have negotiated on behalf of their other enrollees. This is true of United Health Care, which sells Medigap coverage through AARP, and of some Blue Cross/Blue Shield Medigap plans. These various discounts are part of the average prices cited later in this chapter.
Pricing for Insurers and Pharmacy Benefit Managers
The simple pricing model just described applies to cash transactions but not to those in which the retail pharmacy is paid by a group insurer, employer, or other third party at the point of sale. This section will describe mechanisms affecting the prices paid by private third parties that manage drug benefits. Again, because management by a PBM is most common, this term will be used as a shorthand for all private entities managing drug benefits.7
Because a PBM may manage the drug benefit for a large number of individuals, it can negotiate discounts at both ends of the pricing chain: from the manufacturer and from the retail pharmacy. There is little published data about the size of the discounts obtained by PBMs and private insurers, either from manufacturers or from retailers. Discounts from retailers will be estimated later in this chapter, using the MEPS and IMS Health data. Manufacturer rebates are not reflected in these data sources. Rebate agreements are highly confidential and most information about them derives from anecdotes.
Retail Discounts
The price paid to a retail pharmacy for a given drug is negotiated by the PBM and the pharmacy or pharmacy chain. Typically the PBM will take into account its estimate of the cost to the pharmacy of acquiring the drug (usually assuming that the pharmacy has paid something less than the AWP) and offer a dispensing fee above that amount. This dispensing fee is commonly a fixed dollar add-on (in the range of $2.50) that is not related to the cost of acquiring a specific drug.8 Because some PBMs cover a large share of the market, a pharmacy will often accept a price that is less than it would charge to cash customers. The PBM's negotiating power may be offset, however, by its need to assure that its enrollees have access to convenient pharmacies. It might offer a higher price to a large chain than to scattered independent pharmacies.9
Discussions with industry experts conducted during the preparation of this report have provided current information on typical PBM payments to retailers. These experts estimate that payments for brand-name drugs are in the range of AWP minus 13 to 15 percent, plus a $2.50 dispensing fee. (The range from 13 to 15 percent depends primarily on how restricted the pharmacy network is.) The example in Table 3-1 illustrates this type of discount, resulting in a price that is lower than that faced by cash customers but in this case still offers a 12 percent markup over the pharmacy's acquisition price. For some drugs, however, a pharmacy may be forced to accept reimbursement from the PBM that does not cover the pharmacy's cost of acquiring the drug (let alone its operating costs). The PBM has considerable leverage in this relationship, especially as the proportion of drugs sold through PBM-managed arrangements grows (Figure 3-1). The pharmacy is left with an option of refusing the large share of business, raising its prices for cash customers, or reducing its operating margin.
For generic drugs, about three-fourths are reimbursed using limits known as maximum allowable cost (MAC). These limits are established by PBMs, based on the lowest estimated acquisition cost for any of the generic equivalents of a given drug. The MAC tends to be 50 to 60 percent below AWP. The remaining one-fourth of generics are reportedly reimbursed, like brand-name drugs, at AWP minus 13 to 15 percent. The dispensing fee for generics tends to be the same as for brand drugs, but sometimes it is 25 or 50 cents higher, to encourage generic substitution by pharmacies.
Manufacturer Rebates
The second type of discount that the PBM gets is a negotiated rebate paid directly from the manufacturer to the PBM. This rebate does not affect the price paid by a wholesaler to a manufacturer for the drug, the price paid by a retail pharmacy to the wholesaler, or the price paid by the PBM to the pharmacy. It is a separate transaction between the PBM and the manufacturer and thus affects the total amount spent by the PBM. To the extent that a portion of the rebate is passed along, the insurer, employer, or beneficiary may realize a part of these savings.
A key tool in determining whether rebates are available and how large they are is the use of a restrictive formulary, a list of drugs that the PBM has established as preferred for its enrollees. If there are multiple brand-name drugs available for a given condition, the PBM may include some on its formulary and not others. Enrollees who obtain a non-formulary drug may pay higher copayments, or the drug may not be covered at all. Pharmacies dealing with the PBM may be encouraged to contact physicians who have prescribed non-formulary drugs and suggest a formulary alternative.10 Physicians affiliated with the health plan using the PBM may also face pressure to prescribe formulary drugs. In addition, PBMs will commonly require or encourage substitution of generic equivalents for brand-name drugs when these are available. Again, they may charge higher copayments for brand name drugs, or limit reimbursement to the generic price even when the brand-name drug has been dispensed.
Manufacturers of brand-name drugs that treat conditions for which an alternative brand-name treatment is available thus have a strong incentive to grant discounts to the PBM in return for the inclusion of their drugs in the formulary. If generic equivalents are available, the manufacturer may also grant a discount to make the price of its brand-name product more competitive. These discounts usually take the form of direct rebates from the manufacturer to the PBM. For example, in the simplest rebate arrangement, the PBM may report periodically to a manufacturer the number of prescriptions for a given drug that the PBM's enrollees have filled; the manufacturer then pays the PBM an agreed-upon amount for each prescription. In addition or as an alternative to a per-prescription rebate, manufacturers and PBMs also negotiate arrangements where the PBM is reimbursed for moving market share -- causing a significant increase in the number of prescriptions for the manufacturer's drug.
One study by the General Accounting Office attempted to quantify the value of rebates obtained by PBMs contracting with plans participating in the Federal Employees Health Benefits Program (FEHBP). Blue Cross/Blue Shield paid about $1.4 billion for FEHBP pharmacy benefits in 1995 and estimated that its PBM had saved $505 million, of which 21.2 percent was attributable to manufacturer discounts or rebates and 52.3 percent to discounts from retail and mail-order pharmacies.11 If costs in the absence of the PBM would have been $1.9 billion, this suggests retail or mail-order discounts of 14 percent (consistent with the estimates cited above) and manufacturer rebates of 5 to 6 percent (or slightly more, assuming the PBM did not pass all rebates fully to Blue Cross). Overall FEHBP plans estimated that their savings from manufacturer rebates ranged from 2 to 21 percent of total savings. Industry representatives report that rebate savings can be much higher (35 percent) on selected drugs.12
Table 3-1 uses a range of rebate amounts to show the net effect on the price if the unmeasured rebates were taken into account. This amounts to a hypothetical retail price of $30 to $44 (compared to the total $46 price that the pharmacy receives from the customer and the insurer) if the discounts were applied at the pharmacy instead of through rebates to the PBMs. An alternative way to display this discount would be to reduce the manufacturer's price by the rebate or by some portion of it. Because rebates cannot be measured, the analysis reported later in the chapter does not reflect this additional discount.
PBMs that operate under contract to an insurer or self-insured employer are required to pass on most of the rebates. Industry sources report that the insurer or employer typically receives 70 to 90 percent of the rebates. In addition, the PBM will often guarantee a minimum per-prescription rebate, in case actual rebates received from manufacturers are lower than expected. While estimates differ, industry experts report that the value of rebates passed on to insurers or employers may average about $1.00 per claim.13
In addition to cash rebates, industry analysts have reported that PBMs may receive noncash benefits from manufacturers or cash rebates that are not tied to a particular drug. For example, PBMs may receive rebates from manufacturers in return for agreements with regard to the content of their communications with physicians about the use of certain drugs (sometimes called counter-detailing). PBMs that operate their own mail-order pharmacies may receive extra discounts on drugs purchased by those pharmacies. Other PBMs may receive support for development of disease management systems or other research activities. Some industry analysts believe that the value of these other considerations may exceed the amount of cash rebates. The PBM may not be obligated to pass these benefits on to plan sponsors.
Finally, a PBM can realize further savings by encouraging enrollees to use a mail-order pharmacy. (Some PBMs operate their own mail-order pharmacies.) Enrollees may pay a lower copayment when using a mail-order pharmacy or may be required to use mail-order for drugs to be taken over a long period, such as maintenance drugs for chronic conditions. Mail-order pharmacies can operate with a smaller markup than other retail pharmacies, because of economies of scale and lower overhead. They may also be more successful in encouraging prescribing physicians to agree to substitute formulary for non-formulary drugs, because they have more time to contact a physician before filling a prescription.
Pricing for Favored Private Purchasers (HMOs and Hospitals)
Not all drugs dispensed by pharmacies are purchased from wholesalers. Institutions that operate their own outpatient pharmacies, such as hospitals and clinics, may deal directly with manufacturers, either individually or through buying groups. They typically save the wholesaler markup. In addition, they may receive a price lower than that offered by the manufacturer to wholesalers. Table 3-2 shows estimates by the Congressional Budget Office, based on IMS data, of the discounts received by these purchasers, which represent a total of about 14 percent of the market. These discounts are reported relative to the average acquisition price paid by retail pharmacies, excluding mail order, in 1994.
| Percent of average invoice price to retail pharmacies | |
|---|---|
| Retail pharmacies | 100% |
| Hospitals | 91% |
| Long-term care facilities | 95% |
| Health maintenance organizations | 82% |
| Federal facilities* | 58% |
| Clinics | 91% |
|
* Federal facilities include the Department of Veterans Affairs, the Defense Department, the Indian Health Service, and the Public Health Service. Similar prices are available to state pharmaceutical assistance programs. Source: Congressional Budget Office, How Increased Competition from Generic Drugs Has Affected Prices and Returns in the Pharmaceutical Industry, Washington, July 1998. |
|
Rebates and other discounts that do not appear on an invoice are not included in the price relationships reported in this analysis. Because of their direct purchasing relationship with manufacturers, however, these entities are less dependent on rebates based on the amount of a particular drug dispensed. It appears that rebates play a smaller role for these purchasers than for PBMs that do not purchase drugs directly. More complete pricing data are unavailable.
The estimate for HMOs in Table 3-2 reflects discounts to the relatively few HMOs, such as some of the Kaiser Permanente plans and other staff or group-model HMOs, that operate their own pharmacies and buy drugs directly from manufacturers. This is not an estimate of the size of rebates that are received by the majority of HMOs, which manage drug costs and utilization through PBMs and whose enrollees obtain their prescriptions through retail outlets.
In Table 3-1, the HMO price is $34, based on the relationship reported by CBO (Table 3-2) and confirmed by other information obtained from industry sources. There is evidence that some HMOs obtain at least some drugs at prices substantially below the price in this illustration. Recently, the HHS Inspector General reported on two HMOs that were able to purchase a drug at prices considerably below the reported best price for that drug.14
Pricing for Federal Facilities and Agencies
Prices paid to manufacturers by the VA, other federal agencies, and certain other entities, such as Indian tribal governments, are set by the Federal Supply Schedule (FSS). Under the Veterans Health Care Act of 1992, manufacturers must make drugs available to covered entities at the FSS price as a condition of eligibility for Medicaid reimbursement.
FSS prices are negotiated with manufacturers by the VA.15 In general, the FSS price may be no higher than the lowest contractual price charged by the manufacturer to any nonfederal purchaser under similar terms and conditions. In order to determine this price, manufacturers supply the VA with information on price discounts and rebates offered to different customers and the terms and conditions involved. Under certain conditions, the VA may accept an FSS price that is higher than the price offered to some nonfederal customers. According to the GAO, average FSS prices are more than 50 percent below the nonfederal average manufacturer's price.16 This result is somewhat lower than the relationship shown in Table 3-2 based on the CBO study.
For certain drugs sold to the VA, the Department of Defense, the Public Health Service, and the Coast Guard, the manufacturer must charge the lesser of the FSS or a "federal ceiling price."17 The federal ceiling price is set at 76 percent of the average manufacturer's price; this limit may be higher or lower than the FSS. The rule applies only to brand name drugs without competition or innovator multiple-source drugs.18
In the example in Table 3-1, the federal supply schedule price of $24 is about 60 percent of the manufacturer's price and well under the various retail prices charged to different types of customers. Not surprisingly, it is fairly close to the $30 that represents the lowest price that might be achieved by insurers and PBMs (and Medicaid) in cases where they get the largest manufacturer rebates. This makes sense for a "best contractual price" standard. If the table could represent the full range of prices achieved by some buyers for some drugs, there would probably be cases where other third parties achieved prices below this FSS price.
More generally, the fact the FSS price is generally lower than other prices may have a variety of explanations. These include the small share of the market that federal purchasers represent (less than 2 percent), the effectiveness of the VA as a price negotiator, and the interest that manufacturers may have in making sure their drugs are available to federal facilities and agencies (including VA hospitals that train a large number of physicians).
Pricing for Medicaid Programs
Medicaid programs pay retail pharmacies using fixed cost limits and fixed dispensing fees. For single-source drugs (brand-name drugs without generic equivalents), the cost limit is set at the estimated pharmacy acquisition cost for the drug. For multiple-source drugs (with brand name or generic competitors), the limit is based on a MAC. These are similar in concept to the MACs used by PBMs; some PBMs may simply use the Medicaid MACs, while others develop their own. The Medicaid MACs are published by HCFA(now known as CMS) every six months and are set at 150 percent of the lowest published price for any equivalent drug, plus a dispensing fee.19
Under provisions of the Omnibus Budget Reconciliation Act of 1990, Medicaid programs receive rebates from manufacturers.20 Participation is generally required for a manufacturer's drugs to be eligible for Medicaid reimbursement. For single source drugs and innovator multiple-source drugs, the rebate must equal the difference between the average manufacturer price (AMP) - the average paid by wholesalers - and the manufacturer's "best price." The best price is the lowest price offered by the manufacturer to any purchaser at any time during the year, excluding the special prices for federal purchasers and certain other covered entities.21 The minimum rebate must be 15.1 percent of the AMP. For non-innovator multiple source drugs, the rebate is 11 percent of the AMP; the best price concept does not apply.
In Table 3-1, Medicaid reimburses the retail pharmacy for its acquisition price plus a dispensing fee. As noted, states obtain a minimum 15.1 percent rebate, corresponding to a net effective price of $37. Average rebates are in the range of 21 percent, which would yield an ultimate price of $34, but may range higher for certain drugs.
Sources and Uses of Price Data for This Chapter
As described in the previous section, both the processes by which prices are determined and the ultimate price paid for drugs by various purchasers varies considerably. Other factors also lead to wide differences, such as different pricing strategies between generic and brand name drugs and between single-source and multiple-source drugs. In the remainder of the chapter, the focus is primarily on one facet of pricing - the variation in prices faced by different types of consumers at the retail pharmacy. For reasons of data availability, rebates cannot be incorporated into the prices discussed below.
Price data reported in this chapter are derived from the Agency for Healthcare Research and Quality's Medical Expenditure Panel Survey (MEPS) pharmacy component for 1996 and from IMS Health pharmacy audits for 1996 through 1999. The MEPS data include prices for all drugs furnished to participants on an outpatient basis, including prescriptions filled by retail pharmacies, mail-order pharmacies, and pharmacies within outpatient facilities (such as hospitals, some HMOs, and VA facilities). The IMS data used for this report include prices only for prescriptions filled by retail pharmacies. The two data sets complement each other because MEPS allows for comparisons by more precise definitions of coverage status and collects prescription price information across a broader array of pharmacy outlets, while the IMS Health sample size allows more direct drug by drug comparisons of prices paid by cash versus third-party payers. In addition, the 1996 MEPS data are newly released (December, 1999) and have not yet been widely used. The IMS data are a commonly accepted data source on prices, are widely used by industry, and are the most recent data available.
For the purpose of the discussion below, the price of a prescription drug is the total amount paid by all payers to the pharmacy that filled the prescription. For example, if the pharmacy received a $15.00 payment from a third-party payer and a $5.00 copayment from the consumer, the price of the drug is $20.00. Because rebates from manufacturers to PBMs, insurers, or Medicaid programs do not directly affect the amount paid by the third party to the retail pharmacy, the price measured at the retail pharmacy level is often not the true "price" paid by third-party payers. In most cases, the true price is less than the amount paid in the pharmacy transaction. Finally, for the purposes of price comparisons in this chapter, variants of the same drug sold by different manufacturers are each considered as distinct drugs.
Because this analysis focuses on retail price differences and is unable to incorporate rebates, our approach is different from that of the studies by the Minority Staff of the House Committee on Government Reform (Prescription Drug Pricing in the United States: Drug Companies Profit at the Expense of Older Americans, November 9, 1999). The Committee study focused on the difference between best prices obtained in the market by third-party payers and the higher retail prices paid by cash customers. By contrast, our analysis of MEPS and IMS Health data focuses on differences in retail prices at the retail pharmacy level faced by customers with and without access to the discounts negotiated by third-party payers. Data on manufacturer rebates, if available to this study, would have allowed a more complete analysis of price variation within the market.22 Without access to that information, our analysis tends to understate the ultimate price differences for insured and uninsured customers. Rebates paid by manufacturers to insurers or PBMs increase the difference between the total net price ultimately paid for drugs on behalf of those with some type of third-party drug coverage and the price paid by those who pay totally out of pocket at the retail point of sale.
MEPS Data
The 1996 MEPS includes a pharmacy survey to validate prescription drug utilization reported by respondents, and to obtain direct measures of retail prices paid for drugs. For each prescription drug "event" reported by respondents MEPS endeavors to ascertain the price through the pharmacy survey. The pharmacy data, in combination with the comprehensive respondent data, enables pricing analyses with detailed definitions of coverage status.
However, information was collected from pharmacies in only about half the instances in which a household survey participant mentioned using a specific pharmacy. Overall, of the drug events used to estimate utilization and spending in Chapter 2, about 40 percent have data derived directly from a household/pharmacy match for the same person. Prices were imputed for 60 percent of the remaining events using statistically matched pharmacy events for comparable individuals. To avoid introducing measurement error that could bias comparisons, only non-imputed price data - cases in which there was a direct match of information from a household and a pharmacy - are used in this chapter.23 However, sensitivity analyses (not shown) using the imputed data are very consistent with the data reported here.24
One consequence of this exclusion is that, for any single drug (defined by name, manufacturer, form, and strength), the available sample from MEPS is too small to allow reliable comparison of prices paid by different purchasers. Price comparisons for specific drugs will be made using the IMS data, which have a much larger sample size.25
IMS Health Data
IMS data used in this chapter are drawn from the IMS Health Retail Method-of-Payment Report ™ and Price Trak Report™. IMS collects data from a panel of 34,000 retail pharmacies, including independents, chains, and pharmacies within food stores or mass merchandisers. The IMS sample for these products accounts for over 60 percent of retail outlets and over 70 percent of prescriptions filled in the US, but it omits mail-order pharmacies and pharmacies within facilities, such as outpatient hospital pharmacies, VA pharmacies, and those operated by the few HMOs that have their own pharmacies. Through a variety of electronic media, IMS collects acquisition cost, retail price, and payment source for every new and refilled prescription. Three payment sources are identified: cash, Medicaid, and other third party (essentially private insurance). Note that the IMS data would class an individual who paid cash and was later reimbursed by an indemnity policy in the cash category.26
For this study, 1996 and 1999 price data were obtained for 39 Uniform System of Classification (USC) categories of drugs that together included the 100 most commonly prescribed individual drugs in 1996.27 These categories also included 177 of the 200 most commonly prescribed drugs in 1998. These drugs represent a substantial portion of the total market for pharmaceuticals: the 200 most commonly prescribed drugs in 1998 made up 57 percent of the total prescriptions filled at retail pharmacies, and also 57 percent of the total dollar volume of prescriptions in 1998.28 Not all of these drugs were on the market in 1996; price data for that year were obtained for 166 drugs. Nineteen of the 20 drugs most frequently received by Medicare beneficiaries in 1996 are also included.29 We chose to use the most commonly prescribed drugs instead of those drugs with the highest dollar volume. This decision allowed us to focus our analysis on the drugs most used by consumers instead of the highest-cost drugs.
IMS provides data on price for each specific drug name, form (e.g., tablet or capsule), and strength (e.g., 500 milligrams) from each manufacturer. This study uses the most common form and strength in 1999 for each drug name and manufacturer, which is generally representative of the aggregate results across all forms and strength for a given drug.30
The next section presents results from both of these data sources that explore the question of whether individuals without prescription drug insurance coverage and individuals paying cash for prescription drugs pay more for the same drugs than insurers buying drugs on behalf of covered individuals. In general, we use the IMS Health data to compare directly the prices paid on a drug-by-drug basis, which cannot be done with the MEPS data. We use the MEPS data to compare prices paid by Medicare enrollees with and without coverage, and to aggregate across all drugs, neither of which can be done easily with the IMS data.
Unless otherwise noted, all results reported based on MEPS data are statistically significant (at the 0.05 level, based on a two-tailed test). The unique nature of the way IMS collects and reports its data does not allow for statistical testing of results from these audits. However, given the large sample sizes used by IMS (over 70 percent of US prescriptions filled at retail pharmacies), all results reported based on IMS data are highly likely to be statistically significant. See the Introduction of this report for details.
Price Differences from MEPS Data
MEPS data for 1996 show that people with prescription drug coverage received more costly drugs than people without coverage. However, for any specific drug, the total price paid for people with coverage tended to be lower than the price paid by people without coverage.31
This section gives considerable emphasis to analyzing brand name and generic drugs separately for several reasons. First, there are substantial price differences between brands and generics. Second, as mentioned earlier in the chapter, the general approach to pricing is quite different for the two categories of drugs. For example, the difference between a pharmacy's acquisition price and what it gets paid is greater in percentage terms for generics than for brand name drugs.
Both insured and uninsured consumers have incentives to use generic drugs. One widely held notion is that generic substitution may be more common for people with coverage, because insurers or PBMs often require it or charge higher copayments when enrollees receive a brand-name drug for which a generic equivalent is available. On the other hand, it is possible that use of generic drugs will be higher among uncovered individuals, as they have the most incentive to seek less expensive therapy. As shown in Figure 3-2, the MEPS data suggest that there are no real differences in percent of total prescriptions filled with a generic drug between covered and uncovered individuals.32
Figure 3-2. Percent of Prescriptions Filled with Generic Drugs by Coverage Status and Source of Insurance, 1996
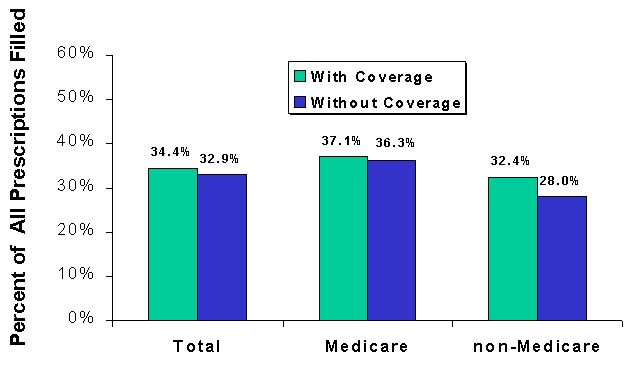
*Approximately 4 percent of drugs could not be classified as brand or generic.
Source: Center for Cost and Financing Studies, Agency for Healthcare Research and Quality: Medical Expenditure Panel Survey Household Component, 1996.
Table 3-3 presents results from MEPS on the average retail unit prices paid for brand name, generic, and all prescriptions in 1996, by people with and without prescription drug coverage.33 The retail unit price accounts for prescription size by dividing the price per prescription by the total number of units.34 It is important to take prescription size into account when comparing average drug prices because there may be differences in the quantity of drugs prescribed for people with different insurance coverage. For example, Chapter 2 noted that among hypertensive patients, those with drug coverage receive more pills per prescription than those without drug coverage.
The results in Table 3-3 show clearly the wide differences between the unit prices of brand name medications compared to generic drugs. For all categories of consumers, brand name drugs are priced at least three times higher on average than generic drugs. The results in this table can also be used to see whether individuals with or without coverage for drugs buy more expensive drugs. The first column of Table 3-3 shows that the average unit price per brand name prescription is $1.65 for people with coverage other than Medicaid, as compared to $1.54 for persons without coverage. The second column shows that, for generic drugs, the average unit price per prescription is $0.53 for people with coverage other than Medicaid, as compared to $0.43 for persons without coverage. For all drugs, the average unit price per prescription is $1.53 for people with drug coverage other than Medicaid, as compared to $1.16 for people without drug coverage.35 These price differences reflect the fact that covered individuals buy a more expensive mix of drugs.
| Coverage Status, Source of Insurance | Average retail unit price per prescription | ||
|---|---|---|---|
| Brand Name | Generic | All Drugs | |
| Total | $1.73 | $0.49 | $1.46 |
| Drug coverage other than Medicaid | 1.65 | 0.53 | 1.53 |
| Medicaid | 2.47 | 0.41 | 1.35 |
| Without drug coverage | 1.54 | 0.43 | 1.16 |
| Medicare | 1.76 | 0.5 | 1.32 |
| Drug coverage other than Medicaid | 1.65 | 0.53 | 1.32 |
| Medicaid | 3.1 | 0.51 | 1.7 |
| Without coverage | 1.31 | 0.38 | 0.97 |
| Non-Medicare | 1.71 | 0.48 | 1.55 |
| Drug coverage other than Medicaid | 1.65 | 0.53 | 1.65 |
| Medicaid | 2.04 | 0.35 | 1.11 |
| Without coverage | 1.83 | 0.52 | 1.45 |
|
Note: Prices shown are point of sale prices and do not include manufacturer rebates. Source: Center for Cost and Financing Studies, Agency for Healthcare Research and Quality: Medical Expenditure Panel Survey Household Component, 1996. |
|||
There exist a variety of other factors, such as form, strength, and mix, that must be taken into account - in addition to the quantity of drugs in a prescription - in order to produce meaningful comparisons of drug prices for those with and without coverage. There is some evidence that physicians may prescribe different, less expensive drugs for people without coverage.36 Table 3-4 shows an illustrative example of how two Medicare beneficiaries - one with drug coverage and one without drug coverage - with the same medical conditions and the same number of prescriptions can receive different quantities, forms and strengths of the same drugs, and a different mix of drugs for the same conditions. These drug differences translate into substantial differences in the average price per prescription (higher for the beneficiary with drug coverage) and average prices paid for the same drugs (higher for the uncovered beneficiary). Thus, to determine whether people with coverage pay less than people without coverage for the same drug, we need to make a more direct comparison - one that holds form, strength, and type of drug, as well as quantity, constant.
| Beneficiary A (has Rx Coverage) | Beneficiary B (no Rx Coverage) | ||||
|---|---|---|---|---|---|
| Drug | Dosage | Monthly Cost | Dosage | Monthly Cost | |
| Same Drugs | Inhaler | 2 puffs/day | $25 | same | $33 |
| Blood Thinner | 5mg, 3X day | $20 | same | $25 | |
| Pain Killer | 20 Tabs/250 mg each | $10 | 15Tabs/125mg each | $7 | |
| Different Drugs | Anti-Hypertensive A | 20 mg/day | $40 | -- | -- |
| AntiHypertensive B | -- | -- | 5mg/day (equivalent) | $25 | |
| Anti-Heartburn A | 10mg/day | $90 | -- | -- | |
| Anti-Heartburn B | -- | -- | 2mg/2X day (equivalent) | $50 | |
| Average Price Per Prescription | $37 | $28 | |||
| Average Price Per Rx for Same Three Drugs | $18 | $22 | |||
Consider two Medicare Beneficiaries, Beneficiary A and Beneficiary B, who suffer from the same conditions: high blood pressure, heartburn, chronic lower back pain, asthma, and atrial fibrillation - a condition which often requires the daily use of a blood thinner. This combination of illnesses is not unusual in the typical Medicare patient.
Both beneficiaries receive five prescriptions per month for their five conditions. However, Beneficiary A has drug coverage and Beneficiary B does not. As illustrated in the diagram above, some of the drugs Beneficiary A receives are the same as Beneficiary B and some are different. Beneficiary A receives discounts on the same drugs for which Beneficiary B pays full price, and pays more for the different drugs because people with coverage often receive newer drugs that are also more expensive. Lastly, in the case of the pain killer, Beneficiary A and B both receive the same drug but Beneficiary A, because he has drug coverage, receives a greater number of pills per month and a higher dosage strength. Thus, the price of the pain killer reflects the difference in total number of pills and any discount Beneficiary A may receive.
As shown, while the average price paid per prescription is higher for Beneficiary A, Beneficiary B still pays more for the same given drug. Moreover, in the case of the pain killer, he actually receives less of the same drug, and at a lower strength.
It should be noted that the prices listed, although used purely as examples, are realistic prices for these types of drugs.
The best way to make comparisons is to use data on the prices paid for a specific form and strength of a drug for different categories of consumers. Ideally, these data would include the effect of manufacturer rebates on the prices paid for those with third-party coverage. No available data set, however, includes these rebates. Although MEPS has insufficient sample size to allow estimates of price differences between covered and uncovered Medicare beneficiaries for any single drug, MEPS allows for the calculation of a powerful summary measure across drugs. To calculate this measure we compare the retail price for each drug to a benchmark price. This benchmark price is obtained from a private company.37
By comparing the retail price to a benchmark price, we can standardize prices across drugs. We calculate the ratio of the retail price to the benchmark price on a unit basis to control for differences in the dispensed quantity. A separate benchmark price is used for each form, strength, and package size (from which each drug was dispensed) of a drug. By calculating this measure separately for each prescription reported in MEPS, we obtain a measure of the relative amount over the benchmark price that the purchaser paid for that prescription.38 We call this measure the percent over benchmark price. Taking the average of this ratio across all drug events gives us a measure that accounts for different forms, strengths, and quantities of medications, and differences in the mix of drugs prescribed, thereby allowing for price comparisons (excluding the effect of rebates) among different subgroups, such as people with and without drug coverage across all of their drug purchases.
Table 3-5 shows the average and median percent over benchmark price for all drugs, by coverage status and source of insurance. Overall, using averages, people without drug coverage paid more when they used the same drugs (57.9 percent over the benchmark) than did people with drug coverage other than Medicaid (33.3 percent over the benchmark). This same result is found using the median percent over benchmark instead of the average, even though the numbers reported are considerably smaller (14.6 percent versus 0.0 percent). In general, these relationships hold up when examining the Medicare and non-Medicare populations separately. In this table, the percent over benchmark for Medicaid beneficiaries is generally closer to individuals without drug coverage than to individuals with coverage. This may be because the prices ultimately paid by Medicaid on behalf of patients are greatly reduced by rebates, which are not captured in these data, and because Medicaid generally pays higher dispensing fees than do PBMs and insurers.
The difference in the magnitude of the results when using medians versus averages suggests that the underlying distributions are skewed. One possible reason for such a skewed distribution is the different pricing patterns for brand name and generic drugs discussed above. In this situation, the median tends to offer a better overall measure of the relationship.
| Coverage Status and Source of Insurance | Average Retail Percent over Benchmark | Median Retail Percent over Benchmark |
|---|---|---|
| Total | 40.5% | 3.0% |
| Drug coverage other than Medicaid | 33.3% | 0.0% |
| Medicaid | 61.8% | 8.3% |
| Without drug coverage | 57.9% | 14.6% |
| Medicare | 40.8% | 3.7% |
| Drug coverage other than Medicaid | 31.3% | 0.1% |
| Medicaid | 58.6% | 7.8% |
| Without coverage | 61.4% | 10.1% |
| Non-Medicare | 40.3% | 2.6% |
| Drug coverage other than Medicaid | 34.5% | 0.0% |
| Medicaid | 63.9% | 9.1% |
| Without coverage | 52.7% | 19.2% |
|
*Percent over benchmark price equals the ratio of the average retail unit price (ARUP) to the benchmark unit price (BUP) minus one, multiplied by 100: Percent over benchmark = ((ARUP/BUP) - 1)*100 Note: Data exclude the effect of rebates for those with Medicaid or with drug coverage other than Medicaid. Source: Center for Cost and Financing Studies, Agency for Healthcare Research and Quality: Medical Expenditure Panel Survey Household Component, 1996. |
||
To test further the robustness of these results, we looked at brand name and generic drugs separately. Table 3-6 shows the median percent over benchmark price for brand name and generic drugs by coverage status and source of insurance. For brand name drugs, the median is 1.0 percent below the benchmark for those with drug coverage other than Medicaid and 10.0 percent above for those without drug coverage. This means that people pay more for brand name drugs, when using the same drugs, if they are not insured. The differences hold up for the Medicare population (-0.6 vs. 7.8 percent) and appear larger for the non-Medicare group (-1.2 vs. 18.6 percent).
| Coverage Status and Source of Insurance | Median Retail Percent over Benchmark | |
|---|---|---|
| Brand Name | Generic | |
| Total | -0.1% | 20.7% |
| Drug coverage other than Medicaid | -1.0% | 15.6% |
| Medicaid | 3.8% | 28.1% |
| Without drug coverage | 10.0% | 45.8% |
| Medicare | 0.2% | 21.6% |
| Drug coverage other than Medicaid | -0.6% | 15.5% |
| Medicaid | 4.3% | 29.3% |
| Without coverage | 7.8% | 44.5% |
| Non-Medicare | -0.3% | 19.5% |
| Drug coverage other than Medicaid | -1.2% | 15.2% |
| Medicaid | 3.7% | 26.0% |
| Without coverage | 18.6% | 49.0% |
|
*Percent over benchmark price equals the ratio of the average retail unit price (ARUP) to the benchmark unit price (BUP) minus one, multiplied by 100: Percent over benchmark = ((ARUP/BUP) - 1)*100 Note: Data exclude the effect of rebates for those with Medicaid or with drug coverage other than Medicaid. Source: Center for Cost and Financing Studies, Agency for Healthcare Research and Quality: Medical Expenditure Panel Survey Household Component, 1996. |
||
The absolute differences in the percent over the benchmark price for generics (about 20 percent for both Medicare and non-Medicare populations) are considerably higher than for the brand name drugs (about 0 percent) because the fixed cost of filling the prescription does not vary with the acquisition cost of the drug. This is consistent with the discussion earlier in the chapter that the fixed cost of filling a prescription - the dispensing fee - is a larger proportion of the total cost of lower generic priced drugs.
For generic drugs, the median percent over benchmark is 15.6 percent for those with drug coverage other than Medicaid and 45.8 percent for those without drug coverage. The differences are similar for the Medicare population and the non-Medicare group. The difference in medians reported for generic drugs, however, is not statistically significant for two of the coverage categories (total and Medicare). Because overall prices are lower, however, price differences tend to be less important to the consumer than for the more expensive brand name drugs. Overall, the results in this table appear consistent with those in Table 3-5, in that those without drug coverage appear to pay more above the benchmark price for their drugs. Further research is needed to understand the pricing of generic drugs.
Finally, Table 3-7 shows the median percent over benchmark price for all prescription drugs by duration of coverage over the year. For the total population, the median percent over benchmark for people who had coverage for only part of the year was 9.0 percent, which was higher than for those with full-year coverage (0.7 percent) and appears lower than for those who never had coverage (14.6 percent).39 This is not unexpected, because people with part-year coverage would have paid cash prices if they purchased drugs during their period without coverage. However, an important implication of this result is that the inclusion of persons with part-year coverage in the simple "covered" group used for the analyses in Tables 3-5 and 3-6 has the effect of understating differences that would be observed if the analyses accounted for duration of coverage. These results are similar to the results on drug spending by duration of coverage reported in Chapter 2.
| Duration of coverage in 1996 | Median Percent over Benchmark |
|---|---|
| Total | 3.0% |
| Always | 0.7% |
| Sometimes | 9.0% |
| Never | 14.6% |
| Medicare | 3.7% |
| Always | 0.2% |
| Sometimes | 0.5% |
| Never | 10.1% |
| Non-Medicare | 2.6% |
| Always | 0.8% |
| Sometimes | 11.9% |
| Never | 19.2% |
|
*Percent over benchmark price equals the ratio of the average retail unit (ARUP) price to the benchmark unit price (BUP) price minus one, multiplied by 100: Percent over benchmark = ((ARUP/BUP) - 1)*100 Note: Data exclude the effect of rebates. Source: Center for Cost and Financing Studies, Agency for Healthcare Research and Quality: Medical Expenditure Panel Survey Household Component, 1996. |
|
Price Differences from IMS Data
IMS data on retail prices for the most commonly filled prescription drugs for the total population show that cash payers usually paid higher prices for the same drug than people with third-party payment. The IMS price data for individual drugs are highly confidential and proprietary, and their use in this report is contingent on their not being directly displayed. Instead the report will show comparisons between prices paid by different purchasers, or prices at different points in time. Here and in the following results, the category of third-party payers does not include Medicaid and the category of cash payers includes people without coverage and people with indemnity coverage.40
Figure 3-3 shows the distribution of differences in average cash-purchaser prices to average third-party prices (excluding the effect of rebates) for nearly 200 of the most commonly prescribed drugs.41 The results reported are for December 1996 and September 1999 (the most recent period that price data were available from IMS). The figure shows a simple distribution of cash to third-party ratios for both years.42 As shown later in this chapter, this distribution is somewhat different when we separate brand name and generic drugs. For the most part, the pattern displayed here reflects patterns for brand name drugs, while the generic drugs are most represented in the tails (especially the right-hand tail) of the distribution.
Figure 3-3. Percent Difference Between Average Price for Cash Purchasers and Average Price for Third Parties (Without Rebates) in 1996 and 1999, for the Most Commonly Prescribed Drugs
Note: Data exclude the effect of rebates for those with third-party coverage.
Source: IMS Price Trak ReportTM, 1996 and 1999.
The median (or midpoint) of the 1996 distribution was 8.3 percent, meaning that for half of the drugs, cash payers paid at least 8.3 percent more than those with a third-party payment, and half the drugs had a cash to third party difference of less than 8.3 percent. For a large majority of drugs (132 of 166, or 80 percent), the price paid is higher for cash purchasers (i.e., a difference of greater than zero, as shown in the figure). By 1999, the distribution has shifted to the right, meaning that the gap between cash and third-party prices has increased substantially. The median ratio has grown to 14.6 percent and cash purchasers are paying more than third parties for virtually all of the drugs (167 of 177 drugs, or 94 percent). For 25 percent of the drugs, the price difference in 1999 was more than 20 percent.
The growth in the price differentials has had a particular impact on Medicare beneficiaries. Figure 3-4 shows cash-purchaser to third-party price differentials (excluding the effect of rebates) for 19 of the 20 drugs most frequently used by Medicare beneficiaries in 1996, as determined by the number of prescriptions reported in MCBS.43 In 1996, the cash price was higher than the third-party price for 12 of the 19 drugs.44 By 1999, the cash price was equal to or higher than the third-party price for all of the 19 drugs.45
Figure 3-4. Percent Difference between Average Price for Cash Purchasers and Average Price for Third Parties (Without Rebates), 1999, for Drugs Most Frequently Used by Medicare Beneficiaries in 1996
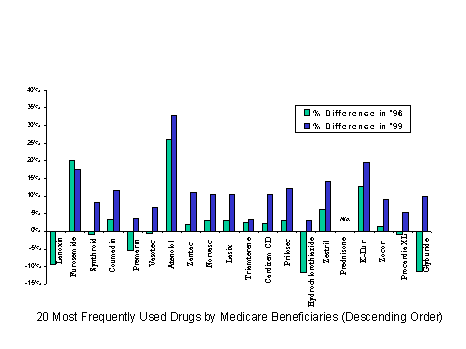
Note: Data exclude the effect of rebates for those with third-party coverage.
1999 difference for Lanoxin = 0%.
Source: IMS Price Trak ReportTM, 1996 and 1999. Drug rankings based on 1996 Medicare Current Beneficiary Survey
Prescription drugs exhibit a high level of price variability (the range in prices paid for a specific drug). All of the comparisons to this point have been based on average prices. Of course some cash purchasers pay more or less than the average, as do some third parties. In either case, however, the variability is considerable, and average prices are likely to be affected by extreme values. One would expect variation in prices to be somewhat greater for third parties than for cash purchasers, because not all insurers negotiate discounts on their own or through PBMs. This is true for the drugs in the IMS data: price variability was somewhat greater for third parties than for cash purchasers in 1999. The price variability percentage for third parties averaged 42.0 percent in 1999, as compared to 29.0 percent for cash payers.46
Median prices are less likely than average prices to be affected by extreme values. Figure 3-5 provides a comparison of the ratios of median cash prices to median third-party prices (excluding the effect of rebates) in 1999. When the medians are used for comparison, the previous results (based on average prices) are confirmed. There are somewhat more drugs for which cash purchasers pay less than third parties, 15 as opposed to 10 when using averages. There are 125 drugs, however, for which cash purchasers paid at least 10 percent more than third parties, and 13 for which cash purchasers paid over twice as much. The median of this distribution is actually somewhat higher at 17.3 percent than for the distribution based on averages (median of 14.6 percent).
Figure 3-5. Percent Difference between Median Price for Cash Purchasers and Median Price for Third Parties (Without Rebates), 1999, for the Most Commonly Prescribed Drugs

Note: Data exclude the effect of rebates for those with third-party coverage.
Source: IMS Price Trak ReportTM, 1999.
A final analysis focuses on the differences between brand name and generic drugs. Unit prices based on IMS data are four times higher for brand name drugs than for generic drugs, similar to the result reported earlier based on MEPS data (Table 3-3). As a result, it makes sense to reexamine the distribution of the differences in average price (excluding the effect of rebates) paid by cash purchaser versus third-party payer, shown in Figure 3-3, separately for brand name and generic drugs. For either category of drugs, cash customers pay higher prices than those with third-party payments at the point of sale (Figure 3-6). For the 111 brand name drugs analyzed, the median of the differences was 14.1 percent.47 The median of the differences was somewhat higher for the 66 generic drugs analyzed (22.1 percent).
Figure 3-6. Percent Difference Between Average Price for Cash Purchasers and Average Price for Third Parties (Without Rebates) in 1999, for the Most Commonly Prescribed Brand and Generic Drugs
Note: Data exclude the effect of rebates for those with third-party coverage.
Source: IMS Price Trak ReportTM, 1996 and 1999.
Figure 3-6 also shows that the differences in average price paid for brand name drugs are considerably more concentrated than for generic drugs. A large majority of the brand name drugs (75 of 111, or 68%) have a percent difference in price paid between 10 percent and 20 percent. In fact, no brand name drug in 1999 cost less for cash purchasers than it did for third-party payers (that is, all the price differences were greater than or equal to zero).
By contrast, the distribution for generic drugs is substantially less concentrated with only a slight peak in the zero to 10 percent range of the distribution. Many generic drugs are in the right-hand tail of the distribution with cash customers paying from 40 percent higher to nearly double that paid by customers with third-party coverage. But, as described previously, the smaller percentage increase on the more expensive brand name drugs amounts to a larger number in absolute dollars paid.
In sum, the analyses from MEPS and IMS tell a consistent story: people without drug coverage and other cash customers generally pay more than insurers for the same drugs at the point of sale. These price differences, however, are not the result simply of differing markups by the retail pharmacy; instead, they contrast prices charged by the pharmacy to one set of customers to the reimbursement received by the pharmacy (plus customer cost sharing) for another set of customers. The story is clearest for brand name drugs, where the absolute price difference matters the most to the cash-paying customer. The IMS data, which are the most recent available, suggest that disparities between prices paid by insurers and by individual consumers appear to have been growing in recent years.
As noted previously, a key limitation on the empirical analysis of prescription drug prices in this study is our inability to incorporate the effect of rebates. The net costs of insurers or PBMs are often further reduced by discount or rebate arrangements with pharmaceutical manufacturers that are not captured in these data. Rebate data, if available, would allow a more complete analysis of the overall differences between the amount paid on behalf of insured customers with the retail price paid by customers 103who lack coverage. Such differences would be larger than the retail price differences observed in this study.
Endnotes
1 - National Association of Chain Drug Stores, The Chain Pharmacy: Industry Profile, Alexandria, VA, 1999.
2 - In general most of the relationships in this table have been established with at least two different sources of data.
3 - IMS Health Retail Method-of-Payment ReportTM 1999.
4 - Cash customers may also include customers with prescription drug coverage who decide to pay full price for a prescription that would cost less than their copay amount or pay full price for a prescription not covered by their insurance.
5 - There are cases in which the holder of a patent licenses other manufacturers to make or market the drug; in this case there can be several brand names and prices for a single drug that is not yet available in generic form.
6 - In establishing upper payment limits for state Medicaid programs, HCFA(now known as CMS) assumes that AWP overstates actual acquisition costs by 10 to 20 percent. (State Medicaid Manual, sec. 6305.1)
7 - A recent study by Mathematica Policy Research reviews the variety of measures used by PBMs to manage utilization of prescription drugs. Anna Cook, Thomas Kornfeld, and Marsha Gold, The Role of PBMs in Managing Drug Costs: Implications for a Medicare Drug Benefit, Mathematica Policy Research, prepared for the Henry J. Kaiser Family Foundation, January 2000.
8 - Although the dispensing fee was originally intended to cover the professional services of the pharmacist, recent information suggests that it is not longer sufficient to cover this cost. Rather it is simply an additional fixed amount that pharmacies collect for each prescription dispensed (Wyeth-Ayerst Prescription Drug Benefit Cost and Plan Design Survey Report, 1999 edition).
9 - The literature cited in Appendix A suggests that pharmacy margins have been falling in recent years.
10 - A pharmacy can usually substitute a generic equivalent for a brand-name drug on its own, although some state laws restrict this. However, the prescribing physician's permission is always needed to substitute one brand-name drug for another.
11 - Most of the remaining savings were attributed to maximum allowable cost limits for drugs with generic equivalents and to preauthorization and utilization review. U.S. General Accounting Office, Pharmacy Benefit Managers: FEHBP Plans Satisfied with Savings and Services, but Retail Pharmacies Have Concerns, Washington, Feb. 1997 [GAO/HEHS-97-47].
12 - HCFA(now known as CMS)'s Office of the Actuary, in estimating net 1997 private insurer drug expenditures for the National Health Expenditure series, assumes average rebates from manufacturers in the range of 7 percent.
13 - Peter D. Fox, Pharmacy Benefits Management-101. Paper presented at The National Health Policy Forum, October 27, 1999.
14 - Letter from Inspector General June Gibbs Brown to Representative Henry Waxman, November 22, 1999.
15 - In some instances the VA has separately negotiated a contractual price that is lower than the FSS established for other federal purchasers.
16 - U.S. General Accounting Office, Drug Prices: Effects of Opening Federal Supply Schedule for Pharmaceuticals Are Uncertain, Washington, June 1997 [GAO/HEHS-97-60].
17 - This "big four" price is not available to other agencies using the FSS.
18 - An innovator multiple-source drug is the version of a drug, for which competition exists, that is sold by the manufacturer that originally obtained FDA approval for the drug.
19 - This limit does not apply when a physician specifies that a brand-name version of the drug is to be dispensed.
20 - Congressional Budget Office, How the Medicaid Rebate on Prescription Drugs Affects Pricing in the Pharmaceutical Industry (January 1996).
21 - This exclusion was not originally part of OBRA 1990; the best price could be the price charged to the VA and other federal facilities. Some manufacturers raised their price to the VA in order to reduce their required Medicaid rebates. In response, Congress added the exclusion and set separate pricing rules for federal purchasers.
22 - Because of these differences in methodology, the findings of this report on price variation at the retail pharmacy level should not be interpreted as inconsistent with findings of the Minority Staff of the House Committee on Government Reform on price variations that were based on differences between observed cash prices in retail pharmacies and prices published in the federal supply schedule.
23 - When statistically matching a MEPS pharmacy drug event to a household drug event between different persons, it was not possible to require that potential payment sources for the two people be exactly matched. This created the possibility that the price of a specific drug paid by a covered person could be statistically matched to a drug mentioned by another person who might not have had drug coverage, or vice versa. The decision to restrict the sample to only matches that were between drugs for the same person, and to eliminate drugs with any price imputations to fill in missing data, or replace outliers in the pharmacy component, was to reduce the potential bias from these measurement error sources.
24 - When all the MEPS data are used-- including both imputed and non-imputed prices-- significant differences remain between the prices paid by the covered and noncovered groups. The full MEPS data therefore demonstrate price differences using a nationally representative sample.
25 - Preliminary efforts using MEPS data were made to estimate price differences for groups of drugs most frequently used by Medicare beneficiaries. Although this method required merging together drugs from different manufacturers or with different forms and strengths with potentially different price differentials, the results were generally consistent with the IMS data for individual drugs in the same groups.
26 - MEPS would treat the same individual as covered. Thus the cash/third-party distinction and the covered/noncovered distinction in the IMS and MEPS results (respectively) cannot be taken as identical. If some individuals with indemnity plans pay the cash price at the point of service, the price differences for people with and without coverage would appear smaller than the price differences faced by cash payers and people whose insurance pays the pharmacy directly.
27 - The USC, or Uniform System of Classification, is a coding system that assigns drugs to broad and then narrower categories, using increasing numbers of digits as the categories become more specific. The 39 categories identified for this study are at the USC3 (3-digit) level: for example, they included 31100, antihypertensives, and 52100, sex hormones.
28 - IMS Health
29 - Prednisone, the 16th most commonly prescribed drug for Medicare beneficiaries was not in the original list of USC categories for which data were obtained from IMS.
30 - In order to assure that cash/third-party price differentials for the most common form and strength were representative of the differentials for other forms and strengths of the same drug, the results for the top 50 drugs were compared to average results for all forms and strengths (weighted either by dollar volume or by number of prescriptions). For the vast majority of the drugs, the results were very similar.
31 - As noted previously, for all analyses in this chapter, the price of a prescription drug is the total amount paid by all payers to the pharmacy that filled the prescription. For example, if the pharmacy received a $15.00 payment from a third-party payer and a $5.00 copayment from the consumer, the price of the drug is $20.00.
32 - A different result was reported in Chapter 2. With regard to hypertension drugs, those with coverage were more likely to use generics. Further research is needed to understand how the use of generics varies by insurance coverage and specific health conditions.
33 - Since the MEPS data are composed of every "drug event" (individual prescription) for the responding individuals, these averages are, in effect, weighted by the volume of prescriptions (i.e., drugs that are more commonly used will be more heavily represented in the average). A different approach would have weighted the averages by drug expenditures. For the tables that show brand name and generic drugs combined, this would have given more importance to brand name drugs. It would have given more weight to extremely high cost drugs and thus would have been less reflective of typical drug use by consumers. The Medicare category in this and other MEPS tables in this chapter includes a small number of elderly individuals--only 10 survey respondents--who did not report having Medicare.
34 - For most drugs, a "unit" is a pill. However, with liquid drugs and inhalers, the unit is the measured and metered dose, respectively.
35 - The comparisons between unit prices for covered and uncovered individuals are not statistically significant for brand name and all drugs. The comparable results for generic drugs are significant on a one-tail test only.
36 - Mott, D.A., Kreling, D.H., "The Association of Insurance Type with Costs of Dispensed Drugs," Inquiry 35: 23-35, Spring 1998.
37 - The private company reports the most common wholesale price charged to the retailer, which we use as our benchmark. It determines this price by a variety of methods. If a manufacturer has a suggested AWP, this value is used as the initial entry of this price. If a manufacturer does not provide a suggested AWP, then a statistical review of the actual wholesale price is conducted, and the price used is the mode (most common) price obtained from the wholesalers surveyed. When no clear mode price is evident, a statistical mean price is computed. The number is also indexed over time through a proprietary model designed by the company. It is important to note that this methodology produces a value different than the AWP (list) price described earlier in the chapter.
38 - This ratio is computed separately for each filled prescription, and then averaged across all drug events. The figure shown is the average of the ratios, not the ratio of the average of all retail prices to the average of all benchmark prices. Thus the differential between the covered and non-covered groups may be interpreted as the difference in percent over benchmark price on the average drug purchase.
39 - In the total and non-Medicare categories, the difference between part-year coverage and no coverage is not statistically significant. In the Medicare category, only the difference between full-year and part-year coverage is not statistically significant.
40 - IMS retail pharmacy data classify indemnity insurance arrangements - in which a customer pays cash for a prescription and then submits a claim to an insurer for reimbursement - as cash payments. Although this type of insurance arrangement has been declining in recent years, the lack of data on these reimbursements means that analyses that use the IMS cash pay group as a proxy for people without prescription drug coverage will incorrectly state the true difference in prices paid between people with and without coverage. However, including indemnity insurance in the cash pay group is not entirely inappropriate. Persons with indemnity insurance bear a higher portion of drug prices directly, relative to other insured groups, because indemnity style insurance more often entails a dollar cap on prescription drug coverage and co-insurance (payment of a percentage of the price) unlike the flat co-pays common with most types of prescription drug coverage.
41 - As discussed previously, to develop the list of the 200 most commonly purchased drugs we used the most recent full calendar year that IMS data were available, 1998. Results do not include 23 of these drugs for 1999 and 34 of these drugs for 1996, because data for these drugs were not included in the USC3 groupings that were obtained from IMS or the drugs were not on the market in 1996.
42 - These results and all those presented subsequently are based on unweighted distributions across the most commonly prescribed drugs. The distributions count each of the top 200 drugs equally, rather than (for example) placing greater weight on higher volume drugs.
43 - The drug ranked 16, Prednisone, was not in the original list of USC categories for which data were obtained from IMS. In a separate IMS data set covering 1998, Prednisone had a 32 percent difference in prices between cash and third-party payers. MCBS, rather than MEPS, rankings were used because of the larger MCBS sample of Medicare beneficiaries. However, 8 of the top 10 MCBS drugs would also have been among the top 10 MEPS drugs.
44 - That the cash price for the most commonly used drugs - especially generic drugs - could be lower than the third-party price is not surprising. These are the drugs that retailers are most likely to discount for cash customers in order to attract their business; in effect, they are the "loss leaders."
45 - During this time period, the proportion of beneficiaries in Medicare risk plans increased substantially (in 1996, 11 percent of Medicare beneficiaries were in risk plans; by 1999, this proportion had increased to 16 percent), and these beneficiaries would be grouped in the IMS third-party payment group.
46 - This percentage is the interquartile range (the 75th percentile value minus the 25th percentile value) divided by the median. The figures cited are the unweighted average for the 166 of the 200 most prescribed drugs for which the data needed for the calculation were available.
47 - When brand name drugs are further separated into the categories of "with generic competition" and "without generic competition," the drugs without competition have a fairly higher median cash and third-party unit price compared to the drugs with competition. However, the difference between the median of differences between drugs with competition and those without was not substantial. Moreover, a distribution of the 111 brand name drugs separated into the "with" and without competition" categories did not differ greatly from the general distribution of all brand name drugs.
Future Research
The results presented in this study provide a wealth of information on prescription drug use in the United States. This study used recent data from multiple surveys, private sector pricing data, and outreach meetings with industry representatives and researchers to provide a detailed examination of multiple factors relating to coverage, utilization, and spending for prescription drugs, particularly by the Medicare population.
The descriptive results presented here raise a variety of issues that are ripe for further investigation. The study has revealed suggestive relationships between demographic factors, insurance status, and prescription drug use; however, we were unable to examine the interrelationships among these factors. Future multivariate analyses will allow us to come to a more nuanced understanding of these relationships. In addition, future research will explore what can be learned from using more sophisticated definitions of drug coverage status and severity of illness than were available for this study. Also, ongoing analyses will allow us to continue to use the most recent data. Rapid change in the pharmaceutical market requires that analyses be refreshed and updated on a continuing basis.
Some possible avenues for future research based on the results of this study include the following:
- What are the characteristics of Medicare beneficiaries with and without drug coverage? Despite the wealth of descriptive material we assembled on this issue, lack of a multivariate analysis hinders our interpretation of the reason why some beneficiaries maintain coverage and others do not. A particularly important question yet to be answered is whether beneficiaries with the greatest need for coverage find it easy or difficult to obtain it. An equally important set of questions includes how stable prescription drug coverage is for those who have it, for which people coverage is least stable, and the consequences of unstable coverage.
- What factors explain drug spending by Medicare beneficiaries? Our study demonstrated that covered individuals use more drugs than those without drug coverage, but we were unable to provide a causal explanation for this difference. Further work is needed to disentangle the effects of barriers to access, adverse selection, and moral hazard in determining the spending patterns of covered and uncovered beneficiaries.
- Can the appropriate use of prescription drugs reduce the need for other health care services like hospitalization or nursing home care? While there have been some studies suggesting that medication can reduce the need for hospital care in the case of specific medical conditions, there is little consensus among researchers on this issue. Most analyses of health care spending tend to look at expenditures for prescription drugs in isolation from other kinds of health care spending and utilization. Our study did not address this question. More research is needed in order to understand the way in which access to drugs affects overall health care spending and the quality of medical care.
- Does drug coverage influence the type of medications used? Do beneficiaries face barriers to access to breakthrough therapies because of their lack of drug coverage? Our preliminary findings indicate that there appears to be a difference in the mix of drugs used by covered and uncovered beneficiaries. We are, as yet, unable to explain that difference or evaluate its significance. Further research could explore this issue through an analysis that includes measures of severity of illness for particular medical conditions, and focuses on specific therapeutic classes of drugs. Simple tabulations, for example, suggest that different types of hypertension drugs are used by those with and without coverage. But a more thorough analysis needs to consider various factors (e.g., severity, coexisting conditions, race) that affect choice of therapy.
- Have changes in the pharmaceutical industry and the organization of prescription drug benefits affected the price of drugs and drug utilization for covered and uncovered beneficiaries? Our analysis of price information and discussions with industry representatives have left us with many questions about how the industry is structured and the dynamics of the pharmaceutical marketplace. Additionally, ongoing trends like direct-to-consumer advertising or the continual emergence of important new "breakthrough" drug therapies have been outside the scope of our study. The complex set of interactions that connect drug manufacturers to patients will require considerably more analysis to learn whether these trends will exacerbate the differences seen in this report for covered and uncovered beneficiaries. In addition, if more data were available on elements of manufacturer pricing, such as rebates, further research could probe more fully the differences in prices paid by different customers.
- How have the techniques developed by PBMs like drug utilization review (DUR) and formulary management affected the quality of pharmaceutical care received by individuals? Our study draws attention to the way in which PBM management of drug benefits has affected the pricing structure of the industry. Further research is needed to determine whether these management strategies have had an impact on access by beneficiaries to appropriate medications and or on the number of adverse drug reactions in the population. Among the questions to be answered include: How much variation exists among PBMs with regard to mechanisms used to screen prescriptions for drug interactions and other safety issues? How are DUR systems updated to incorporate safety information for newly approved drugs and the experience with those drugs among the larger patient population? Is the needed data available to permit timely and rigorous comparisons of competing drug therapies? What effect will emerging strategies such as prior authorization have on access to needed drugs?
- What is the pattern of drug utilization and spending by disabled beneficiaries and beneficiaries in nursing homes and other institutions? Our study, like most previous research, focused only on Medicare beneficiaries living within the community. Yet beneficiaries who are nursing home residents are heavy utilizers of prescription drugs. In addition, while beneficiaries under age 65 were included in this report, they clearly have different utilization patterns that we were not able to pursue here. The prescription drug needs of these populations are largely unexplored. For example, there is a much higher prevalence of mental illness among the disabled compared to the elderly, making behavioral medicine a critical issue for this population group. The disabled also do not have the same access to private Medigap supplements as those over 65 years of age. Further work is needed to explore the utilization and spending patterns of these populations.
- What explains geographical variations in prescription drug utilization and prices? Our analysis revealed striking regional variations in drug prices faced by beneficiaries and in utilization of drugs. Further work is needed to understand what factors are driving these variations and what consequences they have for beneficiaries.
- How are changes in prescription drug coverage affecting utilization and prices? Our analysis has observed a dramatic shift in drug coverage over the past decade from indemnity coverage to third-party coverage at the point of sale managed by pharmacy benefit managers. A better understanding is needed of the effects of this shift, especially on those lacking drug coverage.
- How do Medicare+Choice plans manage their prescription drug benefits? Can their experiences help us to better understand drug utilization by the elderly and improve the quality of care they receive? The Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)) (HCFA(now known as CMS)) has initiated research to study the way in which Medicare+Choice plans currently administer their drug benefits. The aim of the project is to uncover best management practices with regard to improving outcomes, avoiding drug interactions, encouraging appropriate prescribing patterns, and educating beneficiaries about the medications they take.
- How prevalent is underinsurance by Medicare beneficiaries for prescription drugs? The Agency for Healthcare Research and Quality (AHRQ) has begun preliminary work to investigate the Medicare population's prescription drug coverage. They will look at the way in which different sources of coverage meet or fail to meet the needs of Medicare beneficiaries for access to needed medications.
Appendix A. Issues in Prescription Drug Coverage, Pricing, Utilization, and Spending: What We Know and Need to Know
Report prepared for
US Department of Health and Human Services
Office of the Assistant Secretary for Policy and Evaluation
Office of Health Policy
February 18, 2000
by
Bruce Stuart, Ph.D.
Nicole Brandt, PharmD
Becky Briesacher, MA
Cheryl Fahlman, BSP, MBA
Daniel Mullins, Ph.D.
Frank Palumbo, J.D., Ph.D.
Josefina Pizarro, BA
Lynne Stuart, MS
Peter Lamy Center for Drug Therapy and Aging
University of Maryland School of Pharmacy
506 W. Fayette Street, Suite 101, Baltimore, MD 21201
(410) 706-2434
The views expressed in this report are those of the authors and do not necessarily reflect those of OSASPE or the Office of Health Policy.
Section 1. Introduction
This report summarizes the findings from an intensive review of the literature addressing important policy issues in prescription drug coverage, pricing, utilization, and spending. The review had three primary aims:
- To assess the current literature concerning prescription drug costs and trends for Medicare beneficiaries with and without prescription coverage,
- To assess the recent literature on international prices and spending for prescription drugs, and
- To summarize the findings of these reviews from the perspective of what is known and what needs to be known in order to inform the policy debate regarding a Medicare drug benefit.
The study employed a detailed literature search strategy designed to assure that all important research findings relating to the study aims were captured. This strategy involved: (1) traditional index searches of literature in the medical sciences, social and behavioral sciences, economics, health services research, and public policy arenas, (2) searches for relevant statistical data in domestic and international documents, (3) identification and review of relevant ongoing research efforts funded by government agencies and private foundations, (4) identification of relevant working papers and presentations at professional associations based on ongoing and recently completed studies, and (5) a detailed web search with search terms keyed to the study aims. The search was restricted to materials published in the English language since 1995 except for seminal studies published prior to 1995, which are also included.
The report is divided into six sections. Following this introduction is a section that addresses key issues relating to drug costs for Medicare beneficiaries. Seven topic areas are covered in depth:
- Drug cost trends
- Drug prices faced by the elderly
- Sources and determinants of prescription coverage
- Dynamic patterns in prescription coverage
- Impact of prescription coverage on drug use and cost
- Out-of-pocket prescription drug spending by Medicare beneficiaries
- What we know and need to know
The next two sections provide background and context for the Medicare drug benefit debate. Section 3 provides an overview of the market for prescription drugs in the United States including a description of the drug distribution chain and methods for measuring drug prices. Section 4 describes international drug prices and spending. The primary focus of this section is on what can be learned from the international experience particularly in the area of drug price controls. Section 5 is a bibliography of all studies examined in the course of the literature review. Section 6 contains of abstracts of studies considered to be of particular value.
Section 2. Prescription Drug Costs for Medicare Beneficiaries
Spending on outpatient prescription drugs by non-institutionalized Medicare beneficiaries reached $25.1 billion in 19961, the latest year for which accurate statistics are available (Poisal, et al, forthcoming). Four years earlier in 1992, beneficiaries spent $16.2 billion (Laschober and Olin, 1996). Not only does this represent a substantial growth in spending (55 percent), but the rate of increase is also rising, from approximately 9 percent annually between 1992 and 1994 to over 14 percent annually between 1994 and 1996 (Laschober, 1997; Olin and Liu, 1998; Poisal, et al., forthcoming). In per capita terms, the growth rate over this period (44 percent) was somewhat lower (rising from $468 to $674), reflecting the fact that the size of the Medicare population has also continued to grow.
More current data are available on prescription drug costs for beneficiaries enrolled in Medicare HMOs. According to industry surveys, the average per member per month (PMPM) expenditure on prescription drugs rose from $37.53 in 1995 to $52.50 in 1998, a 40 percent increase in just three years (CibaGeneva, 1996; Novartis, 1999). The increase would have been even steeper had HMOs not raised patient copays by more than 25 percent over the same period (CibaGeneva, 1996; Novartis, 1999).
The issue of rising drug costs is at the core of the national debate over whether a prescription drug benefit should be added to Medicare coverage. On the one hand, rising costs are cited as a main reason why Medicare should cover drugs. Despite the fact that drug coverage of Medicare beneficiaries expanded during the first half of the 1990s (see section 2.3); the burden of out-of-pocket drug spending has also grown (see section 2.5). On the other hand, the growth in drug spending will make any meaningful Medicare drug benefit expensive to finance. From either perspective, there is a clear need to answer the question: What factors explain drug spending by Medicare beneficiaries?
The answers lie at both the individual level and the market level. The literature on individual determinants of drug spending by Medicare beneficiaries identifies seven factors: (1) health status, (2) functional impairment, (3) age, (4) gender, (5) race, (6) income, and (7) insurance coverage. Health status is an obvious factor. Beneficiaries reporting excellent health spent an average of $363 on prescription medicines in 1996 compared to $1,107 for persons reporting poor health (Poisal, et al., forthcoming). For beneficiaries with no functional impairment, drug spending per person was $582 versus $1,000 for persons with one or more functional impairments (Poisal, et al., forthcoming). Other highlights from the latest report of Medicare beneficiary drug spending (Poisal, et al., forthcoming) include the following:
- Gender: drug spending by females is higher than for males
- Race: whites spend more than blacks
- Disability: disabled beneficiaries under age 65 spend almost double the amount of aged beneficiaries
- Income: Beneficiaries with annual incomes below the poverty line have the highest per capita spending on prescription drugs, closely followed by those with incomes between 150 and 200 percent of the poverty line.
- Insurance Status: More than three-quarters (79%) of total 1996 prescription drug expenditures by Medicare beneficiaries were spent by persons with drug coverage. The average per capita drug spending by beneficiaries with coverage was $769 and without coverage was $463.
The finding that spending levels differ by socio-demographic status and insurance coverage are not unexpected, but simple descriptive comparisons may mask the true determinants of drug spending behavior. In one of the very few multivariate studies of prescription spending by aged Medicare beneficiaries, Lillard, et al. (1999) find that private health insurance, urban residence, and ill health are positive predictors of drug spending, while age and education level are negative predictors, when other factors are equal. Some factors that appear to be strong correlates of drug spending in descriptive comparisons lack predictive power in a multivariate framework, including gender, income, wealth, and Medicaid coverage.
2.1 Drug Prices Faced by the Elderly
The determinants of drug spending described above operate primarily through the drug selection decisions made by Medicare beneficiaries and their physicians. However, drug prices can be an important factor, particularly for those beneficiaries without stable drug coverage. The question most frequently asked in this regard is: Do Medicare beneficiaries pay more for their prescriptions than other Americans ? The underlying issue is whether the prices for drugs commonly used to treat age-related chronic conditions are either higher or increasing at a faster rate than drugs used for other diseases. Research on this question is difficult because drug prices vary greatly depending on the payor, and private payors rarely disclose their pricing practices. For this reason, most attempts to compare prices across age and disease groups are based on standardized price listings such as the Average Wholesale Prices (AWP), the Producer Price Index (PPI), or the average retail price (ARP). Highlights from three recent studies show that:
- Of the five best-selling drugs used by older Americans in 1997, the average retail pharmacy price (undiscounted) ranged from 78 percent to 299 percent higher than the “best price” negotiated by the Department of Veterans Affairs. (US House of Representatives, 1999).
- The AWP for the 50 prescription drugs most frequently used by the elderly rose more than four times the rate of inflation during calendar year 1998. Over the past five years (1994-1999), the AWP prices of these same drugs rose at twice the level of inflation. During the same period, 22 of the 50 most commonly used drugs by seniors had generic or co-marketed versions available for some portion of the time (Families USA, 1999).
- Between 1990 and 1996, changes in the Producer Price Index (PPI) for drugs in therapeutic classes most frequently prescribed to elders were 51 percent for anticonvulsants, 39 percent for cancer products, 42 percent for cardiovascular agents, 38 percent for diabetes products, 45 percent for diuretics, and 53 percent for nutrients and supplements (Berndt, et al., 1998). Despite these high inflation rates, the authors found no evidence of age-related price inflation differentials at the producer level.
These studies do not resolve the issue of whether age-related price discrimination exists in the market for drug products because of what happens to prices at different points in the chain of distribution. For example, Berndt et al., (1998) found no systematic age-related difference in price increases at the juncture between manufacturer and wholesaler for three classes of drugs (antibiotics, antidepressants, and calcium channel blockers), but from the wholesaler to the pharmacy one of the three classes (antibiotics) showed more rapid growth for the elderly than the young. Furthermore, at the point where the patient makes the purchase in the pharmacy, relative age-related prices for antidepressants favored the young rather than the aged (Berndt, et al., 1998).
2.2 Sources and Determinants of Prescription Coverage
Medicare pays for prescription drugs only if they are administered in institutional settings (e.g., hospital or nursing home) or belong to several special drug categories such as immunosuppressives, erythropoiten, oral anti-cancer drugs, hemophilia clotting factors, and some vaccines. The vast majority of outpatient prescription drugs are not reimbursable under the standard package of Medicare benefits. Supplemental insurance thus plays an important role in understanding how much Medicare beneficiaries spend on prescription medications. What are the characteristics of Medicare beneficiaries with and without drug coverage?
More than 90 percent of Medicare beneficiaries have some form of supplemental health insurance coverage, either by qualifying for public assistance programs or by enrolling in private insurance policies through employee benefits, retiree plans or individually purchased plans. There is no centralized database that records the characteristics of these supplemental policies or of the beneficiaries who obtain them. Instead, our information on coverage rates depends almost entirely on survey data. The best nationally representative survey (and the one used by most researchers in this area) is the Medicare Current Beneficiary Survey (MCBS). Since 1992, longitudinal samples of approximately 12,000 aged and disabled beneficiaries have been queried three times a year about their supplemental insurance (including prescription drug coverage), health status, access to care, and use and cost of all health services. The scientifically rigorous methods used in selecting the respondents and soliciting accurate information make MCBS data the most reliable single source for estimating prescription drug coverage for the Medicare population.
According to 1995 MCBS data, 65.2 percent of community dwelling Medicare beneficiaries had some prescription drug coverage for at least part of the year (Poisal, et al., 1999). Over 70 percent of the disabled Medicare population had drug coverage in that year. Rates were lower among elderly beneficiaries, ranging from 66 percent of those aged 65-69 to just 60 percent of those over age 85 (Poisal, et al., 1999). Males and females have similar coverage rates (70 and 68 percent, respectively in 1995). White beneficiaries were less likely to have coverage (64 percent) than blacks (69 percent) or persons of other races (75 percent). Coverage rates rise with income from 65 percent among beneficiaries with annual incomes below $10,000 to 72 percent among beneficiaries with incomes above $30,000 per year (Poisal, et al., 1999). Beneficiaries who reside in metropolitan areas are much more likely to have prescription coverage (69 percent) compared to those who do not (54 percent) (Poisal et al., 1999). In 1995, 64 percent of beneficiaries reporting excellent health had drug coverage compared to 69 percent of those in poor health (Poisal, et al., 1999). The differences are much greater when health status is measured by counts of chronic conditions. In 1996, 65 percent of Medicare beneficiaries reporting having none of ten common chronic conditions had some prescription coverage compared to 77 percent of beneficiaries with five or more of these conditions (Stuart, et al., 2000).
The reasons for these differences are varied and complex. For beneficiaries above the poverty line, coverage rates rise with income. This could be due to higher purchasing power and the fact that retirees with higher incomes are more likely to qualify for employer-sponsored health insurance. The reason that prescription coverage is higher among nonwhites is probably not because of race per se, but rather because nonwhite retirees have lower incomes and therefore higher Medicaid eligibility rates. The negative correlation of coverage rates and self-reported health status might mean that those who need coverage the most are the most likely to seek it out, but it could also capture the impact of income and Medicaid eligibility status.
To date there has been just a single study of the determinants of prescription drug coverage for Medicare beneficiaries (Lillard, et al., 1997). It showed no evidence of adverse selection into prescription coverage, but the data for this study are quite old (1968-1990). There is a substantial body of research on selection into Medicare supplemental policies independent of benefit coverage. Recent studies of Medicare HMO enrollment (Hellinger, 1995; PPRC, 1996; Riley, 1996; Hamilton, 1999; Call, et al) find strong evidence of favorable rather than adverse selection. Indications of adverse selection in the Medigap market have been found by Ettner, (1997), Wolfe and Goddeeris (1991), and Atherly (1999). Other researchers (Cartwright, Hu, and Huang, 1992; Hurd and McGarry, 1997; Lillard, et al., 1999) find none. Most researchers assume that there is no selection into retiree plans given the nature of plan sponsorship, but Atherly (1999) finds evidence of adverse selection in this market. These thoroughly mixed findings do little to clarify the issue of possible adverse selection into prescription coverage.
2.3 Dynamic Patterns in Prescription Coverage
Although the determinants of who has prescription benefits remain unclear, there is no question that drug coverage rates among Medicare beneficiaries have been rising. In 1992, 57 percent of beneficiaries had prescription coverage (Poisal, et al., 1999). By 1996 the rate was 68.8 percent (Poisal, et al., forthcoming). The movement of Medicare beneficiaries into HMO plans is one reason for the trend. Enrollment into Medicare risk plans increased 30 percent between 1995 and 1996 and HMO enrollees are more likely to have prescription coverage (94.6 percent) than those in any other private supplemental insurance plan. However, these prescription benefits are often inadequate.
From a policy standpoint the critical unanswered question is: Will the trends in prescription coverage of Medicare beneficiaries witnessed in the early and mid 1990s continue? There are no reliable data on prescription coverage rates since 1996. However, it appears unlikely that the trend maintained its steep upward course in the late 1990s. Recent reports show that large employers are scaling back retiree benefits (Hewitt Associates, 1999) that HMOs are not renewing Medicare risk contracts (Barents Group, 1999), and that some Medicare risk plans are dropping drug benefits, lowering payment caps, or imposing stiff premium surcharges for such coverage (Gold, 1999). This is such a volatile mix of factors, that any forecast of beneficiary drug coverage rates is fraught with uncertainty in both short-term and long-term projections.
This uncertainty has its roots in dynamic patterns of coverage that became evident in the mid 1990s. Profiles of typical Medicare supplemental insurance portfolios present a dynamic picture of beneficiaries gaining and losing prescription coverage, holding multiple plans, and changing plans during the year. Poisal et al., (forthcoming) found that nearly nine percent (over 3 million people) of the total non-institutionalized Medicare population switched plans at least once during 1996. This figure is slightly higher than the previous year’s estimate of eight percent (Davis, et al., 1999). However, both of these estimates understate the true degree of change. A month-to-month examination of supplemental insurance in 1996 by Stuart et al. (2000) shows that prescription coverage is often fragmented and non-continuous. They found that 18.9 percent (about 6.3 million people) of beneficiaries with full-year Medicare enrollment had drug benefits for only part of the year, averaging around six months of coverage. Having coverage at some point in the year may be a valuable benefit, but the continuity of that coverage is equally important.
2.4 Impact of Prescription Coverage on Drug Use and Cost
Surely the most compelling issue for policy makers regarding a possible Medicare drug benefit is the question of cost. For any given proposal, actuaries can readily determine that part of the program cost which represents a shift in financial responsibility from beneficiaries or other third party payors. It is much more difficult to predict the impact that coverage will have on the utilization patterns of previously uninsured beneficiaries. This issue has two important components: Does drug coverage encourage Medicare beneficiaries to use more outpatient prescription medications? Does drug coverage influence the type and cost of the medications prescribed and used? The available literature on these topics is primarily descriptive, representing cross-tabulations of utilization and expenditure by presence and source of coverage. Taken at face value, the reported differences suggest that insurance plays a very significant role in the drug utilization decisions of Medicare beneficiaries and their prescribers. A sampling of these findings tells the story:
- Beneficiaries with drug coverage filled an average of 20.3 prescriptions in 1995 compared to 15.3 for beneficiaries without coverage (Davis et al, 1999).
- Beneficiaries enrolled throughout 1996 with full-year prescription coverage filled an average of 22.4 prescriptions compared to 20.7 for those with part year coverage and 16.7 for those with no coverage (Stuart, et al., 2000).
- Average spending on outpatient prescriptions by covered individuals was 66 percent higher in 1996 compared to non-covered beneficiaries (Poisal, et al., forthcoming).
The differences in utilization and annual drug spending by insurance status extend to all major population subgroups categorized by age, income, and health status:
- In 1996, disabled Medicare beneficiaries under age 44 who had drug coverage spent 300 percent more on prescriptions than their counterparts without coverage, $1,077 to $268. (Poisal, et al., forthcoming).
- Among the oldest of the elderly (aged 85 plus), those with drug coverage spent 60 percent more on prescriptions than those without coverage (Poisal, et al., forthcoming).
- Beneficiaries with annual incomes in 1996 below the poverty line and no drug coverage filled only 13.8 prescriptions compared to 25.4 prescriptions if they had coverage (Poisal, forthcoming).
- Beneficiaries reporting their health as fair or poor filled 36.7 prescriptions in 1996 if they had full-year drug coverage, but only 27.2 prescriptions if they had no coverage. Those with part-year drug benefits filled 34.3 prescriptions for the year (Stuart, et al., 2000).
The source of coverage also appears to matter:
- In 1995, Medicare beneficiaries with Medicaid drug coverage filled an average of 27 prescriptions compared to 16.2 for Medicare risk HMO enrollees and 18.6 for those with employer-sponsored coverage (Poisal, et al., forthcoming).
There have been no systematic studies of the relationship between drug coverage and the composition of drug utilization, i.e., the selection of specific therapeutic products for a given disease. However, there is a widely held belief that insurance affords better access to newer, more expensive therapeutic agents. One recent finding consistent with this view shows that Medicare beneficiaries in 1996 who reported good to excellent health utilized drug products with an average cost of $40.70 if insured, but only $28.74 if they were not covered for prescription drugs (Stuart, et al., 2000).
All of these reported findings are subject to a major caveat. Although the differences in drug cost and use associated with insurance coverage appear both large and consistent, they are purely descriptive and do not necessarily mean that drug coverage causes or induces beneficiaries to use more. Indeed, some analysts argue that the differences are primarily due to the fact that sicker individuals who are heavy users of prescription medication are more likely to purchase drug coverage or enroll in public plans that provide it. While it is true that beneficiaries in poorer health are more likely to have prescription coverage (as noted in section 2.3 above), that does not rule out an independent insurance effect on demand.
There is a small but growing analytical literature that attempts to disentangle the various influences on the demand for prescription drugs by the elderly. Early studies by Long and Gordon (1989) and Long (1989) support the view that observed differences in drug use for persons with drug coverage under Medigap plans are the result of adverse selection rather than an insurance effect. These authors find no insurance effect among Medicare beneficiaries with employer coverage (where there is presumably little adverse selection).
More recent work supports the opposite view that drug coverage does induce additional usage. Based on data from a survey of Pennsylvania elderly, Coulson and Stuart (1995) and Coulson et al. (1995) found that prescription coverage increased drug use by approximately 3 percent for every 10 percent reduction in out-of-pocket cost to beneficiaries, all else being equal. These authors also found that the primary effect of drug coverage is to induce additional persons to use prescription medicine as opposed to increasing prescription use among users. This “hurdle” phenomenon associated with patient cost-sharing was first observed in the famous Rand Health Insurance Experiment of the late 1970’s and early 1980’s (Manning et al., 1989). It has since been observed in a number of recent studies of the insurance effect of drug coverage on elderly Medicare beneficiaries (Stuart and Grana, 1998; Stuart and Zacker, 1999; Ya-chen, 1999; Lillard, Rogowski, and Kingston, 1999). These studies all reach the same basic conclusion—drug coverage increases the probability of drug use but has minimal effect on the number of prescriptions filled by users. This conclusion is strengthened by the fact that the studies focused on different groups of elderly using a variety of databases. Stuart and Zacker (1999) used MCBS data to assess the impact of drug copayments on elderly Medicaid/Medicare dual eligibles. Stuart and Grana (1998) analyzed survey data on medicine use for 23 common health problems reported by a large sample of Pennsylvania elderly. Ya-chen (1999) used pharmacy records from the Dialysis and Morbidity and Mortality Study to study non-Medicare covered drug use by ESRD patients. Lillard, Regowski and Kingston (1999) analyzed survey data on prescription use in the RAND Elderly Health Supplement to the 1990 Panel Study of Income Dynamics.
The empirical estimates of insurance effects produced by these studies vary within a relatively narrow range. The addition of drug coverage is estimated to increase the probability of any prescription being filled by between 4 and 16 percent depending on population subgroup and generosity of drug coverage. The effect of adding a comprehensive drug benefit to Medicare is estimated to increase overall drug spending among elderly beneficiaries by between 20 percent (Lillard, Regowski and Kingston, 1999) to 34 percent (Coulson and Stuart, 1995).
2.5 Out-of-Pocket Prescription Drug Spending by Medicare Beneficiaries
Medicare beneficiaries who have no prescription coverage must, by definition, bear the entire cost of their drug purchases out of pocket. However, the obverse does not follow; no prescription plan currently available to Medicare beneficiaries covers the entire cost of outpatient prescription drugs. Were Medicare to adopt a drug benefit, it would presumably provide the greatest benefit to those who are presently most vulnerable to high out-of-pocket spending for drugs. The obvious question for policy makers is: What factors make Medicare beneficiaries most vulnerable to high out-of-pocket drug costs?
The literature on this issue identifies five principle factors: (1) total spending on prescription drugs, (2) insurance coverage, (3) income, (4) health, and (5) functional status. According to the Health Care Financing Administration, half of all drug spending by Medicare beneficiaries in 1995 was paid for out of pocket (Poisal, et al, 1999). The single greatest predictor of high out-of-pocket spending, is high total spending. The National Academy of Social Insurance estimates that 14 percent of Medicare beneficiaries had drug spending exceeding $2,000 from all payment sources in 1999 (Gluck, 1999). Almost a third of these individuals also had out-of-pocket drug spending greater than $2,000 for the year (Gluck, 1999).
The presence of prescription coverage mitigates against high out-of-pocket drug costs, but does not preclude them. A recent study published by AARP, estimates that Medicare beneficiaries with some drug coverage in 1999 spent just 3 percent of income on out-of-pocket drug purchases compared to 6 percent for beneficiaries without coverage (Gibson, et al, 1999). The income of Medicare beneficiaries is often lower in proportion to other populations, thus the out of pocket expenses are relatively burdensome. However, 42 percent of those with out-of-pocket drug spending exceeding $1,000 also maintained some prescription coverage. The principal reason for this is the limited scope of benefits offered by health plans that supplement Medicare, particularly individual Medigap policies (Gibson, et al, 1999). The three Medigap policies with drug coverage all have a $250 deductible followed by a 50 percent coinsurance up to maximum payment caps of $1,250 or $3,000. Stuart et al. (2000) found that Medicare beneficiaries with continuous coverage spent about half as much out-of-pocket in 1996 on prescription drugs ($219) as persons with part year coverage ($424) or no coverage at all ($468).
There is surprisingly little variation in total or out-of-pocket drug spending by income level (Poisal, et al., 1999; Gibson, et al, 1999; Poisal, et al., 1999 forthcoming). In 1995, for example, beneficiaries with annual incomes below $10,000 spent an average of $707 for prescription drugs of which $206 represented out-of-pocket payments. Those with annual incomes over $30,000 spent an average of $642 of which $226 was out of pocket (Poisal, et al., 1999). However, this similarity in dollar spending means that low-income beneficiaries spend proportionately more of their income on prescription drugs. Gibson, et al., (1999) estimate that beneficiaries below the poverty level in 1999 ($8,760 for individuals and $11,334 for couples) spent nine percent of their incomes on drugs compared to just 2 percent for those with income above 400 percent of the poverty line.
Twenty-seven percent of Medicare beneficiaries report being in fair or poor health. These individuals are the heaviest consumers of prescription medicines of any group and bear the highest average burden of out-of-pocket costs. Gibson, et al. (1999) estimate that they paid $590 per-person in out-of-pocket prescription drug payments in 1999, representing seven percent of annual income. Being uninsured and in poor health magnifies the burden. Stuart, et al. (2000) show that beneficiaries self-reporting poor health and no drug coverage spent $732 out of pocket in 1996 compared to just $318 for those with continuous drug coverage for the year.
As this last statistic demonstrates, the burden of out-of-pocket drug expenses varies widely depending on the constellation of attributes that beneficiaries manifest. Because these attributes tend to be inter-correlated, to understand the independent effect of each predictor requires multivariate analysis. To date there are no published papers in this area. A study by Crystal et al. (in press) examines predictors of aggregate out-of-pocket spending by Medicare beneficiaries for all types of health care combined using standard multivariate techniques. They find that self-reported health status, number of medical conditions reported, privately purchased Medicare supplemental health insurance, and functional impairment all increase the level of predicted out-of-pocket payments, while only HMO participation predicts lower payments (Crystal, et al., in press). Given that outpatient prescription drugs comprise approximately a third of all out-of-pocket health spending by beneficiaries (Crystal, et al., in press), one would expect these multivariate findings to hold true for prescription drug spending by itself.
2.6 What We Know and Need to Know
This section reviews what is known as well as that which remains unanswered in the context of the bolded questions presented throughout the discussion above. There are some common themes. The first crosscutting theme is that the literature focuses on drug use and spending almost as if “prescription drugs” were a single homogeneous product. A few studies have analyzed utilization at the level of the therapeutic class, but there has been virtually no research on the nexus between insurance coverage and appropriate or inappropriate drug use. The presumption is that lack of drug coverage is a barrier to appropriate drug therapy and impacts access to medications. It is also possible that lack of coverage reduces exposure to inappropriate drug use and thereby reduces the incidence of adverse drug events. More research is needed to clarify this issue as adverse drug events contribute to additional health expenditures through hospitalizations to treat these events as well as the costly cycle of prescribing additional drugs to treat the side effects of many previously prescribed drugs. Further research is also warranted to identify the relevant elements in insurance contracts that demonstrate empirical evidence of assuring quality and safety in drug use.
A second common theme in the literature is the focus on aged Medicare beneficiaries to the near exclusion of the disabled and beneficiaries with ESRD. Nearly all of the studies reviewed here focus strictly on the elderly or simply subsume the disabled in with the aged. Twelve percent of Medicare beneficiaries or approximately 5 million individuals receive their program entitlement from disability insurance. These beneficiaries are (by definition) all under age 65 and have very different characteristics and prescription drug needs than their aged peers. For example, there is a much higher prevalence of mental illness among the disabled compared to the elderly, making behavioral medicine a critical issue for this population group. Also, the disabled do not have the same access to private Medigap supplements as those over 65 years of age.
Another common characteristic of the literature on drug use and spending by Medicare beneficiaries is that it is largely descriptive rather than analytical. Making public policy on the basis of means, frequency distributions, and cross-tabulations can be perilous given the interdependence of factors relating to drug coverage, use, and spending in this population. There is a need for additional multivariate research in each of the areas addressed in this review as identified below.
What factors explain drug spending by Medicare beneficiaries? This issue is paid considerable attention in the literature, but the studies are predominantly descriptive. We know that beneficiaries in poor health are high spenders, but we have little knowledge of the drugs they use or the illnesses they treat with them. We know that white beneficiaries spend more in total for prescription medicine than blacks, but we do not know whether the explanation is racial or is due to some other co-varying factors such as income or education.
Do Medicare beneficiaries pay more for their prescriptions than other Americans do? There is scant research on this topic and even fewer answers. The question is important, as limitations in prescription drug policies place many beneficiaries with private insurance at risk of significant out-of-pocket purchases. The “best price” practices reserved for large purchasers of prescription drugs means that Medicare beneficiaries who buy prescriptions without the benefit of group volume discounts will tend to pay higher prices. It would be useful to know whether Medicare beneficiaries with supplemental prescription coverage are reaching maximum expenditure caps earlier in the year because of drug price increases. Research is also needed to examine the extent of generic drug use by age and how this is influenced by rising drug prices and drug benefit design.
What are the characteristics of Medicare beneficiaries with and without drug coverage? We know who has drug coverage but not why. Here is another instance where lack of analytic studies hinders interpretation of the descriptive statistics about which beneficiaries maintain coverage and which do not. A particularly important question yet to be answered is whether beneficiaries with the greatest need for coverage find it easy or difficult to obtain it. An equally important question is how stable the prescription coverage is for those who have it.
Will the trends in prescription coverage of Medicare beneficiaries witnessed in the early and mid 1990s continue? Most commentators think not, but the evidence to date is strictly anecdotal. Reliance on the Medicare Current Beneficiary Survey as the primary data source on Medicare drug coverage, means that the most up to date information is almost four years old before it is published.
Does drug coverage encourage Medicare beneficiaries to use more outpatient prescription medications? The answer is yes, but additional studies are critical to improve the precision of the estimated size of the “insurance effect.” Also needed is research that examines the characteristics of beneficiaries most strongly influenced by prescription coverage as well as drug regimens they use (or would use if given the opportunity). We need to know whether the observed difference in utilization rates between the insured and the uninsured arises because the uninsured fail to fill prescriptions or because the insured are simply prescribed more medications.
Does drug coverage influence the type and cost of the medications used? There is little known about this important issue. Research is needed on the potential influence that drug coverage plays on the patient-physician relationship. For example, do Medicare beneficiaries without drug coverage visit the doctor less often (and thereby avoid the problem of getting prescriptions that they would not fill anyway)? Are doctors less likely to prescribe medications to those without coverage? Are they more likely to prescribe less expensive generic drugs to these clients?
What factors make Medicare beneficiaries most vulnerable to high out-of-pocket drug costs? This is perhaps the best understood of all the major issues raised in this review. Although the research to date is descriptive, the range of comparisons is large enough to give a clear profile of which groups of beneficiaries are at greatest economic peril from out-of-pocket drug costs.
Section 3.
3.1 National Trends in Drug Spending
While prescription drugs represent only about seven percent of total national health expenditures, in absolute dollars the market is formidable (Copeland, 1999). In 1998, the prescription drug market represented over $91 billion for approximately 2.5 billion prescriptions at the retail level. The average prescription cost increased 11.9 percent over the prior year (Glaser, 1999). Projections of U.S. retail spending on prescription drugs forecast expenditure levels of $103 billion in 1999 and $143 billion by 2002 (Sager, 1999). Increases in total drug spending are concentrated in a relatively small number of therapeutic categories. Four drug categories account for 30.8 percent of the total $42.7 billion increase in drug spending between 1993 and 1998: antihistamines, antidepressants, cholesterol lowering agents, and anti-ulcerant drugs. (Barents, 1999).
Growth in drug spending is generally associated with two primary factors: (1) higher drug prices; and (2) increased demand for drugs (Smith, 1999). High drug prices account for about 64 percent of the total 1993-98 increase in drug spending (Barents, 1999). This is largely due to the recent flood of new and more costly drugs into the market following revisions in the Food and Drug Administration (FDA) procedures to accelerate the drug approval process. In 1998, the average price per prescription for a new drug was $71.49, which is twice the average ($30.47) for previously available drugs. Forecasters expect a slowdown in these introductions as the industry’s drug development pipeline stabilizes to the new regulatory timelines (Smith 1999).
Increases in the number of drugs that patients use are largely attributed to the drop in out-of-pocket costs, resulting from greater prescription coverage through managed care plans and, until recently, a deceleration in drug price inflation (Smith 1999). Another influence is the change in FDA policy in 1997 (and finalized in 1999) to allow pharmaceutical manufacturers a freer hand in advertising directly to consumers through mass media. In 1998, pharmaceutical manufacturers spent $8.3 billion promoting their products, of which $1.3 billion was in direct-to-consumer advertising (Barents, 1999).
3.2 Drug Distribution Chain
Providing prescription drugs to patients involves an intricate, multifaceted system of drug distribution that includes a number of players such as pharmaceutical manufacturers, wholesalers, retailers, third-party administrators, pharmacy benefit management companies, managed care organizations, and providers. Each of these players participates in the pricing of drugs as either purchasers (who try to reduce their outlays) or providers (who try to maximize profits). The following section summarizes these roles.
While every facet of the drug distribution system attracts attention, pharmaceutical manufacturers tend to attract an inordinate amount because perception holds them as the most profitable link in the distribution chain. At the manufacturer level, there are two major distinctions in product lines: 1) brand name drugs and, 2) generic drugs. Typically, innovator companies who carry the burden of research and development costs that are factored into the prices of marketable products develop brand name drugs. Estimates of the cost of bringing a new drug to market range from $125 million to $500 million. In 1998, pharmaceutical companies spent over $17 billion in research and development (PhRMA, 1998). The costs are, as a rule, borne during the drug development process and recouped after market approval during the time when the patent is still in place. In 1998, the median approval time for a new drug application was 12 months for a New Drug Application and 12 months for a new molecular entity (NME) (CDER, 1998). Research-intensive manufacturers generally try to price their products so that the bulk of research and development costs are recovered before competitors enter the market.
When a patent expires on a brand name prescription product, other manufacturers may enter the market and produce generic equivalents of that particular product. A generic equivalent is essentially a drug that is chemically identical to the brand name drug and is also bioequivalent. Generic pharmaceutical companies invest far less research and development monies to gain FDA approval for marketing their products. A generic manufacturer conducts only one or two bioequivalency studies before entering the market. These studies must demonstrate within certain statistical parameters that the generic version of the drug is absorbed into a patients blood stream at approximately the same rate and duration of time as the brand name drug (CDER, 1998).
Unlike the pricing practices of the brand name manufacturer industry, the generic industry prices its products as another commodity trying to gain market share through lower prices. Generics offer one of the earliest and most frequently used methods of containing costs. Since generic drugs are usually more price sensitive to the market than brand name drugs, generic drugs are often required by third-party programs. It is important to note the program does not necessarily care whether patients receive a brand name drug as long as the price is no greater than that of the generic drug.
Nearly all of major research-intensive brand name manufacturers, numbering approximately 100, are members of the trade association, Pharmaceutical Research and Manufacturers of America, known as PhRMA. PhRMA reported that the domestic U.S. sales of its members were $81 billion in 1998 (PhRMA 1998). In contrast, the generic industry reported sales of $8 billion in 1998 (GPIA, 1998).
Not surprisingly, the marketing techniques of brand name and generic manufacturers differ considerably. Brand name manufacturers tend to focus their marketing strategies primarily on physicians, who will potentially prescribe their products, while the generic manufacturers focus on the purchasers, e.g. pharmacies and hospitals. Since generic drugs tend to be less expensive than brand name drugs, they are as a rule preferred by insurance programs and other third-party payers and even required when available for reimbursement. There are exceptions to this rule, however, such as when a large purchaser may negotiate a brand name price that is competitive if not less expensive than the generic alternative.
Drug wholesalers operate as the middlemen between the drug manufacturers and the retail pharmacy. The wholesaler provides a very useful function to both independent and chain pharmacies because it buys in very large amounts and then distributes in small allotments. This relieves the pharmacy from the burden of dealing with each individual manufacture for every purchase (Smith, 1975). In most cases, wholesalers also provide products within 24 hours, which helps the pharmacy maintain a less costly inventory.
Over the past few years, the wholesale drug industry has become quite concentrated. While there are still a number of wholesalers in operation, the top five wholesalers account for 90 percent of the entire wholesale drug market. (NWDA, 1999) In 1998, the net sales of prescription drugs by wholesalers were 57 billion dollars. Such concentrated purchasing allows these few pivotal entities much influence over drug pricing at this juncture in the distribution chain.
The retail level of the distribution chain includes independent and chain pharmacies, supermarket pharmacies, mail order houses and Internet web-based pharmacies. There are approximately 50,000 independent and chain pharmacies in the United States. In the current market, the number of chain pharmacies is increasing as independents are being purchased by multi-store pharmacies. Chain pharmacies have recently undergone a period of consolidation and now the market power of the surviving chains is considerable. The ability to move large market shares of drug products allows chain pharmacies to command volume discounts from manufacturers and to contract directly with managed care organizations for exclusive distribution rights. The chains are also in a position to negotiate favorable rates with managed care organizations that need the chains in order to serve a geographically dispersed member population.
To reinforce the concept that prescription drugs at the retail level are commodities, it is important to note the eroding gross margins of pharmacies. Gross margins have dropped from a high point of approximately 40 percent to a low point of about 16 percent between 1996 and 1998 (NCP, 1998; Carroll, 1996). Many pharmacies, especially the smaller ones, cannot stay in business within the narrow gross margins permitted by third-party reimbursement programs. One study documented a gross margin decrease of 26.9 percent following the change in one insurance company’s reimbursement formula which caused the pharmacy a net loss on every prescription dispensed for members of that plan (Ganther, 1999).
Retail pharmacies use a variety of methods to control costs and maximize gross margin. One mechanism is to participate in a network of pharmacies to provide services to members of contracted health plans. The pharmacies in a network sign contracts agreeing to comply with the health plans' rules and regulations and to accept the reimbursement levels offered by the plan (Hejna, 1995). Membership in a network allows a pharmacy to participate in third-party reimbursement programs but this does not guarantee a reasonable reimbursement rate. Thus, pharmacies use other mechanisms to control their costs in obtaining prescription drugs. These include joining a buying group where the pharmacies consolidate market power to buy products with volume discounts. Some buying groups maintain warehouses of drug products and distribute them to the pharmacy members of the group as a way to reduce costs.
Internet pharmacies are a new phenomenon so their influence on drug prices is still largely unknown. It is interesting to note that they use mail order to distribute their products and thus are members of the mail order pharmacies’ professional organization, the Pharmaceutical Care Management Association (PCMA). Mail order pharmacy accounts for about 12 percent of the total retail prescription market. Between 1997 and 1998 mail service pharmacy grew by 19 percent (Anon., American Druggist, 1999). This compares to the total prescription market, which grew by 18.5 percent (Glaser, 1999). The proportion of the market accounted for by mail order pharmacy continues to grow at a small annual rate. Internet pharmacies, in addition to providing prescription drugs, provide a wide array of other drug related products such as over the counter medications, some cosmetic products, etc. Both mail order and Internet pharmacies must be licensed and comply with the same level of regulation as any traditional pharmacy establishment. In addition, the mail order pharmacies must also generally register with the states where they mail medications.
Mail order pharmacy is commonly included in prescription drug programs because mail order pharmacies can offer prescription drugs at a discounted price. This is because of the volume of their drug purchases and economies of scale. Patients in plans with mail order options are often provided financial incentives for using this type of pharmacy. These incentives may include discounted or forgiven copayments.
Third party programs in pharmacy cover a variety of players. The third-party is an entity other than the patient or pharmacist who is paying for all or part of the prescriptions. These include insurance companies, third party administrators (TPAs), state Medicaid programs, large employers, and managed care organizations. Third party drug programs provide a wide range of activities. These include claims processing, drug use review, formulary administration (which may include rebates) and disease management programs. TPAs are responsible for processing claims and conducting drug utilization review to ensure the quality of the program. However, TPAs are not financially at risk for any of the payments as are insurance companies or other programs (Gardner, 1986). To capture the growth in third-party programs, it is instructive to note that in 1968, perhaps five percent of prescriptions were paid for by some third-party, whereas in 1999 that figure is closer to 70 percent (Lyles, 1999). This growth includes all types of third-party programs, including insurance programs where an insurance company or other payer for the prescription drugs they would purchase may merely indemnify a patient.
The widespread use of computers in pharmacy practice has made it extremely easy for a plethora of third-party programs to exist and operate seamlessly within a particular practice setting. Typically, the pharmacist enters information into the computer on each patient and dispensed drug. The information is routed through a computerized network as a claim for the product cost and dispensing fee to the patient's third-party plan. Each third-party plan’s system has algorithms or reimbursement rules for coverage, eligibility, deductibles, pricing of the prescription, patient cost-sharing such as copayments, and therapeutic issues such as drug duplication or drug-drug interactions. The third-party plan’s system responds back within minutes with information on potential drug interaction alerts, copayments amounts required from the patient and reimbursement approvals. The plan's computer system may be maintained by the plan themselves, by insurance companies, by third-party administrators or by pharmacy benefit management companies (PBMs).
Pharmacy benefit managers (PBMs) are relatively new to the prescription drug arena and yet they now control a substantial part of the prescription drug market. PBMs act as intermediaries between pharmacists, patients, employers, managed care organizations, and third-party payers. According to Copeland, there are approximately 40 PBMs in the United States with the top five accounting for more than 75 percent of the market (Copeland, 1999). The U.S. General Accounting Office has issued at least two critical reports focusing on PBMs. The first GAO study addresses alliances between PBMs and pharmaceutical manufacturers. Several manufacturers own or legally participate in business alliances with PBMs, which has caused concern over whether this constitutes unfair competition. The theory behind this concern is that if a manufacturer owns a PBM then that manufacturer’s products might appear on the PBM’s formulary in an unfair manner and to the exclusion of other manufacturers’ products. The results of this particular report were not definitive enough to conclude that there was unfair competition (GAO, 1995). The second GAO study examined whether the Federal Employees Health Benefits Program (FEHBP) was satisfied with savings and services related to pharmacy benefit managers. The three FEHBP plans that they studied had contracted with PBMs to manage pharmacy benefit payments. The PBMs did save the plans a substantial amount of money, however the study notes that the plans' decision to use PBMs can shift business away from retail pharmacies to mail-order pharmacies (GAO, 1997). In addition to bias issues, HMO concerns about PBMs include confidentiality of data, disclosure of information to patients, and the HMO’s own oversight of the performance of PBMs (Office of Inspector General, 1997). Despite concerns, PBMs continue to be a dominant force in the prescription drug arena: PBMs currently serve over 150 million patients annually (PCMA, 1999).
As alluded to in one of the GAO reports, the PBMs have substantial relationships with the retail pharmacy sector. While the PBMs negotiate with drug manufacturers on the one hand, on the other hand they must assure adequate sites for patients enrolled in the various plans to obtain their prescription drugs. Therefore, they contract with retail pharmacies to provide this service as well as process claims and provide payment to the pharmacies. Obviously, the PBM will optimize its position by obtaining the widest geographic pharmacy coverage while keeping costs at their lowest. Chain pharmacies, with their considerable market power are able to negotiate more profitably (for them) arrangements with PBMs than are independent pharmacies that do not have the market share or geographic coverage of the chains. Because of this, relationships between independent pharmacies and PBMs are often quite strained (Ganther, 1999; Carroll, 1996).
One important way that PMBs control costs is through formularies. A formulary is a list that the plan uses to make reimbursement decisions about individual drugs. The formulary may be open or closed. An open formulary usually means that the plan will cover all drugs except those listed as exclusions to the drug reimbursement policy. A closed formulary details the specific drugs that meet the plan’s reimbursement policy. Preferred formularies impose lower copayment amounts for formulary drugs versus non-formulary drugs. Ideally, formularies are constructed with two primary considerations, first clinical and then cost (Grabowski, 1997). Restrictive formularies have been the subject of few peer reviewed research articles and the results are mixed. Horn (Horn, et al., 1996) suggested that restrictive formularies may result in increased costs and lower quality care while Walser et al. (1996) concluded that restrictive formularies might be more beneficial. Ideally, the list of covered drugs should first be based on sound clinical considerations after which cost should be taken into account. However, as Schulman (1996) has demonstrated, some PBMs report that the primary consideration is cost followed by clinical.
3.3 Drug Pricing
The pricing for prescription drugs is complex and varies greatly depending on who is the payer and who is the seller. The following is a summary of three major measures of prescription prices: the Producer Price Index, the Average Wholesale Price and the retail pharmacy charge.
The Producer Price Index (PPI) for pharmaceutical drugs measures inflation at the earlier stage of the production and marketing process. It is based on the prices of almost 50 therapeutic classes and indicates the prices paid at the wholesale level. The PPI is often used to determine whether the annual growth rate in drug prices exceeds those of other goods and services. The pharmaceutical manufacturer industry has long criticized the PPI as not properly assessing the impact of new medicines and thus overstating true drug inflation. (PhRMA, 1995)
The average wholesale price of a prescription drug (AWP) is undoubtedly the best known of the pricing terms. It is comparable to a sticker price on an automobile where the manufacturer suggests a certain price but almost everyone pays something different from that price. It tends to be a reliable price reference for brand name drugs but can be misleading with generic drugs since each manufacturer establishes its own AWP for the same product (Cohen, 1995). It is very important to have some reference point for pricing, negotiations, reimbursement, etc., and AWP serves effectively in this role. Unfortunately, the AWP does not really capture actual transaction prices including discounts and rebates (Schweitzer, 1997). The average wholesale price is referenced to some extent in most Medicaid and other third-party programs, but it is virtually unheard of for a plan of any size to pay AWP without extracting some discount from the retailer. Historically, pharmacists used AWP as their basis for pricing. However, they would usually purchase the drugs from wholesalers or manufacturers at some percentage discount from AWP and could thus retain the difference between what they paid for the drug and the cost basis for reimbursement as additional profit. As drugs became more of a commodity at the retail level, third-party payers including Medicaid programs and others, sought to share if not completely capture this "additional" profit. In effect, this substantially eroded the gross margins of pharmacies and thereby created friction between pharmacies and PBMs or other third-party payers. Using state Medicaid programs as an example, some states would reimburse pharmacists at AWP minus 10 percent plus a dispensing fee that varies depending upon the state. The AWP compares to the average manufacturer price (AMP) which is defined as the average price paid by wholesalers for products distributed to retailers (Cohen, 1995). This is obviously one step removed from the retail sector. This term gained widespread public notice with the passage of OBRA 90 where it was used as the reference point for calculating the rebates that drug manufacturers would have to provide to state Medicaid programs (NPC, 1998). Today, it is still used as the primary reference point for determining drug rebates in a variety of prescription drug plans.
The most common method of pricing in a retail pharmacy is to start with a cost basis for the drug and then add a pharmacy-dispensing fee to arrive at a selling price. For mechanical purposes, let us assume that an AWP for a particular prescription is $100 and the dispensing fee is $3. If there were no discounts from AWP included in this particular example, the prescription would be priced at $103. If the pharmacist paid AWP for this product then the pharmacist would gross $3 on that particular prescription. Now let us assume that the pharmacist is being reimbursed at AWP minus ten percent plus the $3 dispensing fee. In this case, the drug cost would be $100 dollars minus $10 (ten percent) or $90. Adding the dispensing fee of $3, the reimbursement price for this particular prescription would be $93. In this case, the pharmacist would need to be able to purchase the product for at least AWP minus 10 percent.
Retail prescription charges are frequently quoted in the lay press but they are difficult to interpret across different types of pharmacies. Drug Topics reports the average value per prescription-drugstore acquisition cost for 1998 at $36.79 per prescription (Glaser, 1999). For 1997, this cost is reported as $32.87 (Gebhart, 1998). However, the average prescription charge for 1997 for independent pharmacies ranged from $24.97 to $34.44 depending on overall sales volume, and was actually higher for pharmacies with high third party activity (NCPA, 1998). A Consumer Reports survey examined retail prices for five commonly prescribed drugs at 26 pharmacies, including chains, independents, supermarkets, mass merchandisers and online/mail order. In general, online pharmacies provided the best prices and independent the highest, although the authors note that the extra service provided by independent pharmacies may be worth it (Consumer Reports, 1999). In general, individual consumers pay more out-of-pocket for prescription drugs than do other more favored purchasers such as HMOs or the federal government (Bettehheim, 1999). The marketplace, as it currently exists, has created a wide range of prices for the exact same product.
One of the thorniest issues surrounding prescription drug prices is rebates. When a PBM is considering adding to or deleting drugs from the formulary, it will negotiate with individual drug manufacturers about providing incentives since the inclusion or exclusion will ultimately mean large dollars to the manufacturer. Generally, these incentives constitute a manufacturer’s rebate calculated as a percentage of the product that flows through the PBM to the plan. The rebate is often set in advance and based on the market share that a manufacturer expects to see for its product within a plan. As an example, assume that a manufacturer and PBM agree that sales of drug X for one year, at average manufacturer price (AMP), will be $5 million. Based on this, the manufacturer agrees to “rebate” back to the PBM 10 percent of the AMP for products dispensed though the particular plan represented by the PBM. The PBM would ultimately receive $500,000 from the manufacturer. The PBM, which is acting on behalf of a client such as a large employer, would receive some of this as a fee for services and the remainder remitted back to the client. The concept of a drug rebate was first widely applied when Congress passed the Omnibus Budget Reconciliation Act of 1990 (OBRA 1990) requiring drug manufacturers to provide rebates back to State Medicaid programs (NPC, 1998). Rebates have revolutionized the prescription drug marketplace. In 1994, rebates to all Medicaid programs totaled $1.8 billion out of total Medicaid outpatient drug payments of $9.5 billion (Lyles, 1999).
Rebates do not directly affect the retail sector, per se. While rebates have a substantial effect on the overall cost of drugs to a particular program, they are negotiated between the manufacturer and PBM or other third-party payer. Thus, when pharmacists dispense a prescription drug to a patient, they are essentially ignorant of any rebate arrangements made for that particular product. This is not to say that the retail sector is totally unaffected. For example, if a plan were not able to obtain the percentage rebates necessary to operate profitably, it may attempt to extract additional from the retail sector.
Section 4. International Drug Prices and Spending
“Why are American drug prices higher when compared to other countries?" Over the years, this has been a topic of much debate and study. Numerous studies have made drug price comparisons between the United States and other countries. Many of these are simply anecdotal reports appearing in the lay media. However, other more sophisticated analyses have also been published in the past decade. They all show that, to varying degrees, Americans do appear to pay higher drug prices than elsewhere in the world. How strong is this evidence? If it is indeed true, what factors are responsible? If the United States were to adopt regulatory policies implemented in other countries would drug prices fall here?
This chapter addresses each of these questions. The next section describes the factors that must be considered in making sound international price comparisons. A review and evaluation of four recent cross-national studies of drug prices follow this. Next is a section comparing prescription drug expenditures among European countries, the United States, and Canada. This section describes some of the reasons for differences in expenditure rates. The fourth section presents an overview of governmental price regulations adopted in other countries with detailed descriptions of the policies in place in Canada, France, Germany, and the United Kingdom. A concluding section summarizes what the United States can learn from these international experiences.
4.1 Making Sound International Price Comparisons
Although the literature appears to support the hypothesis that American drug prices are higher than prices in other countries, special care must be taken when making cross-national price comparisons. A recent article by Danzon and Kim (1998) found that cross-national study results are highly sensitive to methodological choices. These choices include sample selection, unit of measurement for price and volume, relative weight given to consumption patterns in the countries being compared, and exchange rates or purchasing power parities (PPP’s) for currency conversions. This section describes each of these issues.
The first challenge in international price comparisons is finding a list of drugs that is comparable across the study countries. Most comparative studies use small samples of leading, branded, on-patent prescription drugs. This can create selection bias given the fact that some products available in the US are not available in other countries and vice versa. A similar problem arises when products that have been approved and marketed in both the US and a foreign country become accepted therapy in one country but not in the comparator country. This phenomenon can result in large differences in market share, and hence prices, for identical products.
In addition, the life-cycle price profile of products varies among countries. Japan, for instance, has relatively high introductory prices but post-launch prices decline at a faster rate than in other countries. This creates a product age bias that can affect conclusions about average price levels. While this bias can be upward or downward, it is very dependent upon the country being examined. Another source of product selection bias arises when a study excludes lower-priced generic products. In some countries generics account for a third or more of all prescription sales, while in others they are less commonly prescribed. Generic products are sometimes available in foreign markets before or after introduction in the American market. This tends to increase the price of the foreign brand name drug. Excluding generic products will bias the price upward in countries that have a sizable generic market share and/or low generic prices, such as the United States (Danzon and Kim 1998; Schweitzer 1997).
The second challenge is matching units for a given drug. As specification of products become more precise (same compound, same manufacturer, same dosage form, pack size and strength) exact comparator products become more difficult to find. This is because the dosage form, strength or pack size may differ across countries. Researchers often impute prices for missing product strengths or dosage forms based on per unit prices, but this can lead to bias. For instance, in one study of Canadian versus American prices, the Canadian prices were calculated based on the largest package size available in Canada (GAO 1992). When converting prices to U.S. package sizes, the Canadian unit price was multiplied by the number of units in the U.S. package regardless whether the package sizes in the two countries were equivalent. Since unit prices are generally lower for large package sizes, the price comparisons are biased.
A third difficulty in cross-national comparisons is obtaining accurate transaction price data in the U.S. market. Many studies utilize US wholesale list prices when performing price comparisons. The wholesale list price does not incorporate any discounts or rebates afforded to large purchasers, such as Medicaid or large pharmacy chains. Some countries permit bulk packages, which are split by the pharmacist and dispensed to individuals, while many European countries forbid pack split, which leads to an underestimation of the European prices. The combination of an overestimated US price and an underestimated European price will inflate any existing price differentials (Danzon and Kim 1998, Schweitzer 1997).
A fourth challenge is accounting for differences in drug consumption patterns. Most cross-national studies use the drug consumption patterns in the United States as the base rate. Yet, medical norms in treating the same condition differ throughout the world. Differences in the mix of drugs and the significance of domestic products can also alter pharmaceutical consumption patterns. Failure to consider differences in utilization rates creates bias in price comparisons because price levels vary with volume. Less frequently prescribed drugs tend to be priced higher, other things being equal. This means that the popular American drug may appear more expensive in foreign countries, which in turn will bias foreign drug prices upwards. The opposite result occurs if the product requires a prescription in the US but not in the comparator country. In most cases, prescription-only products are more expensive. For instance, Claritin® is available in US as prescription-only but can be obtained in Canada over the counter. A comparison of Claritin® prices between the two countries would show lower prices in Canada, but the comparison would be misleading. Even comparing top selling products across the comparator countries will not alleviate the problem, since the drugs will be different in each country. One possible methodological approach to resolving this problem is to weight the price of the drugs based on level of consumption (Danzon 1997; Danzon and Kim 1998; Schweitzer 1997).
Currency conversions are inherently problematic in cross-national pricing studies. The use of exchange rates versus purchasing power parities (PPP) will yield different results. Using only exchange rates to calculate conversion prices does not directly address price differentials across countries for the same products. The PPP is created from a “basket” of similar drugs in the country used as the study base. Not all these drugs may be available in each country under comparison. Therefore, a PPP calculated for each country would be different, since the basket of drugs is different in each country. In addition, the PPP does not accurately compare purchasing power in different countries. For example, the official exchange rate may be two US dollars to the British pound. Yet, a product that sells for one dollar in the US may be priced at one pound in England. Using only an exchange rate will produce a conversion price for the product sold in Britain as approximately two US dollars. When a PPP is used, the conversion price may appear closer to one US dollar. The key is to employ both exchange rate and purchasing power parity conversions. If a study fails to report both conversions, it may be that the study results would change were the other conversion method employed. (Danzon 1997; Danzon and Kim 1998; Schweitzer 1997).
4.2 Studies of International Differences in Drug Prices
The US General Accounting Office (GAO) conducted one of the first widely cited studies of international price differences in prescription drugs in1992 (GAO 1992). The study compared 200 frequently prescribed drugs in the US and Canada sold by the same manufacturer in both countries. These drugs represented 54 percent of all prescriptions dispensed in US pharmacies in 1990. Since the drugs were supplied in a variety of sizes, strengths and dosage forms, the GAO selected a single commonly used US product for comparison. In Canada, 121 of the 200 were found to be identical to US dosage form, strength, and pack size. The remaining 79 products were not available for comparison. The GAO used the wholesale acquisition cost (WAC) to represent the American manufacturers' prices and the best available price (BAP) from the February 1991 Ontario Drug Benefit (ODB) formulary to represent Canadian prices. The study found that the 121 products cost an average of 32 percent more in the United States than in Canada.
This study represented an important first step in examining the issue of international drug price differentials. However, it also highlights a number of methodological issues discussed earlier (Schweitzer 1997, Danzon 1997). The inability to match more than 60 percent of the drugs originally selected for comparison raises the issue of product selection bias. How does the exclusion of certain products affect the measured price differentials? Those products matched were leading branded patented products in the United States, while the generic equivalents of these products were excluded from the study. If the price differentials for the generics were less than for the brand products, then the GAO procedures would overstate the true differences between the two countries. For those products matched on product strength and dosage form but not pack size, prices were imputed based on the unit price from the ODB formulary. In doing so Canadian prices appear lower, since the ODB unit price is based on the largest pack size available, not the most common pack size used in the US. The wholesale price list used to price US products did not include the discounts and rebates available to large purchasers. This too, tends to overstate the real differences in drug prices between the two countries. The GAO study also assumed that American drug consumption patterns for the products examined were the same as Canadian patterns when they are not (Katz et al, 1998, Pilote et al, 1994, Rouleau et al, 1993). Finally, the GAO used currency exchange rates and did not include a comparison based on the PPP. For all of these reasons, the main study conclusion that drug prices are lower in Canada must be treated with caution (Schweitzer 1997, Danzon 1997). Since 1992, other studies have been published that address some of the methodological concerns present here.
In 1994, the GAO followed up its earlier report with a comparative study of drug prices in the United Kingdom and the United States (GAO 1994a). Of the original 200 US drugs, only 77 were directly matched with products in Britain. In this study, the US price was based on the non-federal average manufacturer price (non-FAMP) from the Department of Veterans Affairs. The non-FAMP is a weighted average of the WAC, the list prices charged to wholesalers, and the lower prices charged to large purchasers such as HMOs. British prices were based on the national wholesale listing. These prices were then adjusted by a standard 12.5 percent discount rate provided to UK wholesalers. The exchange rate was taken as £ 0.5598 per dollar and the purchasing power parity (PPP) was £ 0.652 per dollar. Significant differences in prices were found between the United States and Britain. The GAO estimated that the 77 frequently dispensed drugs cost wholesalers 60 percent more in the US than in the UK.
This study advanced the field in measuring international drug price differences. The most important methodological advances were the use of a US price list that incorporated large purchaser discounts and comparisons based on both currency exchange rates and the PPP. Nonetheless, the study still has methodological shortcomings. By matching on brand name, pack size, manufacturer, strength, and form almost two-thirds of the original products selected for review were dropped from the study sample. As in its earlier Canadian analysis, the GAO imputed pack size price differences based on a single unit price, excluded generic equivalents of the branded products under study, and failed to consider differences in drug consumption patterns between the two countries (Schweitzer 1997, Danzon 1997).
In 1998, Danzon and Kim undertook a study designed specifically to assess the sensitivity of international drug price comparisons to the methodological issues described here. The study examined single ingredient cardiovascular products available between October 1991 and September 1992 in the US and eight OECD countries. The drugs included generic, over-the-counter, and brand name products. All pricing information was obtained from IMS sales data in the various countries. Two methods of product matching were employed; one based on the international product name (IPN), the other by anatomical therapeutic class. The researchers addressed the issue of dosage forms and strengths by converting all products to grams of active ingredient and the number of standard units, thereby permitting aggregation over all dosage forms, strengths and pack sizes. The authors address the issue of international differences in drug consumption patterns by using price indices weighted to reflect utilization rates in the US (a Laspeyres index) and each of the comparator countries (Paasche indices).
Danzon and Kim found that prices for cardiovascular drugs were generally lower in OECD countries than in the US regardless of the specific price index employed the matching criteria used, or the unit measure selected. The robustness of the results to alternative measurement approaches lends particular credibility to the study findings. However, it is worth noting that Danzon and Kim found much closer parity in international drug pricing than in previous studies, and in some cases the direction of the price advantages depended on the choice of methodology. For example, the reported price differentials for cardiovascular drugs in Canada compared to the US ranged from 16.6 percent higher to 6.9 lower depending on which price index was used. Consistently greater price differentials were found when using a Laspeyres index weighted to quantities of drugs used in the US compared to Paashe indices with foreign quantity weights. A similar range of positive and negative price differentials was reported between the US and Japan. Prices in the UK were all lower than in the US, ranging from 28 percent to 54 percent less depending on the index and matching algorithm. The price differentials between the US and Germany, France, Italy, Switzerland, and Sweden were comparable to the US and UK.
The real value of the Danzon and Kim study lies not in its specific findings relating to cardiovascular drug prices, but rather in its approach to making international drug price comparisons. The finding that measured price differences are sensitive to the methods selected is scarcely a surprise. However, the fact that certain methodological choices tend to produce consistently higher (or lower) measured price differentials than other choices should help inform future work in this area.
In 1999, the advocacy group Public Citizen conducted a survey of international prices for five newer antidepressants and three antipsychotic drugs in 17 North American and European countries (Sasich et al. 1999). Not all of the drugs were marketed in every country. All eight were available in the US, Canada, and Sweden, but only five were marketed in Portugal. The researchers contacted one English-speaking pharmacist in each country to obtain the pharmacy acquisition cost for an average 30-day supply for each available drug. Drug costs were converted to US dollars at the exchange rate for that day. Although this study determined that US drug prices were double those in the 17 other countries on average, this finding lacks credibility because of the small sample sizes (one pharmacist per country), poor product matching, failure to consider price discounts or rebates, failure to include generics (in Canada, for example, a generic version of one of the study drugs, Prozac (fluoxetine) has been available since the mid-1990’s), and finally, the failure to consider the impact of different drug consumption patterns on domestic prices.
Although the tools for making credible cross-national comparisons in drug prices have not been perfected, they have reached a level of sophistication that demands the attention of researchers and critics alike. Of the studies reviewed here, the Danzon and Kim analysis of cardiovascular drug prices sets a clear standard for others to follow. Their results confirm the widely held belief that Americans pay more for their prescription drugs than do Europeans, but the differences are lower than reported elsewhere. Moreover, depending on how prices are measured, cardiovascular drugs may actually cost more in Canada and Japan than here. Whether similar patterns hold for other drug groups has yet to be determined. Until further studies are done, the issue of who pays more is still an open question.
4.3 Cross-national Pharmaceutical Expenditures
From the perspective of public policy, the fact that drug prices vary across international boundaries is not as important as the cause of the differences. There are many other factors that can potentially contribute to cross-national price differences. These include national wealth, health insurance coverage of prescription medicine, the demographic makeup of the population, culture and its effect on medical practice patterns, and regulatory policies applied to prescription pricing.
It is essential that cross-national pricing studies be limited to countries with roughly similar levels of national wealth. By this standard, Mexico would not be considered an appropriate comparator country for the US as Mexico is at a less advanced stage of economic development. This means that real wages are lower, per capita incomes are lower, and prices for many goods and services, including pharmaceuticals are lower (Danzon, 1999). Comparisons of US drug prices to those in Canada and Europe are less affected by differences in national wealth given the similarities in level of economic development. However, some OECD countries like Portugal, Greece, and Ireland have substantially lower per capita GDP than there neighbors, and comparisons between the US and these nations should be conducted with caution.
A second important factor is the level of insurance coverage available to the residents of the countries under study. All of the OECD countries, except the United States, have some form of universal health insurance coverage. The level of coverage for pharmaceuticals varies from country to country. Chart 1 shows that France, Germany and the UK have high levels of public funding for pharmaceuticals compared to the US and Canada. The Access and Affordability Monitoring Project (AAMP) found that approximately 70 million or one in four Americans have no prescription drug coverage (Sager and Socolar, 1999). The presence of insurance coverage significantly increases utilization and expenditures for drug products (Lillard et al., 1999). It also provides a mechanism and an incentive for governments to control drug expenditures.

Data for France, Germany and United Kingdom are for 1995 (Lecomte and Paris 1998);
Canadian data are for 1993 ( Angus and Karpetz 1998); United States data are for 1994 (Kane 1997)
Demographics play a significant role in explaining international differences in level of health and pharmaceutical expenditures. However, these differences are generally not evident in simple cross-national comparisons. It is well known that seniors use more prescription drugs per year than other age categories. The expectation would be that countries that have a greater proportion of seniors would have greater health care and prescription costs. This does not hold true when comparing the US and Canada with European countries. France, Germany and the UK all have higher proportions of aged persons than either Canada or the US. Yet, chart 2 clearly shows that the US spends the highest percentage of GDP on health care (GAO 1994b, OECD 1998). In terms of pharmaceutical expenditures per capita, chart 3 shows that only France and Japan surpass the US (PhRMA 1999). Of course, the reason there is no direct correlation between age distributions and drug expenditures is that other factors associated with high health care spending also vary between these countries.
Source: OECD 1998

Source: PhRMA 1999
One methodological flaw present in almost all of the studies reviewed above is the failure to consider difference in drug consumption patterns among the comparator nations. Drug utilization is a function of the underlying norms of medical and health care practice in every country. Norms are part cultural and part imposed. For example, France has mandatory guidelines for physicians that outline “accepted” treatment options for given conditions (Bloor et al 1996). These leave little room for discretionary practice. Germany and the UK have capped prescription expenditure budgets for office-based physicians (Lecomte and Paris 1998). As discussed later, these give physicians a powerful incentive to prescribe generics and over-the-counter products. In the US, physician prescribing is influenced by drug formularies and financial incentives promulgated by managed care organizations. Basic cultural factors play a powerful role in the use of medicines to treat disease. In European countries, the use of alternative medicine, including herbals and homeopathy is an accepted standard of medical care and may be paid for under the insurance system. In the US, alternative medicine is not widely accepted by the medical profession although it is growing in popularity among patients. Still, the notion that no physician visit is complete without a prescription is a strongly ingrained attitude here (Barden et al 1998).
This review of factors that influence pharmaceutical utilization and expenditures is scarcely complete, but it does serve to set the stage for consideration of the importance of pharmaceutical price regulation. The next section describes the basic approaches used in controlling pharmaceutical prices and expenditures in OECD countries. The approaches used by four countries (Canada, Germany, France and the United Kingdom) are examined in detail.
4.4 Pharmaceutical Price Regulation
The OECD countries use a variety of pharmaceutical cost control approaches. The most common tactic in Germany is reference pricing, while France and Canada utilize product price controls and the UK uses profit regulation (Bloom and van Reenen, OECD, 1999; Dickson 1992). The most direct method of controlling drug costs is product price control whereby the government negotiates directly with each manufacturer to determine the reimbursement level for individual products. In a typical system the price is established after the drug product obtains marketing approval but prior to market introduction. The government or a government-sanctioned body reviews the manufacturer's price application and determines whether the requested price is "fair." If the price is not acceptable, the government can set a lower price. If the government-determined price is lower than the manufacturer requested, the company can appeal the decision. The price setting process usually takes one of two forms: internal, which primarily focuses on the manufacturer's price justification, or external, which examines the prices charged for the same or similar product in other countries (Dickson 1992; Earl-Slater 1997). When conducting an internal evaluation of a manufacturer’s price justification, governments take into account a myriad of factors such as the anticipated therapeutic benefit of the new product, anticipated sales volumes, and the company’s contribution to the economy (Le Pen 1996; Earl-Slater 1997). In setting a low price for a particular product, the government may take advantage of the fact that research and development expenses were incurred in another country (like the US) where the selling price is higher. This, in turn, creates an incentive for "parallel trade" in drug products, where drugs from a "low price" country are imported and resold in other countries where they command a higher price (GAO 1994b, Bloor 1996).
Regulatory systems that employ external price justifications for new drug products use different pricing criteria. Where the product under review is already marketed in other countries, these systems compare prices directly based on dose, strength, and pack size considerations. Where the product is not widely marketed elsewhere, countries develop comparative “market baskets” of drugs with the same therapeutic indication. The contents of these market baskets vary from system to system depending on which comparator drugs are included and whether generics are part of the package. The result is that the “fair” price for a given drug product will vary from country to country (Bloor 1996; Anis and Wen, 1998).
By whatever means a country sets the initial drug price, there is also the question of subsequent price changes. All governments that use direct drug price controls require approval for any product price increases after market introduction. (Le Pen 1996; Earl-Slater 1997; Anis and Wen ,1998)
In place of direct product-by-product price controls, some regulatory systems use a method known as “reference pricing”. In such a system, drug prices are compared by three different approaches: the same drug chemical, the same drug class, or therapeutically equivalent drugs that work via different mechanisms of action.2 Dickson and Redwood (1998) define reference pricing as the maximum reimbursement ceiling (reference price) set by the insurer for defined drugs. If the drug costs more than the reference price, the patient or supplementary private insurers are responsible for the difference (Dickson and Redwood 1998). Reference pricing differs from direct price controls in that the pharmaceutical firm can sell the product above the reference price if it believes that the patient is willing to pay the cost difference. The selling price is not strictly regulated on a product-by-product basis. Rather, reference pricing regulates groups of identical or similar drugs. Although reference-pricing systems differ greatly across countries they share four common objectives:
- Modify physicians' prescribing patterns to prescribe less expensive drugs.
- Influence patients to accept cheaper drugs or higher copayments.
- Pressure the pharmaceutical industry to lower prices in order to remain competitive.
- Control expenditures of the payer's drug budget. (Dickson and Redwood 1998; López-Casasnovas and Puig-Junoy 1999)
Reference pricing has been enacted in countries throughout the world, including Germany and British Columbia, Canada. It has become increasingly popular among private payers and Medicaid programs in the United States. The maximum allowable cost (MAC) pricing approach, used by many private insurers and Medicaid, uses reference pricing for drugs that are chemically identical. The effectiveness of reference pricing in influencing market prices depends upon several factors, including:
- The purchasing power of the buyer
- The scope of drugs covered under the reference pricing system
- Consumer's willingness to purchase the product at a given price
- The availability of substitutes for drug therapy, such as surgery or physical therapy (López-Casasnovas and Puig-Junoy 1999).
The general consensus is that reference pricing is effective in controlling drug costs, but the financial savings are short-term and subject to the law of diminishing returns. Initially, a significant reduction occurs in prices for referenced products resulting in short term savings for the insurer. Over time, the cost saving effect diminishes as pharmaceutical companies, physicians, and patients alter prescription consumption towards greater use of the reference-priced product (Dickson and Redwood 1998; Dickson 1992; Bloor 1996; López-Casasnovas and Puig-Junoy, 1999).
Some countries regulate the profits of pharmaceutical manufactures rather than the prices they may charge for their products. Britain and Spain both use this approach. Profit is defined by different methods including the rate of return on capital attributable to sales in that country or negotiated profit margins for each company (Bloor 1996). At regular intervals, a target profit rate is negotiated between each pharmaceutical company and the government. Profit regulation permits companies to price products at their discretion. If a company’s profits are above the negotiated profit rate, the excess is either remitted to the government or prices are reduced (Dickson 1992; Towse 1996; Lecomte and Paris 1998).
4.5 Country Specific Cost Control Approaches
The following sections present more detailed discussion of the drug cost control mechanisms used in Canada, Germany, France, and the UK.
4.5.1 Canada
In Canada, the provinces have responsibility for assuring their citizens access to health care under a Federal mandate that establishes general eligibility and benefit standards. All provinces provide some level of prescription coverage for the elderly and social assistance recipients. Coverage for the remainder of the population varies by province. Many of the consumer-oriented cost control techniques used in the United States are also used in Canada, such as formulary management, drug utilization reviews and copayments (Angus and Karpetz 1998).
In addition to consumer-oriented cost control mechanisms, Canada regulates the entry price of newly patented pharmaceuticals through the Patented Medicines Prices Review Board (PMPRB). The primary mandate of the board is “to prevent brand name firms from abusing their monopoly position during the market exclusivity period” (PMPRP 1998). The PMPRB is responsible for regulating the maximum prices charged by manufacturers for all patent-protected prescription and non-prescription drugs sold in Canada. The board has no authority over the prices charged further down the distribution chain by wholesalers or retailers. Furthermore, once a drug goes off patent its price is no longer regulated by the PMPRB. (Angus and Karpetz 1998; Anis and Wen 1998; PMPRB 1998).
The PMPRP issues pricing guidelines that defines “excessive” drug prices for three categories of products:
- Line extensions of existing medications, such as different strengths of the same product. A price is presumed to be excessive if it does not bear a reasonable relationship to the price of other strengths of the same drug sold by the company.
- Breakthrough drugs. A price is presumed to be excessive if it is greater than the prices of all other medications in the same therapeutic class and exceeds the median factory price charged for the same strength and dosage form in Germany, France, Italy, Sweden, Switzerland, the United Kingdom, and the United States.
- New chemical entities offering moderate, little or no therapeutic improvement. Price is presumed excessive if it exceeds the prices of all other medications in the same therapeutic class (Anis and Wen 1998; López-Casasnovas and Puig-Junoy 1999; PMPRB 1994).
The PMPRB also regulates prices of patent-protected products already on the market. In this case, a price increase is considered excessive if it exceeds the rise in the general consumer price index (CPI) (Angus and Karpetz 1998; Anis and Wen 1998; Dickson 1992; PMPRB 1994). If the price is found to be “excessive” the PMPRB can negotiate or order a price reduction. The company may appeal this decision through the court system. If the PMPRB and the company are unable to reach an agreement the PMPRB can revoke the patent on the product (Dickson 1992; PMPRB 1994).
Although Canada has the second lowest per capita spending on pharmaceuticals in the G7 nations, there is still an ongoing struggle for the government and drug benefit plans to contain costs. Provincial governments employ a number of drug cost control approaches such as patient copayments and formularies. The British Columbia government has instituted a policy of reference pricing for select therapeutic categories (López-Casasnovas and Puig-Junoy 1999). Initial research findings suggest that the regulation has produced a shift toward prescribing of the reference product in each therapeutic class and that drug expenditures within the targeted therapeutic categories have declined as a result (Narine 1999).
4.5.2 Germany
Over the past decade, the German government has introduced a number of substantial health care reforms affecting the sales of pharmaceuticals. Germany uses a combination of cost control mechanisms to manage its national drug budget. Both negative and positive “Lists of Reimbursable Drugs” (comparable to open and closed formularies) are used in formulary management (Schöffski 1996). In addition, Germany uses office-based physician drug budgets and prescription copayments along with reference pricing. Office-based physicians receive an annual budget for drug expenditures based on the number of patients under their care. Similar to a capitation system, this regulation places physicians at financial risk for their prescribing behavior.
The German government uses reference pricing to help control prices for drugs for which there are exact or close substitutes on the market. The reference price system assigns covered products to one of three levels:
- Level 1: Those products with identically active substances.
- Level 2: Those products with pharmacologically similar active ingredients
- Level 3: Those products with similar therapeutic effects (therapeutic reference pricing)
In 1993, the government introduced further reforms that froze drug prices and set an aggregate cap on drug reimbursements. If expenditures exceed the cap, financial penalties could be imposed on the medical professional associations and the pharmaceutical industry. Evaluations of the reform found some dramatic effects. After the cap was imposed, physicians prescribed more generics, decreased their prescribing of products with unproven efficacy, and increased referrals to specialists and hospitals. The cap did not affect drugs administered in hospitals where specialists had a significantly higher cap. Use and price of drug products not covered by the National Insurance system also increased after the cap was imposed. Finally, some generic manufacturers increased their prices to the reference price level (Schöffski 1996; Lecomte and Paris 1998; Ulrich and Wille 1996; von der Schulenburg and Uber 1997; von der Schulenburg 1997; Drummond 1997; López-Casasnovas and Puig-Junoy 1999).
4.5.3 France
Compared to other members of the OECD, the French pharmaceutical market is characterized by low prices complemented by high demand (Le Pen 1996). Pharmaceutical firms are free to set prices for all drugs not covered by the National Insurance plan, the Sécurité Sociale. The pricing committee, Comité Économique du Médicament (CEM) sets prices of covered drugs sold through non-hospital channels. The Ministry of Health determines the list of reimbursable drugs and the level of coverage for each drug. The CEM determines drug prices based on anticipated therapeutic benefit, anticipated sales volume, and other considerations. During the 1990’s, the French government instituted a number of innovations in an attempt to control drug expenditures. These included:
- A series of mandatory “good practice procedures" for physicians called Références Médicales Opposables (RMOs);
- An agreement between the state and the industry (Syndicat National de l’Industrie Pharmaceutique or SNIP); and
- A definition of enforceable regional spending targets (objectifs opposables) (Le Pen 1996; Lecomte and Paris1998; Pauriche and Rupprecht 1998).
The agreement between the industry and the state was designed to reduce sales volumes in exchange for price increases. This agreement was negotiated with each pharmaceutical company and outlined the conditions for permitting price increases. In return, the companies agreed to reduce promotional expenditures, promote “proper drug use” and provide information about its activities. The regional targets (objectifs opposables) represented negotiated agreements between the Sécurité Sociale and the physician associations. Separate targets were negotiated for specialist and general practitioner fees and drug prescriptions. In case of regional overspending, local physicians could be required to compensate the Sécurité Sociale for the excess spending. Despite the reforms, the French remains heavy consumers of prescription drugs relative to their OECD neighbors. The principal impact of the reforms appears to have been a slight shift toward less expensive drug products for treatment of medical conditions where RMOs have been implemented (Le Pen 1996; Lecomte and Paris 1998; Pauriche and Rupprecht 1998).
4.5.4 United Kingdom
The United Kingdom uses a variety of different methods to control drug costs. The primary method is through regulation of manufacturer profits. The Pharmaceutical Price Regulation Scheme (PPRS) regulates the rate of return on capital attributable to pharmaceutical sales in Britain. The government and the Association of the British Pharmaceutical Industry (ABPI) negotiate a target rate of return for each company based on brand-drug sales to the national health plan, known as the National Health Service (NHS). To reach the target rate (currently between 17 to 21 percent), the amount of research and development (including promotional expenditures) is set at about 9 percent of sales. Since the PPRS does not regulate prices per se, it permits the company significant flexibility in the launch price of new products. Once a product is marketed, any subsequent price increases require prior NHS authorization. In the event a manufacturer surpasses the target profit rate, it is permitted a 25 percent margin above the target rate, called the “gray area”. If the company earns more than the permitted "gray area” it must provide either reimbursement to the NHS or reduce the price. The PPRS does not address the pricing of generic products or products not reimbursed by the NHS (Lecomte and Paris 1998; Towse 1996; Bloom and van Reenen 1999; Burstall 1997; Burstall 1999).
Most prescription drugs available in the UK market are eligible for reimbursement from the NHS, but a few are included on a “Selected List” that prohibits government payment. Most drugs covered by the NHS are provided free of patient charge. Copayments are charged to certain segments of the population but more than half of all individuals are exempt. In all, only about 12 percent of prescriptions filled in the UK require a copayment, and for this reason copays contribute little in cost savings to the NHS drug budget (Towse 1996; Burstall 1997; Freemantle and Bloor 1996).
Another policy used in Britain to control drug spending targets doctors' prescribing of drugs (Towse 1996; Burstall 1997; Freemantle and Blood 1996). The NHS has introduced the Prescribing Analysis and Cost Information System (PACI) in an attempt to alter physician-prescribing patterns. This policy allows physicians to compare their prescriptions with a “theoretical” practice of patients with similar demographic characteristics. The practice guidelines are reported monthly. The PACT data have recently started to include price information on generics in order to encourage their use. Britain has also introduced voluntary drug budgets for general practitioners, called general fund holding practices.
Although the latest reforms in drug cost control in the UK have yet to be evaluated, the combination of control mechanisms is generally considered to have contributed to the fact that Britain has the lowest level of pharmaceutical expenditures per capita among the G7 nations (Burstall 1997).
4.6 Conclusion
Comparisons of prescription drug prices between the United States and other developed countries generally show that prices are higher here. That said, we end where we began with four major questions: (1) How much higher are drug prices in the US? (2) For which drug products are the differences the greatest? (3) Why are drug prices lower elsewhere? and (4) What can be done about it?
The primary difficulty in assessing the true magnitude of international drug price differentials arises from the fact that methodological problems make meaningful comparisons difficult. Different methods produce different results, as the Danzon and Kim (1998) study results show so dramatically. For this reason, it is critical that future research employ sensitivity analysis to test the robustness of study results to alternative assumptions regarding the types of drug products compared, their relative importance in terms of domestic consumption, and how they are priced.
Clearly, if we cannot answer the first question, the answer to the second is also unknown. Nonetheless, the findings from the Danzon and Kim (1998) study offer some clues and some lessons for future research. Their analysis of cardiovascular drugs found that prices were consistently higher in the US than in Europe regardless of alternative pricing and product matching criteria used in the study. However, the comparisons between the US and Canada and Japan showed that prices were lower in the US if a certain set of pricing/product matching conventions was used, but higher otherwise. The lesson here is that future research should concentrate on price comparisons of products within defined therapeutic classes.
Finding (and verifying) cross-national drug price variation is just the first step. Explaining why variation occurs is equally if not more important. The most important reasons are probably the country’s wealth and the extent of prescription coverage offered to its citizens. Other important factors include cultural morays and medical practice norms. Demographic differences play a role. The size and characteristics of a nation’s pharmaceutical industry matter. Drug price regulation surely plays a role. However, there is no body of empirical research that tells us which of these actors is the most important. This represents an important field for future investigation once we have better answers to the question of where drug price differentials are the greatest.
The last, and arguably most important question of what the US does about rising drug prices and expenditures is only tangentially related to the issue of whether Americans pay more for the drugs they use. Other countries in the OECD and elsewhere did not look at the American experience when deciding to embark on drug price and profit controls. Those decisions were based on domestic concerns. The same should be true here. Having good information about the effects of drug price controls (both positive and negative) is important, and the European experience is instructive in this regard. However, the US is unique in the size and innovativeness of its pharmaceutical industry. Most economists would agree with the pharmaceutical industry’s contention that current levels of pharmaceutical research and development in the US would decline under administered pricing. There is considerable disagreement about the magnitude of the potential drop. However, research and development is not the only matter of importance. All developed countries struggle to achieve a balance between health care expenditures, provider prices, and the rate of medical innovation. The key is to take explicit account of the tradeoffs.
Bibliography
American Association of Retired Persons (1997) Profile of Older Americans.
Angus D. Karpetz H. (1998) Pharmaceutical Policies in Canada. Pharmacoeconomics. 14 (Suppl. 1): 81-96.
Anis A. Wen Q. (1998) Price Regulation of Pharmaceuticals in Canada. Journal of Health Economics. 17:21-38.
Anon. (1999) Mail Order Drug Sales Leap 10 Percent. American Druggist. July: 1.
Anon. (1999) Relief for the Rx Blues. Consumer Reports. October: 38-46
Atherly A. (1999). The Effect of Medicare Supplemental Insurance on Medicare Expenditures. Department of Health Policy and Management. School of Public Health. Emory University. Atlanta. GA. Unpublished Paper.
Barents Group LLC. (1999) Factors Affecting the Growth of Prescription Drug Expenditures. National Institute for Health Care Management Research and Educational Foundation.
Berndt ER. Cockburn IM. Cocks DL. et al. (1998). Prescription Drug Prices for the Elderly. Monthly Labor Review September: 23-34.
Bettelheim A. (1999) Drugmakers Under Siege. CQ Researcher. 9(33): 753-776.
Bloom N. Van Reene J. (1999) Regulating Drug Prices: Where do we go from here? Institute for Fiscal Studies. L5, L6, D4. 19 (3): 321–342. London. England.
Bloor K. Freemantle N. (1996) Lessons from International Experience in Controlling Pharmaceutical Expenditure II: Influencing Doctors. British Medical Journal. 312(7045): 1525-1527.
Bloor K. Maynard A. Freemantle N. (1996) Lessons from International Experience in Controlling Pharmaceutical Expenditure III: Regulating Industry. British Medical Journal. 313 (7048): 33-35.
Burstall. M. (1997) The Management of the Cost and Utilization of Pharmaceuticals in the United Kingdom. Health Policy. 41 (Suppl): S27-S43.
Burstall M. Reuben B. Reuben A. (1999) Pricing and Reimbursement Regulation in Europe: An Update on the Industry Perspective. Drug Information Journal. 33: 669-688.
Call K. Dowd B. Feldman R. Maciejewski M. (1999) Selection Experiences in Medicare HMOs: Pre-Enrollment Expenditures. Health Care Financing Review. 20(4): 197-209.
Carroll NV. Miederhoff PA. Waters LW. (1996) Profitability, Third Party Reimbursement, and Access to Community Pharmacies. Clinical Therapeutics. 18 (4): 702, 703-715.
Cartwright WS. Hu TW. Huang LF. (1992). Impact of Varying Medigap Insurance Coverage on the Use of Medical Services of the Elderly. Applied Economics. 24: 529-539.
Center for Drug Evaluation and Research (CDER). (1998) Report to the Nation: Improving Public Health through Human Drugs. Food and Drug Administration. U.S. Department of Health and Human Services. Washington D.C.
Center for Drug Evaluation and Research (CDER). (1998) Approved Drug Products with Therapeutic Equivalence Evaluations. 18th ed. Food and Drug Administration. United States Department of Health and Human Services. US Government Printing Office. Washington DC.
CibaGeneva. (1996) Pharmacy Benefit report: 1996 Trends and Forecasts. CibaGeneva Pharmaceuticals. Summit. NJ.
Cohen K. (1995) Glossary. Principles and Practices of Managed Care Pharmacy. Foundation for Managed Care Pharmacy. Alexandria. VA.
Cohen RA. Bloom B. Simpson G. et al. (1997) Access to Health Care. Part 3: Older Adults. National Center for Health Statistics. Vital Health Statistics 10 (198).
Copeland D. (1999) Prescription Drugs: Issues of Cost, Coverage, and Quality. EBRI Issue Brief. 208.
Coulson NE. Stuart, BC. (1995) Insurance Choice and the Demand for Prescription Drugs. Southern Economic Journal. 61(4): 1146-1157.
Crystal S, Harman J, Sambamoorthi U, Johnson R, Kumar R. (2000) Out of Pocket
Health Care Costs Among Older Americans. Journal of Gerontology:
Social Sciences, In press.
Danis M. Biddle AK. Henderson G. Garrett JM. DeVellis RF. (1997) Older Medicare Enrollees' Choices for Insured Services. Journal of the American Geriatrics Society, 45(6): 688-94.
Danzon PM. (1997) Pharmaceutical Price Regulation: National Policies versus Global Interests. The AEI Press. Washington DC.
Danzon P. Kim J. (1998) International Price Comparisons for Pharmaceuticals: Measurement and Policy Issues. Pharmacoeconomics. 14 (Suppl. 1): 115-128.
Davis M. Poisal J. Chulis G. Zarabozo C. Cooper B. (1999) Prescription Drug Coverage, Utilization, and Spending Among Medicare Beneficiaries. Health Affairs. 18(1): 231-43.
Dickson M. (1992) The Pricing of Pharmaceuticals: An International Comparison. Clinical Therapeutics. 14 (4): 604-610.
Dickson. M, Redwood H. (1998) Pharmaceutical Reference Prices: How Do They Work in Practice? Pharmacoeconomics. 14(5): 471-479.
Drummond M. Jönsson B. Rutten F. (1997) The Role Of Economic Evaluation in the Pricing and Reimbursement of Medicines. Health Policy. 40: 199-215.
Earl-Slater A. (1997) A Study of Pharmaceutical Policies in the EU. Policy Studies. 18 (3/4): 251-267.
Hillman A. Pauly M. Escarce J. Rippley K. Gaynor M. Clouse J, Ross R. (1999) Financial Incentives and Drug Spending in Managed Care. Health Affairs. 18 (2): 189-200.
Ettner S. (1997) Adverse Selection and the Purchase of Medigap Insurance by the Elderly. Journal of Health Economics. 16: 543-562.
Families USA. (1999) Hard to Swallow: Rising Drug Prices for America’s Seniors. Publication No. 99-107. November.
Freemantle. N. Bloor. K. (1996) Lessons from International Experience In Controlling Pharmaceutical Expenditure I: Influencing Patients. British Medical Journal. 314(7044): 1469-1470.
Ganther JM. Kreling D. (1999) Effect of a Change in Third Party Reimbursement Rate on Prescription Gross Margin. Journal of the American Pharmaceutical Association. 39(3): 346-352.
Gardner T. (1986) Structuring Drug Benefits. In final report of APhA Commission on Third-party Programs.
Gebhart F. (1998) Annual Rx survey: The New Golden Age. Drug Topics. March 16: 71-83.
Generic Pharmaceutical Industry Association (GPIA). (1998) Facts and Figures. Washington, DC.
Gibson M. Brangan N. Gross D. et al. (1999) How Much Are Medicare Beneficiaries Paying Out-Of-Pocket For Prescription Drugs? Washington DC: AARP Public Policy Institute.
Glaser M. (1999) Boom year. Drug Topics April 5: 47-53.
Gluck ME. (1999) A Medicare Prescription Drug Benefit. National Academy of Social Insurance.
Gold M. et al. (1999) Medicare Managed Care: Preliminary Analysis of trends in Benefits and Premiums, 1997-1999. Mathematica Policy Research. Washington D.C.
Grabowski H. Mullins CD. (Aug 1997) Pharmacy Benefit Management, Cost-Effectiveness Analysis and Drug Formulary Decisions. Social Science Medicine. 45(4): 535-44.
Gross DJ. Alecxih L. Gibson MJ. Corea J. Caplan C. Brangan N. (1999) Out-of-Pocket Health Spending by Poor and Near-Poor Elderly Medicare Beneficiaries. Health Services Research, 34(1 Pt 2): 241-54.
Hamilton BH. (1999) HMO Selection and Medicare Costs: Bayesian MCMC Estimation of a Robust Panel Data Tobit Model with Survival. Health Economics and Econometrics. 8: 403-414.
Hejna Chester Pharmacy Networks: Origins, Functions and Future Directions. (1995) Principles and Practices of Managed Care Pharmacy. Foundation for Managed Care Pharmacy. Alexandria. VA: 119.
Hellinger F. (1995) Selection Bias in HMOs and PPOs: A Review of the Evidence. Inquiry. 32: 135-148.
Hensley S. (1999) Prescription Costs Harder to Swallow. Providers and Payers Get a Big Dose of Reality with Explosive Spending and Patient Demand for New Drugs. Modern Healthcare. 29(34): 30-4.
Hewitt Associates (1999) Retiree Health Coverage: Recent Trends and Employer Perspectives on Future Benefits. Menlo Park, CA: The Henry J. Kaiser Family Foundation.
Horn SD. Sharkey PD. Tracy DM. et al. (1996) Intended and Unintended Consequences of HMO Cost Containment Strategies: Results from the Managed Care Outcomes Project. American Journal of Managed Care. 2: 253-64.
Hurd MD. McGarry K. (1997). Medical Insurance and the Use of Health Care Services by the Elderly. Journal of Health Economics. 16: 129-154.
Jacobzone. S. (1999) How can Pharmaceutical Policy Reconcile Social Objectives and Industrial Efficiency? A View from the Recent Experience of OECD Countries. Organization for Economic Cooperation and Development (OECD). Paris France.
Johnson RE. Goodman MJ. Hornbrook MC. Eldredge MB. (1997) The Impact of Increasing Patient Prescription Drug Cost Sharing on Therapeutic Classes of Drugs Received and on The Health Status of Elderly HMO Members. Health Services Research.32(1):103-22.
Kane. N. (1997) Pharmaceutical Cost Containment and Innovation in the United States. Health Policy. 41(Suppl): S71-S89.
Laschober M. (1997) Health and Health Care of the Medicare Population: Data from the 1993 Medicare Current Beneficiary Survey. Rockville MD. Westat, Inc.
Laschober M. Olin G. (1996) Health and Health Care of the Medicare Population: Data from the 1992 Medicare Current Beneficiary Survey. Rockville MD. Westat, Inc.
Le Pen C. (1996) Drug Pricing and Reimbursement in France: Towards a New Model? Pharmacoeconomics. 10 (Suppl. 3): 26-36.
Lecomte T. Paris V. (1998) Controlling Drug Expenditures in France, Germany, and the United Kingdom. Économie et Statistique. 312-313: 109-124.
Lillard LA, Rogowski J. Kington R. (1997) Long-Term Determinants of Patterns of Health Insurance Coverage in the Medicare Population. The Gerontologist. 37(3): 314-323.
Lillard LA. Rogowski J. Kington R. (1999) Insurance Coverage for Prescription Drugs: Effects on Use and Expenditures in the Medicare Population. Medical Care. 37(9): 926-936.
Long S. (1994) Prescription Drugs and the Elderly: Issues and Options. Health Affairs. 13(2): 157-74.
Long S. Gordon N. (1989) Updated Estimates of Medicare's Catastrophic Drug Insurance Program. Congressional Budget Office (CBO). Washington D.C.
López-Casanovas G. Puig-Junoy. J. (1999) Review of the Literature On Reference Pricing. Working Paper. Research Center on Health and Economics. Department of Economics and Business. Universitat Pompeu Fabra. Barcelona Spain. February.
Lutchmie N. Mahil S. Smith T (1999) Evaluating Reference-Based Pricing: Initial Findings and Prospects. Canadian Medical Association Journal. 161(3): 286-288.
Lyles A. Palumbo FB. (1999) The Effect of Managed Care on Prescription Drug Costs and Benefits. Pharmacoeconomics. 15(2): 129-140.
McArdle F. Copock S. Yamamoto D. et al. (1999) Retiree Health Coverage: Recent Trends and Employer Perspectives on Future Benefits. The Henry J. Kaiser Family Foundation: Menlo Park, CA.
Manning WG. Newhouse JP. Duan N. et al. (1987). Health Insurance and the Demand for Medical Care: Evidence from a Randomized Experiment. The American Economic Review. 77 (3): 251-327.
Minority Staff Report. (1998) Prescription Drug Pricing in the United States: Drug Companies Profit at the Expense of Older Americans. Committee on Government Reform. United States House of Representatives. Washington DC.
National Pharmaceutical Council (NPC) (1998) Pharmaceutical Benefits Under State Medical Assistance Programs. NPC. Reston VA.
Novartis, (1999) Pharmacy Benefit Report Facts and Figures: 1999 Edition. Novartis Pharmaceutical Company. East Hanover NJ.
National Wholesale Druggists' Association (NWDA) (1999) Industry Profile and Health Care Fact Book. Addendum.
Office of Inspector General. (1997) Experiences of Health Maintenance Organizations with Pharmacy Benefit Management Companies. United States Department of Health and Human Services. Washington D.C. April. Report Number OEI-01-95-00110.
Olin G. Liu H. (1998) Health and Health Care of the Medicare Population: Data from the 1994 Medicare Current Beneficiary Survey. Rockville MD. Westat Inc.
Organization for Economic Co-operation and Development (OECD). (1998) Social and Health Policies in OECD: A Survey of Current Programmes and Recent Developments. Paris France.
Pauriche P. Rupprecht F. (1998) Pharmaceuticals in France: a Review of Current Trends. Économie et Statistique. 312-313:7-20.
Pharmaceutical Care Management Association (PMCA) (1999) Facts and Figures, 1999.
Pharmaceutical Research and Manufacturers Association. (PhRMA) (1999) Pharmaceutical Industry Profile 1999. Washington D.C.
Pharmaceutical Research and Manufacturers Association. (PhRMA) (1998) Pharmaceutical Industry Profile 1998. Washington D.C.
Pharmaceutical Research and Manufacturers Association. (PhRMA) (1995) PhRMA Facts: Current Drug Price Increase Below General Inflation: GAO Says Past Increases Much Lower Than Reported. June. (http://www.phrma.org)
Physician Payment Review Commission (PPRC) (1996). Annual Report to Congress. ProPAC. Washington D.C.
Poisal JA. et al. (2000) Medicare Beneficiaries And Drug Coverage. Health Affairs. (forthcoming).
Poisal JA. Murray LA. Chulis GS. Cooper BS. (1999) Prescription Drug Coverage and Spending for Medicare Beneficiaries. Health Care Financing Review. 20(3): 15-27.
Riley G. Tudor C. Chiang YP. et al. (1996) Health Status of Medicare Enrollees in HMOs and Fee-for-Service in 1994. Health Care Financing Review. 17(4): 65-76.
Rowgowski J. Lillard LA. Kington R. (1997) The Financial Burden of Prescription Drug Use Among Elderly Persons. Gerontologist, 37(4): 475-482.
Sager A. Socolar D. (1999) Affordable Medications for Americans: Problems, Causes and Solutions. Prescription Drug Task Force. United States House of Representatives. Washington D.C.
Sasich L. Torrey E. Wolfe S. (1999) International Comparison of Prices for Antidepressant and Antipsychotic Drugs. Public Citizen’s Health Research Group. Washington DC.
Schöffski O. (1996) Consequences of Implementing a Drug Budget for Office-Based Physicians in Germany. Pharmacoeconomics. 10 (Suppl. 2): 37-47.
Schulman KA. Rubenstein LE. Abernathy DR. et al. (1996) The Effect of Pharmacy Benefit Managers: Is It Being Evaluated? Annals of Internal Medicine. 124: 906-13.
Schweitzer S. (1997) Pharmaceutical Economics and Policy. New York: Oxford University Press.
Smith H. (1975) Principles and Methods of Pharmacy Management. Lea and Febiger. Philadelphia PA.
Smith S. Freeland M. Heffler S. et al (1998) The Next Ten Years of Health Spending: What Does the Future Hold? Health Affairs. 17(5): 128-140.
Stuart B. Coulson NE. (1994) Use of Outpatient Drugs as Death Approaches. Health Care Financing Review. 15(3): 63-82.
Stuart B. Grana J. (1998) Ability to Pay and the Decision to Medicate. Medical Care. 36(2): 202-211.
Stuart B. Grana J.(1995) Are Prescribed and Over-the-Counter Medicines Economic Substitutes? A Study of the Effects of Health Insurance on Medicine Choices by the Elderly. Medical Care, 33(5): 487-501.
Stuart B. Shea D. Briesacher B. (2000) Prescription Drug Coverage For Medicare Beneficiaries: Coverage and Health Status Matter. The Commonwealth Fund. New York.
Stuart B. Zacker C. (1999) Who Bears the Burden of Medicaid Drug Copayment Policies? Health Affairs. 18(2): 201-12.
Towse A. (1996) The UK Pharmaceutical Market. Pharmacoeconomics. 10 (Suppl. 3): 14-25.
Ulrich V. Wille E. (1996). Healthcare Reform and Expenditure on Drugs: The German Situation. Pharmacoeconomics. 10(Suppl. 2): 81-88.
U.S. Department of Health, Education and Welfare. (1968) Task force on Prescription Drugs. Approaches to Drug Insurance Design: Background Papers. Washington, DC. Office of the Secretary of Health.
U.S. Department of Health and Human Services Office of Inspector General. (1997), Experience of Health Maintenance Organizations with Pharmacy Benefit Managment Companies. Washington, D.C. Government Document #OEI-01-95-00110.
U.S. General Accounting Office. (1992) Prescription Drugs: Companies Typically Charge More in the United States than in Canada. Washington D.C. Report Number GAO/HRD-92-110.
U.S. General Accounting Office. (1994) Prescription Drugs: Companies Typically Charge More in the United States than in the United Kingdom. Washington D.C. Report Number GAO/HEHS-94-29.
U.S. General Accounting Office. (1994) Prescription Drugs: Spending Controls in Four European Countries. Washington D.C. Report Number GAO/HEHS-94-30.
U.S. General Accounting Office. (1995) Pharmacy Benefit Managers, Early Results on Ventures with Drug Manufacturers. Washington D.C. Report Number GAO/HEHS-96-45.
U.S. General Accounting Office. (1996) Health, Education and Human Services Division, Pharmacy Benefit Managers, Early results on Ventures with Drug Manufacturers. Washington D.C. Report Number GAO-96-45.
U.S. General Accounting Office. (1997) Pharmacy Benefit Managers: FEHBP Plans Satisfied with Savings and Services, but Retail Pharmacies Have Concerns. Washington D.C. Report Number GAO/HEHS-97-47.
U.S. General Accounting Office. (1999) Prescription Drug Benefits - Implication for Beneficiaries of Medicare HMO Use of Formularies. Washington D.C. Report Number GAO/HEHS-99-166.
United State House of Representatives, Committee on Government Reform and Oversight, Minority Staff Report (1998) Prescription Drug Pricing in the United States: Drug Companies Profit at the Expense of Older Americans. Washington, D.C.
von der Schulenburg J. (1997) Management of Cost and Utilization of Pharmaceuticals in Germany. Health Policy. 41(Suppl): S45-S53.
von der Schulenburg J. Uber A. (1997) Current Issues in German Healthcare. Pharmacoeconomics. 12(5): 517-523.
Walser BL. Ross-Degnan D. Soumerai SB. (1996) Do Open Formularies Increase Access to Clinically Useful Drugs? Health Affairs. 15(3): 95-108.
Wolfe JR Goddeeris JH. (1991) Adverse Selection, Moral Hazard, and Wealth Effects in the Medigap Insurance Market. Journal of Health Economics. 10: 433-459.
Ya-Chen T. (1999). Effects of Insurance on Prescription Drug Use by ESRD Beneficiaries. Health Care Financing Review. 20(3): 39-54.
Abstracts
The articles abstracted below represent some of the best sources of material found
during the course of this study. The source of the abstract is listed at the end of each study summary. If no source is listed, it is the authors’ original abstract.
American Association of Retired Persons (1997) Profile of Older Americans
This AARP report provides a socioeconomic profile of older Americans’ in the United States for 1996. These variables include population in U.S. and by state, projected population growth, living arrangements, racial and ethnic composition, geographic distribution, income and poverty, employment, education, health and health care. It identifies older population-persons as 65 years of age and older at 33.9 million, representing 12.8%. Projections for future growth show the most rapid increase is expected between the years of 2010 and 2030 when the “baby boom” generation reaches 65. The study projects that older persons will represent 20% of the population by 2030, an increase of 100% from 1996 (staff).
Bloom N. Van Reenen J. (1998) Regulating Drug Prices: Where Do We Go from Here? Institute for Fiscal Studies, L5, L6, D4.19(3): 321–342.
This paper examines the arguments for changing the ways that UK drug prices are regulated. In the UK, NHS pharmaceutical expenditures on branded drugs, currently worth about £3 billion a year, have been regulated by the Pharmaceutical Price Regulation Scheme (PPR) since 1978. We argue that, in publicly funded healthcare systems, pharmaceutical price regulation is necessitated by a tendency towards excessive government expenditure because of over prescription and the monopoly power of firms with on-patent drugs. We briefly explain the operation of the PPRS, which is based on rate-of-return regulation, and discuss its merits and drawbacks. We then consider five alternative pricing systems: free pricing, therapeutic benefit pricing, international reference pricing, therapeutic reference pricing and RPI – X price regulation. However, we reject all these alternatives in favour of a reformed PPRS. We suggest three potential reforms of the PPRS: an RPI – X cost allowance if feasible or a widening of the rate-of-return bands otherwise; the introduction of a marketing innovation allowance; and greater regulatory transparency.
Carroll NV. Miederhoff PA. Waters LW. (1996) Profitability, Third-Party Reimbursement, and Access to Community Pharmacies. Clinical Therapeutics, 18(4): 702, 703-715.
The purpose of this study was to analyze the extent to which third-party reimbursement programs have affected the profitability and availability of community pharmacies. Data were taken from records maintained by the Virginia Board of Pharmacy and a survey of 177 community pharmacies. Between 1989 and 1994, 258 outpatient pharmacies opened and 342 closed. Chain and independent pharmacies suffered net losses, and supermarket and mass merchandiser pharmacies experienced net increases. Few significant changes occurred in the distribution of pharmacies over the study period. Fifty-nine chain and independent pharmacies and 1 supermarket pharmacy chain provided usable profit and reimbursement data. These pharmacies experienced declines in profits and increases in the percentage of prescriptions reimbursed by private third-party prescription programs over the last several years. Regression analyses indicated that higher ratios of sales of private third-party prescriptions to private-pay prescriptions were associated with lower profits. All respondents indicated that changes in private third-party reimbursement had substantially reduced profits over the past 5 years. The results indicate that the growth of private third-party payment has led to lower pharmacy profits but has not yet resulted in problems of consumer access.
Cohen RA. Bloom B. Simpson G. et al. (1997) Access to Health Care. Part 3: Older Adults. National Center for Health Statistics. Vital Health Statistics 10(198).
This report uses data from the 1993 Access to Care, the 1993 Health Insurance, and the Year 2000 surveys of the National Health Interview Survey to describe access to care for older adults. The findings indicate that health insurance status played a key role in an individual’s ability to obtain health care services. Elderly persons with Medicare only were 3 times as likely to go without prescription medicine or glasses as those with Medicare and private insurance coverage. Approximately 600,000 elderly persons were unable to get prescription medicine. (Staff)
Danis M. Biddle AK. Henderson G. Garrett JM. DeVellis RF. (1997) Older Medicare Enrollees' Choices for Insured Services. Journal of the American Geriatrics Society, 45(6): 688-94.
RESERACH OBJECTIVES: The aim of this study was to: (1) explore the possibility of allowing Medicare enrollees to choose limited insurance packages personally; (2) identify which services elderly adults would like insured; and (3) elucidate the reasons for their choices. STUDY POPULATION: We recruited 118 patients (77% response rate) receiving non-urgent care from the general internal medicine practice of a major teaching institution. STUDY DESIGN: We used in-person interviews and an innovative instrument as part of an anonymous, cross-sectional survey. The instrument, a "puzzle-like" board and 12 benefit cards, simulated the health insurance purchase decision by presenting information about premium costs and covered services to allow study subjects to "purchase" coverage for desired services. PRINCIPAL FINDINGS: Only 2% of subjects selected the package currently offered by Medicare; 52 different packages were chosen. Twenty-one percent of subjects selected a package that included coverage for hospital, intensive care unit, home care, outpatient care, prescription drugs, and either vision care (13%) or preventive services (8%). Only 9% elected to purchase coverage for long-term care services. Although there was some variation, costs and current or anticipated need were the most frequently cited reason for selection of coverage. Individuals forgoing long-term care coverage deemed other services as more essential (40%); did not anticipate a need (17%); or would rely on family for care (21%). CONCLUSIONS: The preferences expressed suggest that a majority would purchase a different combination of coverage than is currently offered; coverage for prescriptions was highly desired. RELEVANCE TO POLICY: These results suggest that patients can understand the need for limiting resource use and would rather not defer these decisions entirely to policy makers. Care must be taken in designing benefit packages, requiring, at minimum, coverage of certain core services. (HealthSTAR)
Davis M. Poisal J. Chulis G. Zarabozo C. Cooper B. (1999) Prescription Drug Coverage, Utilization, and Spending Among Medicare Beneficiaries. Health Affairs, 18(1): 231-43.
Outpatient prescription drugs are not a covered benefit under Medicare. There have been proposals in the past to expand Medicare benefits to include drug coverage, and current discussions dealing with "modernizing" the Medicare benefit package have raised the issue again. Using data from the 1995 Medicare Current Beneficiary Survey (MCBS), we describe the sources and extent of drug coverage among Medicare beneficiaries. The data show that 65 percent of Medicare beneficiaries have some level of drug coverage--a figure much higher than previous published numbers--and that 95 percent of Medicare health maintenance organization (HMO) enrollees have drug coverage. The data provide a baseline to observe future changes in the level of coverage, particularly among Medicare managed care plans.
Dickson M, Redwood H. (1998) Pharmaceutical Reference Prices. How Do They Work in Practice? Pharmacoeconomics, 14(5): 471-9
Reference pricing systems are reimbursement ceilings set by payers in an effort to constrain pharmaceutical expenditure for a private or public drug benefit. In recent years, many governments have adopted reference pricing either as a replacement or in addition to product specific price controls. Program administrators should consider whether these policies are providing the intended benefits or whether there may be a more effective method. This article provides a review of reference pricing in Europe, North America and other countries. There are many similarities in the reference price policies but the markets to which they apply are more likely to be different. The European experience gives a 'once-for-all' lowering effect on pharmaceutical expenditure, often at the expense of compromises on prescribing. In Germany and The Netherlands, reference pricing has been relatively ineffective in lowering expenditure which has led to a succession of other interventions to achieve expenditure control goals. The US also has reference pricing, but it occurs in a very competitive market which may be responsible (at least in part) for the relatively modest growth in expenditure compared with European countries. The review of countries with reference pricing policies suggests that such policies are less effective than competitive markets in moderating pharmaceutical expenditure. Nonetheless, governments continue to pursue reference pricing strategies.
Ganther JM. Kreling DH. (1999) Effect of a Change in Third Party Reimbursement Rate on Prescription Gross Margin. Journal of the American Pharmacy Association, (Wash) 39(3): 346-52
OBJECTIVE: To measure the effect of a change in an insurance company's reimbursement formula on prescription department gross margins for all prescriptions and subgroups of prescriptions. DESIGN: Retrospective descriptive analysis. SETTING: Wisconsin. PARTICIPANTS: Two units of a chain pharmacy. INTERVENTION: Reimbursement changed from usual and customary price to average wholesale price less 10% plus a $2.00 dispensing fee for single-source products, and maximum allowable cost plus a $2.00 dispensing fee for multisource products. MAIN OUTCOME MEASURE: Gross margins for prescriptions dispensed in the month before and after the reimbursement change. RESULTS: The average estimated gross margin decreased 26.9% after the change in reimbursement, and the effect on the average gross margin for generic prescriptions was nearly twice that of the effect on the average gross margin for brand name prescriptions. The effect of the reimbursement change on different therapeutic classes ranged from an increase of 0.7% in the cardiovascular class to a decrease of 68.2% in the eyes, ears, nose, and throat class. The effect of the reimbursement change was greater for low-cost prescriptions than for high-cost prescriptions. CONCLUSION: The large effect of the reimbursement change, combined with continued growth in third party prescriptions, raises concerns about whether pharmacies can accept third party contracts with low reimbursement rates and still maintain current profitability and service levels.
Grabowski H. Mullins CD. (1997) Pharmacy Benefit Management, Cost-Effectiveness Analysis and Drug Formulary Decisions. Social Science Medicine, 45(4): 535-44.
Pharmacy benefit management companies (PBMs) have evolved over the past decade in response to the increased demand for health care cost containment. Their activities include the implementation of drug formularies and the negotiation of rebates from manufacturers. Our analysis of this industry is based on interviews and materials provided by the top five ranked PBM companies which account for over 80% of beneficiaries covered within formulary plans. The formularies of these companies are relatively inclusive, but they are becoming more restrictive over time. At present the use of cost-effectiveness (C-E) studies in the formulary decisions of PBMs has been limited. In this regard, the surveyed PBMs emphasized that most C-E studies have not compared therapeutic substitutes in populations with characteristics that are similar to those of their clients. Pharmacy benefit management companies also have had strong incentives to focus narrowly on drug costs because they typically manage drug benefits on a "carved-out" basis. However, PBMs anticipate a growing future role in the integrated management of patient care (disease management) for certain high cost chronic diseases and conditions. All of the leading firms we surveyed have disease management programs in development. The importance of C-E studies to PBM decisions is expected to increase significantly as disease management programs are implemented. The data infrastructure inherent to the PBM industry and the increasing number of employees with advanced training in pharmacoeconomics will permit firms to perform their own internal C-E studies. They are also establishing various alliances and joint ventures with drug manufacturers, health maintenance organizations, and academic institutions to perform these analyses. The leading PBMs tend to favor active participation in the development of methodological approaches to C-E studies over government regulations such as those proposed by the FDA in 1995.
Gross DJ. Alecxih L. Gibson MJ. Corea J. Caplan C. Brangan N. (1999) Out-of-Pocket Health Spending by Poor and Near-Poor Elderly Medicare Beneficiaries. Health Services Research, 34(1 Pt 2): 241-54.
OBJECTIVE: To estimate out-of-pocket health care spending by lower-income Medicare beneficiaries, and to examine spending variations between those who receive Medicaid assistance and those who do not receive such aid. DATA SOURCES AND COLLECTION: 1993 Medicare Current Beneficiary Survey (MCBS) Cost and Use files, supplemented with data from the Bureau of the Census (Current Population Survey); the Congressional Budget Office; the Health Care Financing Administration, Office of the Actuary (National Health Accounts); and the Social Security Administration. STUDY DESIGN: We analyzed out-of-pocket spending through a Medicare Benefits Simulation model, which projects out-of-pocket health care spending from the 1993 MCBS to 1997. Out-of-pocket health care spending is defined to include Medicare deductibles and coinsurance; premiums for private insurance, Medicare Part B, and Medicare HMOs; payments for non-covered goods and services; and balance billing by physicians. It excludes the costs of home care and nursing facility services, as well as indirect tax payments toward health care financing. PRINCIPAL FINDINGS: Almost 60 percent of beneficiaries with incomes below the poverty level did not receive Medicaid assistance in 1997. We estimate that these beneficiaries spent, on average, about half their income out-of-pocket for health care, whether they were enrolled in a Medicare HMO or in the traditional fee-for-service program. The 75 percent of beneficiaries with incomes between 100 and 125 percent of the poverty level who were not enrolled in Medicaid spent an estimated 30 percent of their income out-of-pocket on health care if they were in the traditional program and about 23 percent of their income if they were enrolled in a Medicare HMO. Average out-of-pocket spending among fee-for-service beneficiaries varied depending on whether beneficiaries had Medigap policies, employer-provided supplemental insurance, or no supplemental coverage. Those without supplemental coverage spent more on health care goods and services, but spent less than the other groups on prescription drugs and dental care-services not covered by Medicare. CONCLUSIONS: While Medicaid provides substantial protection for some lower-income Medicare beneficiaries, out-of-pocket health care spending continues to be a substantial burden for most of this population. Medicare reform discussions that focus on shifting more costs to beneficiaries should take into account the dramatic costs of health care already faced by this vulnerable population. (Medline)
Hillman A. Pauly M. Escarce J. Rippley K. Gaynor M. Clouse J, Ross R. (1999) Financial Incentives and Drug Spending in Managed Care. Health Affairs. 18(2): 189-200.
This study estimates the impact of patient financial incentives on the use and cost of prescription drugs in the context of differing physician payment mechanisms. A large data set was developed that covers persons in managed care who pay varying levels of cost sharing and whose physicians are compensated under two different models: independent practice association (IPA)-model and network-model health maintenance organizations (HMOs). Our results indicate that higher patient copayments for prescription drugs are associated with lower drug spending in IPA models (in which physicians are not at risk for drug costs) but have little effect in network models (in which physicians bear financial risk for all prescribing behavior).
Johnson RE. Goodman MJ. Hornbrook MC. Eldredge MB. (1997) The Impact of Increasing Patient Prescription Drug Cost Sharing on Therapeutic Classes of Drugs Received and on The Health Status of Elderly HMO Members. Health Services Research. 32(1):103-22.
OBJECTIVE: To assess the impact of increased prescription drug copayments on the therapeutic classes of drugs received and health status of the elderly. HYPOTHESES TESTED: Increased prescription drug copayments will reduce the relative exposure to, annual days use of, and prescription drug costs for drugs used in self-limiting conditions, but will not affect drugs used in progressive chronic conditions and will not reduce health status. STUDY DESIGN: Each year over a three-year period, one or the other of two well-insured Medicare risk groups in an HMO setting had their copayments per dispensing increased. Sample sizes ranged from 6,704 to 7,962. DATA SOURCES/DATA COLLECTION: Automated administrative data systems of the HMO were used to determine HMO eligibility, prescription drug utilization, and health status. ANALYSIS DESIGN: Analysis of variance or covariance was employed to measure change in dependent variables. FINDINGS: Relative exposure, annual days of use, and prescription drug costs for drugs used in self-limiting conditions and in progressive chronic conditions were not affected in a consistent manner across years by increases in prescription drug copayment. Health status may have been adversely affected. Larger increases in copayments appeared to generate more changes. CONCLUSIONS: Small changes in copayments did not appear to substantially affect outcomes. Large changes in copayments need further examination.
Lecomte T. Paris V. (1998) Controlling Drug Expenditures in France, Germany, and the United Kingdom. From Économie et Statistiques, no. 312-313 Insee Studies no. 30.
In the 1990s, France, Germany, and the United Kingdom have all tested procedures to curb the growth of public expenditures on pharmaceuticals. Their diversity reflects the major differences between the three countries' healthcare systems. France combines a consensual regulation of drug prices with incentives to foster greater restraint in doctors' prescriptions (through the guidelines known as Références Médicales Opposables). Germany has introduced competitive mechanisms via regulations based on groupings of equivalent products. In the United Kingdom, a series of reforms has opened up the traditionally State-controlled healthcare sector to market rules aimed at promoting cost-conscious behavior among system players. The main innovation consists in giving doctors the opportunity to manage a budget tailored to the size of their practice. The success of these policies varies considerably from one country to another. France combines a higher individual expenditure with a lower co-payment by the State. The national differences concern not only the overall consumption level, but also the type of medicine: the French consume more anti-anxiety drugs, while the British consume more analgesics. Consumption volume appears to be a more powerful engine of spending growth than prices.
Lillard LA. Rogowski J. Kington R. (1999) Insurance Coverage for Prescription Drugs: Effects on Use and Expenditures in the Medicare Population. Medical Care, 37(9): 926-936.
BACKGROUND: Although Medicare covers most of the elderly, they potentially face large out-of-pocket costs for their health care because of excluded services. Aside from nursing home care, the exclusion of prescription drugs is one of the most significant. Several earlier policy initiatives have proposed adding prescription drug coverage to the Medicare program. To determine the effects of such an expansion, one must account for the potential increase in the demand for prescription drugs from providing insurance coverage. METHODS: The study uses a new data source, the RAND Elderly Health Supplement to the 1990 Panel Study of Income Dynamics (PSID). The endogenity of insurance coverage is tested using instruments that exploit the longitudinal nature of the data. Equations are estimated on 910 persons (> or = 66 years) using a two-part model. RESULTS: Insurance coverage for prescription drugs significantly increases the probability of use, but not of total expenditures, among those who use prescription drugs. However, insurance coverage significantly lowers out-of-pocket expenditures, thereby decreasing the financial burden on elderly households associated with prescription drug use. Medicaid coverage has effects that are smaller than those for private insurance do, but the magnitude is less precisely estimated. These findings imply that if prescription drug coverage were added to Medicare, expected expenditures on drugs would rise by on average $83 for each elderly Medicare beneficiary (in 1990 dollars), although this increase is significant only at the 90% level. If the benefit had been included under Medicare expected spending on prescription drugs by the elderly would have risen by approximately 20%, or $2.6 billion in 1990. (Medline)
López-Casasnovas G. and Puig-Junoy J. (1999) Review of the Literature on Reference Pricing. Research Center on Health and Economics. Department of Economics and Business. Universitat Pompeu Fabra. Barcelona Spain. February.
This paper reviews the literature on reference pricing (RP) in pharmaceutical markets. The RP strategy for cost containment of expenditure on drugs is analyzed as part of the procurement mechanism. We review the existing literature and the state-of-the-art regarding RP by focusing on its economic effects. In particular, we consider: (1) the institutional context and problem-related factors which appear to underline the need to implement an RP strategy; i.e., its nature, characteristics and the sort of health care problems commonly addressed; (2) how RP operates in practice; that is, how third party-payers (the insurers/buyers) have established the RP systems existing on the international scene (i.e., information methods, monitoring procedures and legislative provisions); (3) the range of effects resulting from particular RP strategies (including effects on choice of appropriate pharmaceuticals, insurer savings, total drug expenditures, prices of referenced and non-referenced products and dynamic efficiency; (4) the market failures which an RP policy is supposed to address and the main advantages and drawbacks which emerge from an analysis of its effects.
Lyles A. Palumbo FB. (1999) The Effect of Managed Care on Prescription Drug Costs and Benefits. Pharmacoeconomics, 15(2):129-40.
This review discusses the approaches to prescription drug payment practices taken by managed care to influence drug use and costs, and presents the research evidence supporting these interventions. In the US, drugs were infrequently covered as an ambulatory benefit under fee-for-service indemnity insurance; however, health maintenance organizations almost always provide outpatient drugs and consequently have developed approaches to influence drug use and manage its costs. Managed care as a set of tools and as an organizational form is moving toward more restrictions on direct access to pharmaceuticals as a covered benefit. Options for influencing drug use and cost may address access, ingredient costs, dispensing fees and cost sharing. The formulary process is the foundation for a managed pharmacy benefit and integrates these options. The limited empirical evidence for an effect of managed care on drug costs and use is reviewed. A proposed research agenda includes evaluation of the effects of restrictive formularies, capitation, disease management and other programs to influence the cost and use of pharmaceuticals.
McArdle F. Copock S. Yamamoto D. et al. (1999) Retiree Health Coverage: Recent Trends and Employer Perspectives on Future Benefits. The Henry J. Kaiser Family Foundation: Menlo Park, CA.
This study uses the Hewitt Associates client database of large employers’ benefits plans to show trends in retiree health plans. The analysis showed that the overall health insurance coverage has declined between 1991 to 1998: there was a 13-percentage point drop in portion of large employers offering health coverage to age 65+ retirees. More than 95% of large employers offer prescription benefits to retirees. Prescription drug expenditures account for 40 to 60 percent of retiree plan costs. (Staff)
Poisal JA. Murray LA. Chulis GS. Cooper BS. (Spring 1999) Prescription Drug Coverage and Spending for Medicare Beneficiaries. Health Care Financing Review, 20(3):15-27.
Very little is known about which persons within the Medicare population have drug coverage from other sources. Using 1995 data from the Medicare Current Beneficiary Survey (MCBS), the authors present information on who has coverage by various sociodemographic categories. The data indicate higher-than-average levels of coverage for minority persons, beneficiaries eligible for Medicare because of disability, and those with higher incomes. (HealthSTAR).
Rowgowski J. Lillard LA. Kington R. (1997) The Financial Burden of Prescription Drug Use Among Elderly Persons. Gerontologist, 37(4): 475-82.
This study, using a new data source, the 1990 Elderly Health Supplement to the Panel Study of Income Dynamics (PSID), demonstrates that, among elderly persons, insurance coverage for drugs reduces the fraction of household income spent on prescription drugs by 50 percent. Groups most likely to benefit from insurance coverage are elderly women and those with common chronic conditions, low incomes, and rural residences. (Medline)
Sager A. Socolar D. (1999) Affordable Medications for Americans: Problems, Causes and Solutions. Prescription Drug Task Force. United States House of Representatives. Washington D.C.
The authors define certain existing problems with the high costs of prescription drugs and make recommendations to redistribute financial resources in order to make prescription drugs more cost effective and accessible to the American public. According to the report, ¼ of all Americans do not have prescription drug coverage. Yet, current expenditures on prescription drugs in the U.S. are more than sufficient to purchase all the medications that Americans need. U.S. retail prescription drug spending is predicted to rise to $103 billion in 1999 and $143 billion by 2002. The cost of prescription drugs is rising about three times as fast as overall health costs. According to the authors, reasons for such high expenditures include: lack of government regulation of drug pricing activities; drug manufacturers charge Americans higher prices than they do to other countries. They assert that it may be a myth that the funds are needed for research activities. The costs of pharmaceutical research ought to be shared globally. Pharmaceutical drugs in 1998 were the most profitable industry, and maintained remarkably high profits for seven decades. The profits of this industry exceed research costs in many of the top 10 U.S. drug firms. Some solutions recommended by the authors include: 1) lowering drug prices to raise demand, thus offsetting some revenue loss for drug makers, 2) pooling statewide buying power to negotiate discounts and rebates with drug manufacturers, 3) federal and state governments to negotiate “in-kind’ donations with drug manufactures, 4) partnering public and private efforts to more efficiently manage expenditures related to drug manufacturers’ advertising, public relations, and lobbying, and 5) financing should be planned cooperatively among all stakeholders. In light of these recommendations, the authors advocate for comprehensive reform of the prescription drug manufacturing industry in order to contain expenditures and make prescription drugs more affordable and accessible to the American public. (Staff)
Schulman KA. Rubenstein LE. Abernethy DR. Seils DM. Sulmasy DP. (1996) The Effect of Pharmaceutical Benefits Managers: Is It Being Evaluated? Annals of Internal Medicine, 124(10): 906-13 .
Over the last decade, the number of pharmaceutical benefits managers has increased, and their influence has expanded rapidly. These managers now provide prescription drug coverage to more than 100 million Americans. The effect of pharmaceutical benefits managers on health care delivery remains unclear. We review the development of these organizations, their current role in the delivery of pharmaceutical therapies to patients, and their relationship with pharmaceutical manufacturers. We discuss potential advantages and disadvantages of pharmaceutical benefits manager practices and suggest ways in which these organizations can be made more accountable to the employer groups that hire them.
Stuart B. Grana J. (1998) Ability to Pay and The Decision to Medicate. Medical Care. 36(2): 202-211.
OBJECTIVES: It is widely recognized that ability to pay affects access to hospital and physician services. Much less is known about the economic determinants of prescription drug use, particularly among the elderly. The authors hypothesize that persons with higher incomes and better health insurance coverage are more likely to medicate common health problems than those with lower incomes and less comprehensive coverage. METHODS: A random sample of 4,066 elderly Pennsylvania Medicare beneficiaries were asked to complete a mail survey on health insurance, income, and medicine use for 23 common health problems. The relationship between ability to pay and medication decisions was analyzed using logistic and Poisson regression models with covariates for socio-demographic characteristics and health status. RESULTS: A strong and consistent relationship was found in the hypothesized direction. Other things being equal, elderly persons with Medicare supplementation were between 6% and 17% more likely to use prescription medicine to treat their health problems than are persons with Medicare coverage alone. The presence of prescription drug coverage significantly increased the odds of prescription treatment for 10 of the 22 conditions examined. The insurance effects were generally--but not exclusively--more pronounced for less serious compared with serious health problems. Income also was shown to have a strong independent effect on medication decisions. Elderly with annual incomes greater than $18,000 were 18% more likely to treat problems with prescription drugs than were persons with annual incomes less than $6,000. CONCLUSIONS: In sum, economic factors appeared to play an important role in medication decisions by the elderly. The magnitude of the impact was sufficiently high that it could have major negative consequences on the health of elderly persons who are poor and lack drug coverage. (Medline)
Stuart B. Grana J. (1995) Are Prescribed and Over-the-Counter Medicines Economic Substitutes? A Study of the Effects of Health Insurance on Medicine Choices by the Elderly. Medical Care, 33(5):487-501.
Insurance coverage on the selection of over-the-counter (OTC) and prescribed (Rx) medicines in treating less serious health problems. Because health insurance policies typically provide no coverage for OTC products, a low list price for an OTC may exceed the after-insurance expense associated with a much higher-priced prescription. Under these circumstances, rational individuals with insurance will choose prescribed medicines even if OTCs are equally effective. Ten common health problems typically managed with either Rx or OTC medicines were selected for analysis. The study population consists of elderly Pennsylvanians surveyed during 1990 who reported suffering one or more of these conditions (N = 2,962). Multivariate analysis confirmed that 1) people with prescription coverage are significantly more likely to medicate a given problem than are those without it; and 2) given the decision to medicate, the presence of insurance significantly increases the level of Rx use and significantly reduces the level of OTC use. As expected, the effect was strongest among people with the most complete prescription insurance coverage. The article discusses the implications of these findings in the context of national health reform and Food and Drug Administration policy regarding Rx-to-OTC switches. (Medline)
United States Department of Health and Human Services Office of Inspector General. (1997), Experience of Health Maintenance Organizations with Pharmacy Benefit Managment Companies. Washington, D.C. Government Document #OEI-01-00110.
This report focuses on the experience of HMO’s in using Pharmacy Benefit Managers (PBMs). As enrollment in managed care continue to grow, and because PBMs can significantly affect patients’ use of prescription drugs, it is important for the Health Care Financing Administration(now known as Centers for Medicare and Medicaid Services(CMS)) (HCFA(now known as CMS)), as well as private payers, to be informed about the HMOs experiences with them. The data used in this report are derived from a mail survey of all HMOs in the country. The response rate was 71%. Information collected also drew from discussions with staff of the U.S. Department of Health and Human Services (HHS) and several state Medicaid agencies, non-government experts, and a literature review. The report had three recommendations: 1) the HCFA(now known as CMS) should take steps to ensure that its Medicare HMOs are sufficiently accountable for the quality of the services their PBMs provide to beneficiaries, 2) State Medicaid agencies should take steps to ensure that their Medicaid HMOs are sufficiently accountable for the quality of the services their PBMs provide to beneficiaries, and 3) HCFA(now known as CMS), the Food and Drug Administration (FDA), and the Health Resources and Services Administration (HRSA), should work together with external organizations to develop quality measures for pharmacy practice that can be used in managed care settings. (Staff)
U.S. General Accounting Office (1996), Pharmacy Benefit Managers, Early Results on Ventures with Drug Manufacturers, Washington D.C. Report Number GAO-96-45
This GAO report responds to congressional concerns over recent trends among the largest pharmaceutical manufacturers in merging or forming alliances with some of the largest pharmacy benefit management companies (PBMs). Questions raised by the study focused on four general areas including: 1) the role of the PBMs in the health care industry; 2) the objectives of these ventures; 3) specific concerns about the effect of these ventures on competition in markets served by drug manufacturers and PBMs; and 4) the extent, if any, to which the PBMs have given preference to their manufacturer partners’ drugs after a merger. Information was collected though discussions with Wall Street analysts, pharmaceutical economists, health plan sponsors, pharmaceutical trade associations, and PBMs. The results indicate that drug manufacturer have merged or allied with PBMs to help maintain and/or increase the manufacturers’ profits at a time of increasing competition. It is believed that PBMs have substantial market power due to their increasing special role in health care by administering prescription drug benefits for health plan sponsors. They also have been able to affect substantial cost savings through their ability to negotiate drug discounts and rebates from both drug manufacturers and pharmacies. The objective of the manufacturers’ ventures with PBMs was to bolster profits, increase market share for their drugs, and develop new programs for treating specific diseases. While the number of people covered by PBMs has increased significantly, the market for PBMs services continues to involve a small number of firms. Although there are over 40 PBMs in the U.S., the 5 largest manage benefits for over 80% of the health plan enrollees covered by PBMs. They include PCS Health Systems, Medco, Value RX, DPS, and Caremark International Inc.’s Prescription Service Division. (Staff)
U.S. General Accounting Office (1999) Prescription Drug Benefits - Implication for Beneficiaries of Medicare HMO Use of Formularies. Washington D.C. Report Number GAO/HEHS-99-166.
This GAO report is a study of the impact of drug formularies on beneficiaries in Medicare HMOs. Medicare HMO enrollees face complex issues in selecting a plan to best meet their needs because formularies within different HMOs vary and can affect their drug benefits as well as their out-of-pocket costs. Plans are not required by HCFA(now known as CMS) to notify beneficiaries of formulary changes and may also make it difficult for physicians to obtain exceptions to allow patients to remain on their existing medication at no additional cost if it is dropped from the formulary. Ten of the 16 HMOs studied use closed formularies that limit coverage to certain drugs. Twelve of the 16 HMOs require the use of generic drugs when available. Seven of the 16 use variable co-payments, with a larger amount charged for brand-name drugs than for the generics. Currently, Medicare beneficiaries can join or leave an HMO plan on a monthly basis. However, in 2002, making informed choices among health plans will become even more important because under the Balanced Budget Act of 1997 (BBA) (Staff)
U. S. General Accounting Office (1997), Pharmacy Benefit Managers: FEHBP Plans Satisfied with Savings and Services, but Retail Pharmacies Have Concerns. Washington D.C. Report Number GAO –97-47
The Office of Personnel Management (OPM) estimates that nearly 9 million federal employees, retirees, and dependents are covered by a Federal Employees Health Benefits Program (FEHBP). Additionally, they estimated that pharmacy benefit payments for the five largest FEHBP plans amounted to about $2 billion in 1995, and accounted for an increasing share of the total FEHBP health care costs –growing from 12% in 1990 to 19% in 1995. In order to control prescription drug costs, FEHBP plans contract with PBMs to manage pharmacy benefits on behalf of plan sponsors. As a result, the role of PBMs in managing pharmacy benefits for federal enrollees is growing. The report raised four basic questions: (1) why FEHBP plans have contracted with PBMs to provide pharmacy benefits, (2) what types of services and savings the PBMs provide FEHBP plans, (3) how FEHBP plans evaluate PBM customer service, and (4) the concerns of retail pharmacists about the quality of PBM pharmacy services and the effect of some PBM practices on the retail pharmacy business. The range of cost control services offered by PBMs to FEHBP plans include negotiating lower prices on prescription drugs, developing mechanisms that support lower drug costs such as mail order pharmacy sales, checking prescriptions for adverse drug reactions, and customer services, etc. Reimbursement mechanisms employed by PBMs include industry standards such as the drug’s usual and customary price, average wholesale price (AWP), or maximum allowable cost (MAC), plus a dispensing fee. The PBMs also require network pharmacies to substitute generic drugs for brand name drugs when possible. In directing market share of certain drug formularies, manufacturers give rebates to the PBMs amounting to over $113 million collectively in 1995. These rebates accounted for between 2% and 21% of plans’ estimated savings. Because the FEHBP plans use open formularies, enrollee reimbursement is not limited to the drugs listed on the formularies. Other cost saving interventions used by the PBMs include drug utilization review (DUR), generic and therapeutic interchange programs, and disease management programs. (Staff)
U.S. House of Representatives, Committee on Government Reform and Oversight, Minority Staff Report, (1998). Prescription Drug Pricing in the United States: Drug Companies Profit at the Expense of Older Americans. Washington D.C.
This report summarizes investigations of prescription drug pricing conducted by the minority staff in 20 congressional districts. The findings indicate that many older Americans pay high prices for prescription drugs and have a difficult time paying for the drugs they need. Large corporate and institutional customers with market power are able to purchase at discounted prices, but seniors and individual customers who pay for drugs out of pocket must pay at a much higher price. By one estimate, more than one in eight older Americans has been forced to choose between buying medications and food. Case studies conducted in several states illustrate these hardships. The staff recommendations include legislation to reduce the cost of prescription drugs for seniors. (Staff)
Walser BL. Ross-Degnan D. Soumerai SB. (1996) Do Open Formularies Increase Access to Clinically Useful Drugs? Health Affairs. 15(3): 95-109.
Before 1990 many state Medicaid programs maintained "restrictive" formularies, which denied reimbursement for unlisted prescription drugs. This type of formulary has been criticized for denying important medications to poor, medically needy persons. As part of the Omnibus Budget Reconciliation Act of 1990, restrictive formularies in Medicaid programs were disallowed. Based on research into the 200 top-selling prescription drugs in the United States, we conclude that eliminating Medicaid restrictive formularies improved access to a subset of the 200 best sellers, but that the majority of these products offered only questionable or no additional therapeutic benefit.
Endnotes
1 - The true expenditure levels may be higher than this figure considering the possibility of under-reporting of prescription drug use in consumer surveys.
2 - An example of drugs within the same chemical class would be all brands of Tagamet® available on the market. An example of drugs within the same drug class would be all H2 antagonists such as Zantac® and Tagamet®. An example of different drugs used to treat the same medical condition would be all drugs used to treat ulcers such as Zantac®, Tagamet®, Prilosec®and Prevacid®. These drugs have similar effects but they use different methods to effect change. Under reference pricing would be considered equivalent (Narine et al. 1999).
Appendix B. Development of Mcbs and Meps Estimates for This Report
This report uses 1996 Medicare Current Beneficiary Survey (MCBS) data to estimate prescription drug coverage, utilization, and spending for Medicare beneficiaries. 1996 Medical Expenditure Panel Survey (MEPS) data are used to provide estimates for the non-Medicare population. The MEPS surveyed a sample of the US civilian, noninstitutionalized population, including Medicare beneficiaries. MEPS data could therefore have been used for both the Medicare and non-Medicare groups.
There are several reasons for using the MCBS data instead of MEPS for the Medicare population. MCBS has a much larger sample of Medicare beneficiaries than MEPS, 10,869 used for this study, as opposed to 2,892 in the MEPS panel. This allows more reliable estimates for subgroups of the Medicare population defined by demographic or health-related factors. In addition, MCBS has been conducted continuously since 1992. It therefore allows some analysis of trends over time. MEPS began in 1996; its predecessor, the 1987 National Medical Expenditure Survey (NMES) was so different in its data collection for prescription drug spending and coverage that meaningful comparisons are not possible. The one exception where MEPS was used instead of MCBS for estimates relating to Medicare beneficiaries is the analysis of prices in Chapter 3. As discussed below, MEPS had data not available in MCBS for comparing drug prices.
Data collection and estimation procedures under the two surveys are different in a number of respects. This means that, while the MCBS results for the Medicare population and the MEPS results for the non-Medicare population are often quite similar, they cannot be directly compared. In addition, MCBS estimates for the Medicare population are different from the Medicare estimates that would have been produced if MEPS had been used. Use of MEPS results would not have affected the most important findings of this report: that a large share of the Medicare population is without drug coverage, that beneficiaries without coverage receive fewer and less costly prescription drugs, and that they pay higher prices for the drugs they receive than are paid by beneficiaries with coverage for the same drugs. Estimates of the magnitude of these effects would, however, have been different. The following is a brief overview of how coverage, utilization, and spending are estimated under MCBS and MEPS, and of how their findings differ.
Establishing Coverage
Both the MCBS and the MEPS household survey asked participants if they had prescription drug coverage. MEPS had a second source of information: for respondents who named the pharmacies they used and gave interviewers permission to contact the pharmacy (the pharmacy follow-back), it was possible to determine the sources of payment for prescriptions filled at participating pharmacies. Sometimes, when an individual reported that he or she did not have drug coverage, it was nevertheless found through the pharmacy component that an insurer or public program had in fact paid for one or more prescriptions. In this case, the person was deemed to have drug coverage through the source of payment for the prescription.
MCBS did not collect information from pharmacies, but did collect information from respondents about each of the prescriptions they received during the year and the sources of payment for those prescriptions. If an individual reported no drug coverage but nevertheless reported a third-party payment for a prescription, the individual was assigned to drug coverage. However, because MCBS relied almost entirely on respondents’ own reports,1 it found fewer such people with coverage than MEPS found through its separate pharmacy component.
A second key difference between MEPS and MCBS is in the treatment of Medicaid beneficiaries and Medicare HMO enrollees. Some Medicaid beneficiaries are QMBs and SLMBs without a drug benefit, and not all Medicare HMOs provide drug coverage. MEPS is not able to distinguish whether Medicare HMO enrollees or Medicaid recipients have coverage; all of these beneficiaries are deemed to have coverage in MEPS. For these populations, MCBS distinguishes among those who did and did not have a drug benefit. In the future, it may be possible to use Medicare administrative data on health plan enrollment to determine drug coverage among Medicare HMO enrollees surveyed in the MEPS.
Finally, MCBS uses a consistency edit that treats as noncovered those beneficiaries who reported having drug coverage but paid more than $250 out of pocket for drugs and reported no insurance payment, while MEPS leaves these beneficiaries in the covered category.
Table B-1 shows the proportion of Medicare beneficiaries with different types of drug coverage for at least one month as estimated by MEPS and MCBS. The MEPS numbers have been adjusted to account for the difference between MEPS and MCBS in the treatment of Medicaid recipients and Medicare HMO enrollees, and to account for the consistency edit that MCBS uses and MEPS does not use.2 The adjustments are: a .8% percent reduction from employer and a .3% reduction from nongroup for the “consistency edit” and a .6% reduction in Medicare Risk HMO and a 1.4% reduction in Medicaid to account for the proportion of beneficiaries in those categories who lack drug coverage. The lower bound of the 95% confidence interval for the adjusted MEPS estimate is 71.6 percent of beneficiaries with coverage. The upper bound of the MCBS estimate is 69.7 percent. A statistically significant difference, therefore, remains between the MCBS estimate and the adjusted MEPS estimate.
| MEPS | MCBS | |
|---|---|---|
| Employer | 33% | 32.3% |
| Private nongroup and other private | 13.2% | 11.3% |
| Medicare risk HMO | 10.4% | 10.0% |
| Medicaid | 10.9% | 11.8% |
| Veterans and other | 6.1% | 3.3% |
| Total with coverage for at least one month during 1996 |
73.6% | 68.8% |
| None | 26.4% | 31.2% |
| Total | 100.0% | 100.0% |
|
Note: Percents may not add to 100 because of rounding. Source: Center for Cost and Financing Studies, Agency for Healthcare Research and Quality: Medical Expenditure Panel Survey Household Component, 1996 and Health Care Financing Adminstration, Medicare Current Beneficiary Survey, 1996. |
||
Much of the remaining difference is due to the larger number of beneficiaries who failed to report coverage but were identified as covered by MEPS through the pharmacy follow-back. Most of the beneficiaries assigned to the covered category in this way were classified as either having private nongroup insurance or as being covered through the Veterans Administration.
As was noted in chapter 1, the VA does not actually provide insurance. It supplies prescription drugs directly, at no cost or nominal cost, to qualifying veterans whose prescriptions are written by VA physicians and filled at VA pharmacies. While this benefit is undoubtedly an important source of financial assistance for those who receive it, it cannot be described as insurance in the same sense as other forms of drug coverage. However, it is counted as coverage for the purposes of this study. Private nongroup coverage is insurance, but it is the least generous and the most costly to beneficiaries of the major types of coverage. Moreover, it is the least stable: as table 1-2 indicated, beneficiaries with this type of coverage are the most likely to be without protection at some time during the year.
In summary, after correction for the likely overstatement of Medicare HMO and Medicaid drug coverage in MEPS, there remains a small but statistically significant difference in the MEPS and MCBS estimates of the extent of drug coverage in the Medicare population. Much of the difference is attributable to estimates for types of coverage that may not be meaningful or stable. The MCBS data are used in this report because they allow a closer look at the characteristics of the covered and noncovered populations and at trends over time.
Estimates of Utilization and Spending
Gathering information through household surveys about prescription drug use and spending is much more difficult than for some other kinds of medical services. While most people can readily recall, for example, whether they were hospitalized in the last year and for what reason, filling a prescription is not so memorable an event. People who filled only one or two prescriptions may not remember them at all. Those who had many medications may not recall just how many they had or may not be able to name every drug they were taking. Even people who can accurately report what drugs they used may not be able to tell what those drugs cost. This is especially true when the drug cost is covered by an insurer or a public program; the respondent may know only the amount of his or her required copayment, not the total charge.
The MEPS and MCBS surveys use a variety of techniques to overcome these barriers to collection of complete and accurate information about prescription drugs. The following is a brief overview of how the two surveys go about answering two key questions:
- How many prescriptions did participants fill during the period covered by the survey?
- What was paid for those drugs, including both out-of-pocket payments by the respondents and payments by insurers or other third parties?
Utilization Estimates
The MCBS and the MEPS household component both involve multiple rounds of interviews with each respondent. In each interview, the respondent is asked about services used in the period since the last interview. MEPS separately surveys pharmacies named by respondents, and can thus supplement the information gathered in the household survey. Because MCBS has no pharmacy component, it must collect all utilization and price information through the household survey.
MCBS uses a variety of devices to promote fuller reporting by participants. Between interviews, participants are asked to keep track of the services they used on a calendar and to retain bills, receipts, check stubs and other relevant documentation. For a filled prescription they are asked to save and bring to the interview the actual bottle, the package it came in, and any receipts or other statements they received with the prescription. To further aid recall, the interviewer in each round has information about the drugs reported in the previous interview, and thus can ask whether the respondent is still taking the same drugs. Still, the information is probably incomplete. Some respondents may not keep the requested records, or the proxy who actually participates in the survey for individuals unable to be interviewed may not have full information.
MEPS collects less information through the household survey. For example, charge and payment data are not obtained from participants who report that their insurance pays the pharmacy directly. Instead, MEPS obtains some information through the pharmacy component. Each participant who reports having a prescription is asked to identify the pharmacy that filled it and to give permission for that pharmacy to share information. Permission was obtained for 73 percent of possible person-pharmacy pairs, and pharmacies responded to data requests for 67 percent of these pairs. Thus there is information from pharmacies for about half the instances in which a participant named a specific pharmacy.
The pharmacy component provided more detailed data on specific drugs used and on their costs, but it did not actually increase MEPS estimates of total drug utilization. It was expected that pharmacies would sometimes report filling more prescriptions for an individual than the individual had reported in the household survey. While this sometimes occurred, aggregate utilization estimates derived from the household data alone are actually larger than those derived from the pharmacy survey alone. Thus there remains the possibility of some undercounting of total prescriptions. However, aggregate MEPS data benchmark fairly well to external data on prescription drug utilization.
The MEPS data, based on household responses supplemented with some reporting from pharmacies, can be compared to IMS data, which are based on pharmacy audits. MEPS estimates a total of 2.1 billion prescriptions, excluding free samples, for the noninstitutionalized population in 1996. IMS data show total pharmacy prescription volume of 2.41 billion, about 15 percent more. However, the IMS data include prescriptions for people in institutions, including the 1.6 million people who were in nursing homes in 1996 and who are often heavy users of prescription drugs. MEPS excluded people in institutions (along with individuals in the military, prisoners, and non-resident individuals). The difference in populations covered may account for much of the difference in total prescription counts.
For the Medicare population, MEPS reports 9.8 percent more prescriptions than does MCBS. The two surveys are almost identical in their estimates of the proportion of beneficiaries who received any prescription during the year; the difference is chiefly in their estimates of the annual number of prescriptions for each person who received any prescription drug. A portion of this difference is attributable to somewhat different treatment of diabetic supplies in MEPS and MCBS. The MEPS estimate includes expenditures for insulin and diabetic supplies totaling nearly $2 billion. MCBS includes insulin in prescription drug expenditures, but excludes diabetic supplies. Insulin purchases do not require a prescription, but a prescription is generally needed to receive third party payments.
Because the focus in this report is on relative utilization by the covered and noncovered populations, whether the MCBS prescription count is complete is less important than whether the possible undercount is more serious for one or the other of these groups. This is difficult to ascertain, however, because of the differences in assignment of coverage status between MEPS and MCBS. MCBS reports more prescriptions than MEPS for beneficiaries without coverage and fewer for beneficiaries with coverage. This is probably partially attributable to the fact that MCBS treats as noncovered some beneficiaries who used prescription drugs and would have been assigned coverage under MEPS on the basis of a third-party payment. Since people with coverage tend to have higher utilizaton than those without coverage, having peole with coverage in the noncovered category will tend to raise estimates of average spending and utilization for the noncovered category.
Spending Estimates
MEPS data for 1996 indicate that 27 percent of people who used prescription drugs paid for those drugs in full at the time they received them.3 Most people with public or private coverage pay only a copayment or coinsurance amount. (Exceptions include people who have not yet met a deductible or who have exceeded a cap on their coverage and people who have an indemnity plan, which reimburses the policyholder instead of the pharmacy.) As a result, household survey data alone can provide cost information for only a portion of all prescriptions.
Because MEPS has a pharmacy component, it can obtain full price information for prescriptions paid for by third parties. As noted earlier, however, pharmacy survey responses were not always available. When neither the individual nor a pharmacy provided price data for a prescription, the price had to be imputed through statistical matching: a total price was assigned to the prescription based on price data for the same drug furnished to a similar individual. There were also instances in which a total price was available for the prescription, but the amounts paid by the insurer and/or the individual were missing. Again, values were imputed for each such prescription.
MCBS also uses a process of imputation in cases in which household respondents were unable to supply price and payment information. Because MCBS has no pharmacy component, imputation is needed for more prescriptions than under MEPS, and the method of assigning prices is different.4 The MCBS average price per non-Medicaid prescription, $35.23, is quite close to the average price of $35.90 found by MEPS for Medicare beneficiaries without Medicaid.5 However, because MCBS counts fewer prescriptions per beneficiary using prescription drugs, its aggregate spending estimates are lower than MEPS estimates for Medicare beneficiaries.
Because the process of imputation leads to some potential measurement errors in comparing prices paid for a particular drug by individuals with different coverage statuses, all the price comparisons in chapter 3 are based on MEPS drug purchases for which the price was established through a direct match of pharmacy and household survey information. However, for the spending estimates in chapter 2, both the MCBS and the MEPS data used include prescriptions for which prices were imputed. Dropping all prescriptions for which prices were imputed would have left nationally unrepresentative populations and samples too small to estimate aggregate spending differences for different subgroups. Imputation is designed to generate accurate aggregate estimates, but may misrepresent pricing, especially for individual drugs.
It should be emphasized that all of the MEPS spending information, and most of that under MCBS, reflects only the amounts paid to pharmacies.6 These amounts are not adjusted for any rebates that may be paid by the manufacturer to the insurer, because these rebates are generally not reflected in the prices charged at the point of sale. There is one exception: under MCBS, but not MEPS, spending data for Medicaid beneficiaries are reduced to reflect rebates received from manufacturers by state Medicaid programs. Rebate estimates are derived from state financial reports to HCFA(now known as CMS). Thus, all other things being equal, we would expect aggregate spending estimates for MCBS to be somewhat lower than for MEPS.
Effects of Coverage Estimates
As was discussed earlier, MEPS produces higher estimates of Medicare beneficiaries who had drug coverage than MCBS for three basic reasons. First, MEPS assigns coverage to larger numbers of people who did not report having drug coverage but who were found to have a third-party payment for a prescription. Second, MEPS treats all Medicaid and Medicare risk HMO enrollees as having drug coverage, although in fact not all do. Third, MCBS screens out beneficiaries whose reported coverage is inconsistent with their payment history. Each of these differences has potential implications for estimates of relative spending by covered and noncovered beneficiaries.
While the availability of payment-source data from pharmacies increases the coverage estimates under MEPS relative to those under MCBS, the method necessarily misses people with drug coverage who failed to report it and who did not have any drug expenditure during the year. These people remain in the noncovered group, while the people assigned to coverage on the basis of a drug expenditure are by definition all utilizers of drugs. This biases utilization and spending estimates upward for the population treated as covered and downward for the noncovered group, widening differences between the two groups.
This effect is partially offset by the MEPS assignment of drug coverage to all Medicaid beneficiaries and Medicare HMO enrollees. MCBS finds lower utilization by beneficiaries in these groups who did not have a drug benefit. Their inclusion in the MEPS counts of covered people thus depresses the MEPS estimates of spending for people with coverage.
Finally, MCBS treats as noncovered those beneficiaries who reported having coverage but paid more than $250 out of pocket and reported no insurance payment, while MEPS leaves these beneficiaries in the covered category. The effect is to raise the MEPS estimates of out-of-pocket spending for covered people and to lower the estimates for noncovered people. This effect does not appear to be very large.
In combination, these factors mean that MEPS data show larger utilization and spending differences between covered and noncovered beneficiaries than those shown in the MCBS data used in chapter 2. This would merely reinforce the key point of that chapter, that insurance has an important effect on use of prescription drugs. The MEPS data would also show a larger proportion of covered beneficiaries, and a smaller proportion of noncovered ones, receiving any prescription drugs at all.
MEPS and MCBS also differ in their estimates of out-of-pocket spending by Medicare beneficiaries. They are close in their estimates of the proportion of total spending that is paid out of pocket, 47 percent under MCBS and 50 percent under MEPS. However, MCBS finds beneficiaries with coverage paying about 33 percent of their own expenses, compared to 43 percent under MEPS. This difference is partially attributable to the differences in coverage estimates described above. However, the difference persists for groups, such as those with employer coverage, for whom the MEPS and MCBS coverage estimates are quite close. Further investigation will be needed to fully account for this difference. Use of the MEPS data would have shown smaller differences in out- of-pocket spending for people with and without coverage than are shown in chapter 2. As a corollary, however, MEPS data show that many people who have coverage have insurance that leaves them exposed to high out-of-pocket costs.
MEPS Price Data
It should be emphasized that, while some aspects of the MEPS method of assigning coverage may introduce potential biases into its estimates of relative utilization by covered and noncovered beneficiaries, there is no reason to believe that chapter 3 overstates the price differences for the two groups. It was noted earlier that, while MEPS treats as having drug coverage people who failed to report coverage but had a third-party payment, there was no way of identifying people who failed to report their coverage and who received no drugs during the year. For the purpose of pricing comparisons, the omission of the latter group from the covered category does not affect the results (since they had no prescription to price). There does remain the issue of treating all Medicaid beneficiaries and Medicare HMO enrollees as having drug coverage. Some noncovered people in these populations have been included in the covered category. However, because people without coverage routinely pay more for their prescriptions than people with coverage, the probable result is that the MEPS data understate rather than overstate price differentials for the covered and noncovered.
Endnotes
1 - The one exception was Medicare HMO coverage, which was identified through HCFA(now known as CMS) enrollment records.
2 -The MEPS estimates are adjusted for two reasons. First, the adjusted MEPS estimates can be directly compared to the MCBS estimates. Second, the unadjusted MEPS estimates would overstate the percentage of beneficiaries who have drug coverage.
3 - IMS Health data for 1996 indicate that about 33 percent of prescriptions are paid for fully in cash at the time they are filled.
4 - For each drug reported by a participant, MCBS must first establish exactly what drug was received and in what quantity. It then establishes, using First Data Bank data, an average wholesale price (AWP) for the specific drug. Finally, it must estimate the retail price charged for the prescription by applying a markup factor to the AWP. Factors vary according to the source of payment for the drug—cash, Medicaid, or other third party—and also according to the range of the wholesale unit price, because pharmacies tend to charge higher markups for drugs with a lower AWP.
5 - The difference in price estimates for beneficiaries with Medicaid is larger, because MCBS reduces prices to account for manufacturer rebates to states, while MEPS does not.
6 - This term includes any entity furnishing outpatient prescription drugs to noninstitutionalized individuals, including retail pharmacies, outpatient pharmacies within hospitals, HMOs, or other facilities, and mail-order suppliers.
Appendix C. State and Regional Variations
One area of inquiry that is not discussed in the main report is regional and state variation. Preliminary analysis from several different sources suggests that there are substantial regional and state variations in spending and utilization of prescription drugs and in prices faced by consumers. At this point, we can observe some differences, but within the limited time available for this report, it has not been possible to investigate those differences systematically or to offer explanations for them. This appendix describes some of the variation observed. All of the information presented on price variation is limited to the pharmacy level–no information was available on state and regional variation in rebates or discounts to large volume direct purchasers. Tests of statistical significance were not performed on observed differences. Further research is needed to draw informed conclusions on the factors influencing the observed geographic variation.
The discussion below uses data from several different sources. Drug coverage, use rates, and expenditure estimates are from MEPS and are only available at the regional level for 1996. The MCBS cannot be used for regional or state analysis because the sample was not drawn to be representative at the regional or state level. Data from IMS Health are used to compare price differences between cash and third party payers across states and regions in 1998. State-level data were obtained from IMS Health for 25 drugs at their most common form and strength. The drugs were chosen by combining the 20 most frequently prescribed drugs for the Medicare population based on 1996 MCBS data and the top 10 most frequently prescribed drugs for the total population in 1998 based on IMS Health data. Because there was some overlap in the two lists, the final number of drugs compared was 25.1 Among the 25 drugs, seventeen are brand name drugs and 8 are generic drugs. Comparisons between cash and third party payers across states and regions are based on the difference in price paid per unit at the retail pharmacy for each drug on the list. As with the analysis in Chapter 3, this analysis excludes the effect of rebates from manufacturers to PBMs or insurers – for which data were unavailable. While this list includes many common drugs, it is a relatively small sample of drugs to use for state comparisons. Thus, results using a different or larger number of drugs might be different than those shown here.
Coverage
Table C-1 shows variation in coverage levels by region using MEPS data on drug coverage for the civilian, noninstitutionalized population. The highest coverage rates are in the Midwest and Northeast with the lowest coverage rate in the South. The pattern observed is similar to analyses of general health insurance coverage elsewhere.
| Region | Percent with drug coverage |
|---|---|
| National | 77.0% |
| Northeast | 78.6% |
| Midwest | 80.6% |
| South | 73.7% |
| West | 76.9% |
| Source: Center for Cost and Financing Studies, Agency for Healthcare Research and Quality: Medical Expenditure Panel Survey Household Component, 1996 | |
Utilization and Spending
In Chapter 2, various data were presented that showed spending differences between individuals with and without insurance coverage for prescription drugs. Here we consider those same comparisons within regions. Because size of prescription may vary due to differences in local medical practice patterns and variations in state Medicaid regulations on prescription size, neither average number of prescriptions nor average prescription prices are particularly good measures for regional comparisons. Rather, we compare spending and use rates by region using MEPS data.
Table C-2 shows that expenditures on prescription drugs vary somewhat between geographic regions. Residents of the Midwest and South appear to have the highest average drug spending per person while spending for residents in the West and Northeast appears somewhat lower. Generally, we would expect average spending to be higher where coverage is higher, yet the South has the lowest coverage rate and relatively higher spending than two other regions. Use rates – percent of people with at least one prescription – vary in a similar pattern to average drug spending.
| Region | Average Rx Spending per Capita | Average Spending for Those With Coverage | Average Spending for Those Without Coverage | Percent With at Least One Prescription | Average Rx Spending per User | Average Health Spending |
|---|---|---|---|---|---|---|
| National | $265 | $315 | $103 | 65.2% | $406 | $2081 |
| Northeast | $232 | $284 | $74 | 64.5% | $360 | $2228 |
| Midwest | $305 | $341 | $120 | 68.0% | $448 | $2435 |
| South | $285 | $333 | $132 | 66.2% | $431 | $1947 |
| West | $220 | $282 | $61 | 61.3% | $358 | $1795 |
| Source: Center for Cost and Financing Studies, Agency for Healthcare Research and Quality: Medical Expenditure Panel Survey Household Component, 1996 | ||||||
The difference in average total spending on drugs for those with and without coverage varies by region as well. Nationally, the uncovered spend about a third as much as those with coverage for prescription drugs. In each region, a similar relationship persists. But the magnitude of the difference varies. In the South, those without coverage spend about 40 percent as much as those with coverage, while in the West, those without coverage spend only 22 percent as much as those with coverage.
Interestingly, drug spending seems to vary somewhat differently across regions than total health spending. Although the West is the lowest on both measures and the Midwest is the highest, the average total health spending in the South is relatively lower while average drug spending is relatively higher and the opposite is true for the Northeast.
As discussed in Chapter 2, total drug spending is a combination of the price, volume and mix of drugs used. Further analysis will be needed to determine the role these factors play in regional differences in prescription drug spending.
Prescription Drug Prices
Given the national nature of many of the major players in the prescription drug market—drugs are produced by national manufacturers, distributed through mostly national PBMs and in many cases through national pharmacy chains—we might not expect to see large price differences by region. We would expect to see small differences based on local price variations. But in fact, we see a fairly substantial price variation across the country.
The key research question for Chapter 3 was the extent and direction of retail price differences between cash customers and those with third party coverage. Here we focus again on that distinction and we find some striking state and regional differences. As in Chapter 3, this analysis considers only differences between retail prices and does not consider manufacturer rebates that PBMs typically receive. Because rebate information is proprietary, no information is available on whether rebate amounts vary by region or state. Retail price differences also do not capture discounts that other large volume direct purchasers may receive. If these rebates and other discounts were included in this analysis, the differences observed would be larger.
Table C-3 shows the median price differential between cash customers and third party payers across states and regions based on IMS Health data for the 25 drugs described above. As in Chapter 3, the term cash customer refers to those paying in full for their prescriptions at the retail counter and includes those with indemnity insurance coverage who file claims at a later time. Median retail price differences for the 25 listed drugs are nearly twice as large in the Northeast and West as in the Midwest or South. The District of Columbia (21.6%) and New York (17.7%) had the highest median retail price difference (excluding rebates) among the states. Nebraska (-0.9%) was the only state where the retail price for third party payers was typically lower than that for cash payers. Wisconsin (0.3%), New Mexico (0.7%), and Florida (1.3%) were the next lowest states. Table C-3 also shows the number of drugs that fall into four ranges of price differentials: less than 0 (third party greater than cash), 0 to 9.9 percent, 10 to 19.9 percent and 20 percent or above. Looking at the differences in numbers of drugs falling in the four ranges shows substantial variation in price differentials within states as well as across states.2
The reasons for the substantial geographic variations have not been determined. Analysis of systematic sources of variation based on simple measures of managed care penetration and other variables showed no obvious relationships. In addition, the relationship between the share of the retail market attributable to cash customers (discussed below) and price differences is not clear. One hint may come in limited evidence that there is greater state variation in the prices charged to cash customers than in the prices charged to third party payers (data not shown). Further study of these differences, however, will be needed to understand state and regional variations.
MEPS data comparing average percent over benchmark price across all drug purchases in 1996 for those with and without drug coverage are shown by region in Figure C-1.3 The difference in average percent over benchmark price appears to be lowest in the Northeast where the average percent over benchmark price for those without coverage is 39 percent compared to 31 percent for those with coverage. The difference in prices relative to the benchmark price is much greater in the other three regions. This contrasts with the IMS results where the data show larger differences in the Northeast. Interestingly, the average percent over benchmark price is similar for Medicaid recipients and those with other non-Medicaid coverage in the Northeast and West, but in the Midwest and South average percent over benchmark price for Medicaid recipients is much closer to the higher levels shown for those without coverage (Medicaid data not shown). As discussed in Chapter 3, rebates from manufacturers to the states are not included in these data.
Figure C-1. Average Percent Over Benchmark Price, By Region
Note: (1) Percent over benchmark price equals the ratio of the average retail unit price (ARUP) to the benchmark unit price (BUP) minus one, multiplied by 100: Percent over benchmark = ((ARUP/BUP- 1)*100.
(2) Data exclude the effect of rebates for those with drug coverage.
Source: Center for Cost and Financing Studies, Agency for Healthcare Research and Quality: Medical Expenditure Panel Survey Household Component, 1996.
Cash Share of the Market
Cash payments have accounted for a declining share of the retail market for prescription drugs, as noted in Chapter 3. The share of prescriptions for which the purchaser pays in cash at the time of the transaction has declined from 63 percent in 1990 to 25 percent in 1998.4 One possible explanation for some of the differences just observed could be state and regional variation in the shares of the market attributed to cash payment (which here includes those with indemnity coverage where payment is not made at the point of sale).
Table C-3 also shows cash shares by state for 1998 from IMS data published by the National Association of Chain Drug Stores.5 States in the Northeast have relatively lower shares of cash payments – including two of the three lowest cash-share states: Rhode Island (13.4%) and New York (14.8%). Among the other three regions, the share of cash transactions averages about 26 percent. There is a great deal of variation across states in these regions, however. For example, the two states with the highest share of cash payments–North Dakota (44%) and South Dakota (42%)–are both in the Midwest as is Michigan (14.3%), the state with the second lowest cash share. It is likely that differences in cash share are related to differences in levels of insurance coverage and penetration of PBMs and other types of managed pharmacy benefits.
| State and Region | Median Price Difference | Distribution of Price Differences | Cash Share | |||
|---|---|---|---|---|---|---|
| <0 | 0-9.9 | 10-19.9 | >=20 | |||
| National | 7.4% | 6 | 12 | 4 | 3 | |
| Northeast | 11.4% | 2 | 7 | 13 | 3 | |
| Connecticut | 14.6% | 2 | 7 | 11 | 5 | 21.2% |
| Maine | 8.2% | 0 | 16 | 3 | 6 | 21.0% |
| Massachusetts | 12.8% | 8 | 4 | 5 | 8 | 15.1% |
| New Hampshire | 10.6% | 1 | 9 | 8 | 7 | 21.8% |
| New Jersey | 9.6% | 2 | 13 | 7 | 3 | 16.6% |
| New York | 17.7% | 0 | 1 | 14 | 10 | 14.8% |
| Pennsylvania | 7.6% | 4 | 14 | 3 | 4 | 14.6% |
| Rhode Island | 8.8% | 2 | 11 | 8 | 4 | 13.4% |
| Vermont | 10.7% | 1 | 9 | 9 | 6 | 23.6% |
| Midwest | 4.5% | 6 | 13 | 3 | 3 | |
| Illinois | 4.1% | 8 | 10 | 4 | 3 | 24.6% |
| Indiana | 5.4% | 6 | 13 | 3 | 3 | 25.2% |
| Iowa | 2.5% | 5 | 15 | 1 | 4 | 31.8% |
| Kansas | 4.3% | 4 | 14 | 2 | 5 | 34.7% |
| Michigan | 11.2% | 1 | 9 | 10 | 5 | 14.3% |
| Minnesota | 6.1% | 4 | 14 | 4 | 3 | 20.1% |
| Missouri | 3.0% | 6 | 13 | 3 | 3 | 23.4% |
| Nebraska | -0.9% | 15 | 6 | 2 | 2 | 28.1% |
| North Dakota | 6.1% | 1 | 17 | 2 | 5 | 43.8% |
| Ohio | 5.8% | 5 | 14 | 4 | 2 | 18.8% |
| South Dakota | 5.0% | 2 | 16 | 3 | 4 | 41.5% |
| Wisconsin | 0.3% | 12 | 9 | 2 | 2 | 22.0% |
| South | 5.1% | 5 | 13 | 4 | 3 | |
| Alabama | 5.9% | 2 | 16 | 2 | 5 | 26.0% |
| Arkansas | 5.3% | 3 | 16 | 2 | 4 | 34.2% |
| District of Columbia | 21.6% | 0 | 1 | 10 | 14 | 20.0% |
| Delaware | 11.1% | 0 | 9 | 10 | 6 | 18.7% |
| Florida | 1.3% | 11 | 10 | 3 | 1 | 24.3% |
| Georgia | 8.5% | 0 | 14 | 5 | 6 | 26.1% |
| Kentucky | 4.9% | 5 | 14 | 3 | 3 | 22.3% |
| Louisiana | 4.3% | 4 | 16 | 3 | 2 | 25.0% |
| Maryland | 10.8% | 1 | 10 | 9 | 5 | 15.9% |
| Mississippi | 6.2% | 1 | 18 | 2 | 4 | 27.6% |
| North Carolina | 9.9% | 0 | 13 | 9 | 3 | 28.5% |
| Oklahoma | 4.4% | 3 | 16 | 2 | 4 | 28.7% |
| South Carolina | 8.3% | 0 | 15 | 5 | 5 | 26.1% |
| Tennessee | 5.5% | 5 | 14 | 3 | 3 | 23.9% |
| Texas | 3.8% | 6 | 14 | 3 | 2 | 23.8% |
| Virginia | 7.9% | 4 | 14 | 4 | 3 | 19.9% |
| West Virginia | 4.8% | 7 | 12 | 3 | 3 | 19.1% |
| West | 11.4% | 2 | 9 | 9 | 5 | |
| Alaska | 7.5% | 0 | 16 | 6 | 3 | 30.2% |
| Arizona | 9.9% | 5 | 8 | 4 | 8 | 23.0% |
| California | 15.6% | 2 | 4 | 10 | 9 | 20.2% |
| Colorado | 7.9% | 5 | 9 | 6 | 5 | 22.4% |
| Hawaii | 11.3% | 2 | 9 | 10 | 4 | 18.6% |
| Idaho | 6.6% | 2 | 16 | 3 | 4 | 28.3% |
| Montana | 5.8% | 2 | 15 | 6 | 2 | 40.0% |
| Nevada | 10.7% | 2 | 9 | 9 | 5 | 21.6% |
| New Mexico | 0.7% | 12 | 8 | 3 | 2 | 23.2% |
| Oregon | 8.2% | 2 | 14 | 3 | 6 | 24.4% |
| Utah | 10.6% | 1 | 11 | 6 | 7 | 21.3% |
| Washington | 5.5% | 4 | 14 | 5 | 2 | 24.5% |
| Wyoming | 6.0% | 4 | 14 | 2 | 5 | 38.3% |
|
Note: Data exclude the effect of rebates for those with drug coverage. Source: IMS Health,1998 |
||||||
Endnotes
1 - Claritin, Premarin, Zantac, Prozac, Trimox, Prilosec, Lanoxin, Hydrocodone w/APAP, Norvasc, Lipitor, Furosemide, Synthroid, Coumadin, Vasotec, Atenolol, Lasix, Triamterene/HCTZ, Cardizem CD, Hydrochlorothiazide, Zestril, Prednisone, K-Dur, Zocor, Procardia XL, and Glyburide.
2 - Analysis using a different set of drugs might yield different results at the state level.
3 - Recall from Chapter 3 that the percent over benchmark price equals the ratio of the average retail unit price (ARUP) to the benchmark unit price (BUP) minus one, multiplied by 100.
4 - The National Association of Chain Drug Stores, “The Chain Pharmacy Industry Profile.” 1999. Based on data from IMS Health.
5 - Ibid.
